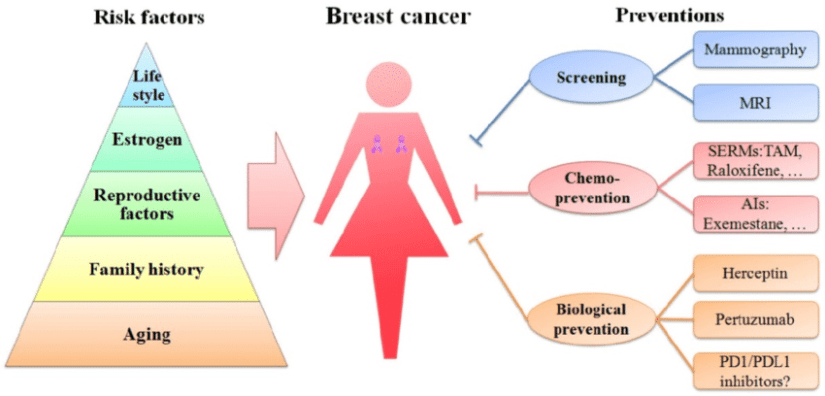
- Breast awareness:
- Starting at age 18 years:
- Women should be familiar with their breasts and promptly report changes to their health care provider:
- Periodic, consistent breast self-exam (BSE) may facilitate breast self-awareness
- Premenopausal women may find BSE most informative when performed at the end of menses
- Periodic, consistent breast self-exam (BSE) may facilitate breast self-awareness
- Women should be familiar with their breasts and promptly report changes to their health care provider:
- Starting at age 18 years:
- Clinical breast exam, every 6 to 12 months:
- Starting at age 25 years:
- Randomized trials comparing clinical breast exam versus no screening have not been performed:
- Rationale for recommending clinical breast exam every 6 to 12 months is the concern for interval breast cancers
- Randomized trials comparing clinical breast exam versus no screening have not been performed:
- Starting at age 25 years:
- Breast screening:
- Age 25 to 29 years:
- Annual breast MRI screening with contrast:
- or Mammogram with consideration of tomosynthesis:
- Only if MRI is unavailable:
- Breast MRI is preferred due to the theoretical risk of radiation exposure in pathogenic /likely pathogenic variant carriers
- Only if MRI is unavailable:
- or Mammogram with consideration of tomosynthesis:
- Individualized based on family history if a breast cancer diagnosis before age 30 is present
- Annual breast MRI screening with contrast:
- Age 30 to 75 years:
- Annual mammogram with consideration of tomosynthesis and breast MRI screening with contrast:
- The criteria for high-quality breast MRI include:
- A dedicated breast coil
- The ability to perform biopsy under MRI guidance
- Radiologists experienced in breast MRI
- Regional availability
- Breast MRI is preferably performed on:
- Days 7 to 15 of a menstrual cycle for premenopausal women
- The criteria for high-quality breast MRI include:
- Annual mammogram with consideration of tomosynthesis and breast MRI screening with contrast:
- Age greater than 75 years:
- Management should be considered on an individual basis
- For women with a BRCA pathogenic / likely pathogenic variant who are treated for breast cancer and have not had a bilateral mastectomy:
- Screening with annual mammogram with consideration of tomosynthesis and breast MRI should continue as described above
- The appropriateness of imaging modalities and scheduling is still under study. Lowry KP, Lee JM, Kong CY, et al. Cancer 2012;118:2021-2030.
- Lehman CD, Lee JM, DeMartini WB, et al. Screening MRI in women with a personal history of breast cancer. J Natl Cancer Inst 2016;108.
- Screening with annual mammogram with consideration of tomosynthesis and breast MRI should continue as described above
- Age 25 to 29 years:
- Discuss option of risk-reducing mastectomy:
- Counseling should include a discussion regarding:
- Degree of protection
- Reconstruction options, and risks
- In addition, the family history and residual breast cancer risk with age and life expectancy should be considered during counseling
- Counseling should include a discussion regarding:
- Recommend risk-reducing salpingo-oophorectomy (RRSO):
- Given the high rate of occult neoplasms:
- Special attention should be given to sampling and pathologic review of the ovaries and fallopian tubes
- Typically between 35 and 40 years, and upon completion of child bearing
- Because ovarian cancer onset in patients with BRCA2 pathogenic / likely pathogenic variants is an average of 8 to 10 years later than in patients with BRCA1 pathogenic / likely pathogenic variants:
- It is reasonable to delay RRSO for management of ovarian cancer risk:
- Until age 40 to 45 years in patients with BRCA2 pathogenic / likely pathogenic variants unless age at diagnosis in the family warrants earlier age for consideration of prophylactic surgery.
- Counseling includes a discussion of:
- Reproductive desires
- Extent of cancer risk
- Degree of protection for breast and ovarian cancer
- Management of menopausal symptoms
- Hormone replacement therapy, and related medical issues
- Salpingectomy alone is not the standard of care for risk reduction, although clinical trials of interval salpingectomy and delayed oophorectomy are ongoing:
- The concern for risk-reducing salpingectomy alone is that women are still at risk for developing ovarian cancer
- In addition, in premenopausal women, oophorectomy likely reduces the risk of developing breast cancer but the magnitude is uncertain and may be gene-specific
- It is reasonable to delay RRSO for management of ovarian cancer risk:
- Given the high rate of occult neoplasms:
- Limited data suggest that there may be a slightly increased risk of serous uterine cancer among women with a BRCA1 pathogenic / likely pathogenic variant.
- The clinical significance of these findings is unclear
- Further evaluation of the risk of serous uterine cancer in the BRCA population needs to be undertaken
- The provider and patient should discuss the risks and benefits of concurrent hysterectomy at the time of RRSO for women with a BRCA1 pathogenic / likely pathogenic variant prior to surgery
- Women who undergo hysterectomy at the time of RRSO are candidates for estrogen alone hormone replacement therapy, which is associated with a decreased risk of breast cancer compared to combined estrogen and progesterone, which is required when the uterus is left in situ (Chlebowski R, et al. JAMA Oncol 2015;1:296-305)
- Address psychosocial and quality-of-life aspects of undergoing risk-reducing mastectomy and /or salpingo-oophorectomy
- For those patients who have not elected RRSO:
- Transvaginal ultrasound combined with serum CA-125 for ovarian cancer screening:
- Although of uncertain benefit, may be considered at the clinician’s discretion starting at age 30 to 35 years
- Transvaginal ultrasound combined with serum CA-125 for ovarian cancer screening:
- Consider risk reduction agents as options for breast and ovarian cancer, including discussion of risks and benefits
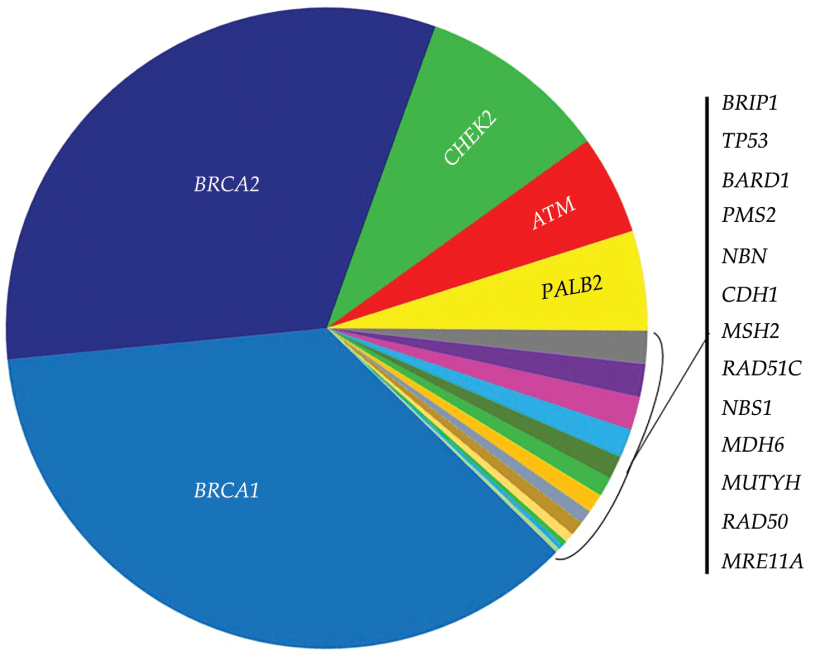
#Arrangoiz #CancerSurgeon #BreastSurgeon #BreastCancer #BRCA1 #BRCAPositiveBreastCancer #SurgicalOncologist #CASO #CenterforAdvancedSurgicalOncology