👉Rodrigo Arrangóiz MS, MD, FACS collaborated in the COVIDSurg-Cancer study published in the Journal of Clinical Oncology, which highlights the importance of having hospitals with protocols and “COVID-free routes” for the surgical care of cancer patients.
👉Millions of surgeries in the world have been postponed due to the pandemic.
👉Surgery for many conditions must continue despite the threat of a second wave.
👉Hospital areas with “COVID-19 free routes” should be established to allow surgery to be carried out safely.
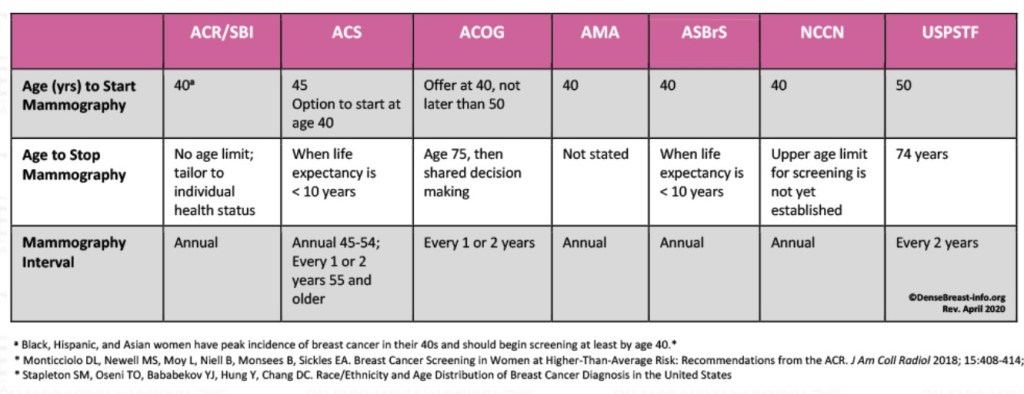
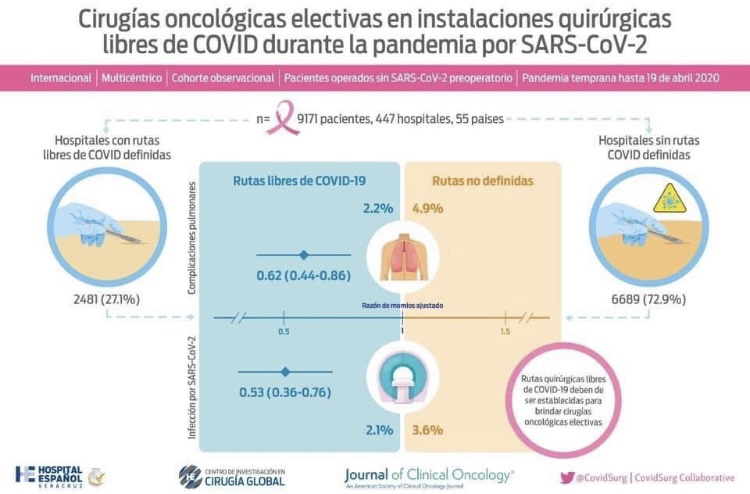
👉Data from 9,171 patients from 55 countries were examined since the onset of the pandemic.
👉Pulmonary complications (2.2% vs 4.9%) and post-op mortality (0.7% vs 1.7%) were lower in patients treated in hospitals with protocols and “COVID-19 free routes”.
👉Surgical care must continue during the pandemic. Hospitals must have protocols for the care of COVID and NON-COVID patients, detection programs for SARS-CoV-2 infection in asymptomatic personnel, and differentiated routes.
👉Hospitals without protocols represent a higher risk of complications and mortality for patients undergoing cancer surgeries. The Hospital Español de Veracruz has the COVID SAFE program to protect our patients and continue safe surgical care.
👉Full article: 👉🏼 https://ascopubs.org/doi/pdf/10.1200/JCO.20.01933
#Arrangoiz #CancerSurgeon #BreastSurgeon #ThyroidSurgeon #ParathyroidSurgeon #HeadandNeckSurgeon #SurgicalOncologist #CASO #CenterforAdvancedSurgicalOncology #PalmettoGeneralHospital