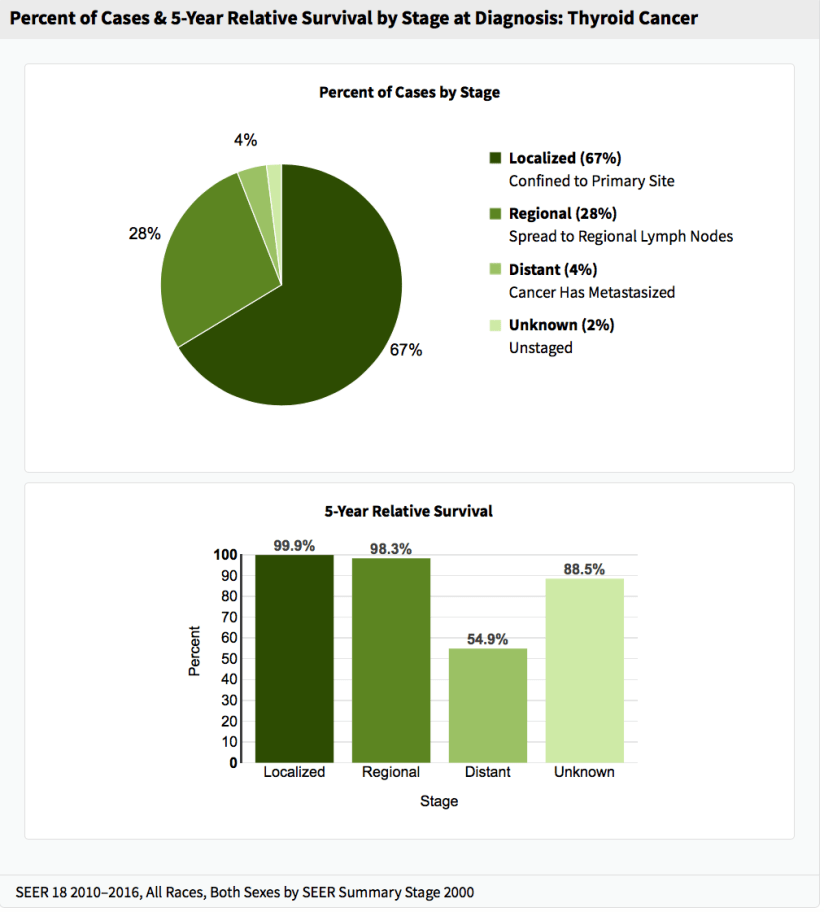
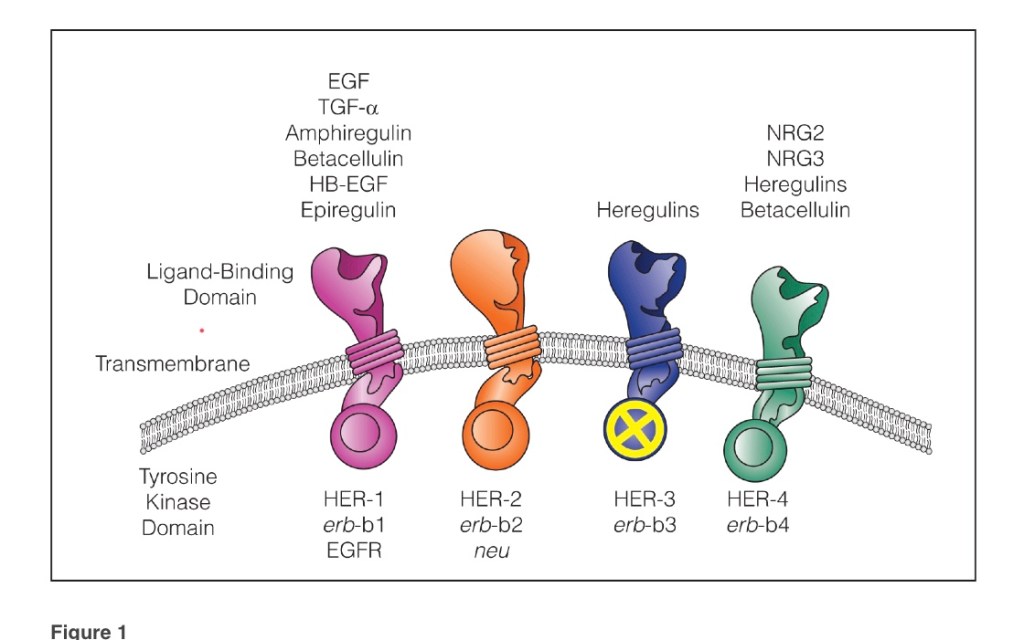
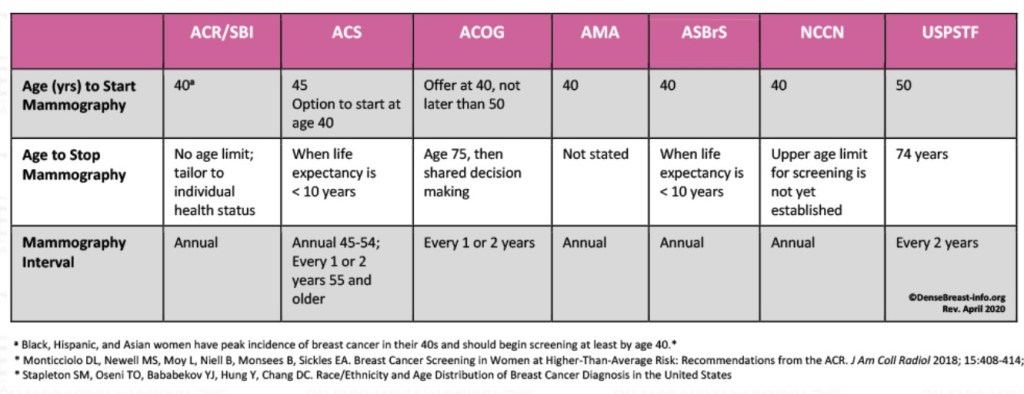
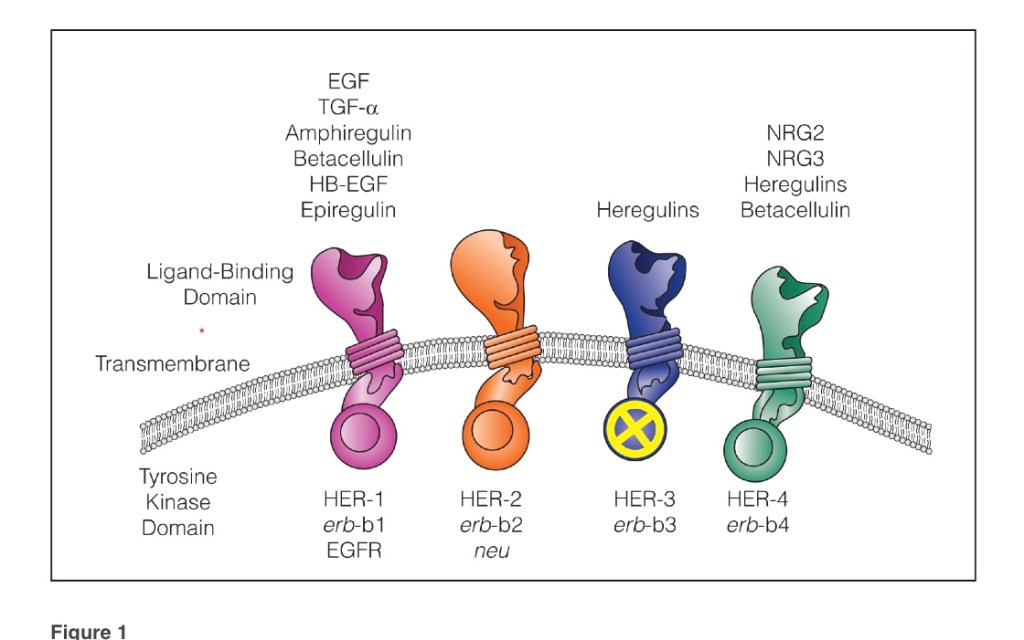
- The Cancer and Leukemia Group B (CALGB) 9343 trial:
- Enrolled 647 patients:
- From 1994 until 1999
- Long-term follow-up data:
- Were published in 2013:
- With a median follow-up of:
- 12.6 years
- With a median follow-up of:
- Women age 70 years or older:
- With clinical stage I:
- cT1, cN0, cM0
- ER-positive breast cancer
- Treated by lumpectomy
- With clinical stage I:
- Were randomly assigned to receive:
- Tamoxifen plus radiation therapy (TamRT) or Tamoxifen alone (Tam)
- At 10 years:
- 98% of women:
- Receiving TamRT were free from local and regional recurrences
- Compared to 91% of those receiving Tam
- 98% of women:
- The 10-year estimates of overall survival (OS) were:
- 67% (95% confidence interval [CI], 62–72%) in the TamRT group versus
- 66% (95% CI, 61%–71%) in the Tam group:
- But the difference was not statistically significant
- In addition to concluding that:
- While RT (in addition to Tam):
- Reduces locoregional recurrence:
- The authors noted that “the impact of breast cancer in this select group of older women is much smaller than that of comorbid conditions”:
- Only 3% of women in study have died as a result of breast cancer whereas 49% have died as a result of other causes
- The authors noted that “the impact of breast cancer in this select group of older women is much smaller than that of comorbid conditions”:
- Reduces locoregional recurrence:
- While RT (in addition to Tam):
- Were published in 2013:
- Enrolled 647 patients:
- References:
- Hughes KS, Schnaper LA, Bellon JR, et al. Lumpectomy plus tamoxifen with or without irradiation in women age 70 years or older with early breast cancer: long-term follow-up of CALGB 9343. J Clin Oncol. 2013;31:2382-2389
#Arrangoiz #BreastSurgeon #CancerSurgeon #SurgicalOncologist #CASO #CenterforAdvancedSurgicalOncology #BreastCancer