- Fibroadenomas of the breast are benign tumors:
- Composed of stromal and epithelial elements:
- That are commonly seen in young women
- Multiple or complex fibroadenomas:
- May indicate a slightly increased risk for breast cancer:
- The relative risk of breast cancer in patients with such fibroadenomas:
- Is approximately twice that of patients of similar age without fibroadenomas
- A patient’s age determines the preferred imaging method:
- In general, ultrasonography (US) is preferred:
- If a palpable mass is found
- If a patient is younger than 30 years
- If the patient is pregnant
- Mammography and US are both useful if the patient has:
- A palpable mass
- Is older than 30 years
- Is not pregnant
- In patients younger than 30 years:
- The most appropriate modality is ultrasound:
- Because the patient is spared radiation exposure and because the likelihood for fibroadenoma is high
- Mammography is not indicated as the primary imaging study in women younger than 30 years:
- Unless high-risk factors are present
- Computed tomography (CT) scanning:
- Is not initially indicated for assessing a palpable lump in a woman in women younger than 30 years:
- Because of radiation exposure
- The inability of CT to demonstrate micro-calcifications
- The lack of specificity in the findings
- Magnetic resonance imaging (MRI):
- Is not initially indicated for assessing a palpable lump in women younger than 30 year:
- Mainly because of its high cost and the high likelihood of false-positive findings
- Positron emission tomography:
- Is expensive and is not universally available
- On mammograms:
- Fibroadenomas typically appear as:
- Circumscribed oval or round masses:
- Which occasionally have coarse calcifications
- Circumscribed oval or round masses:
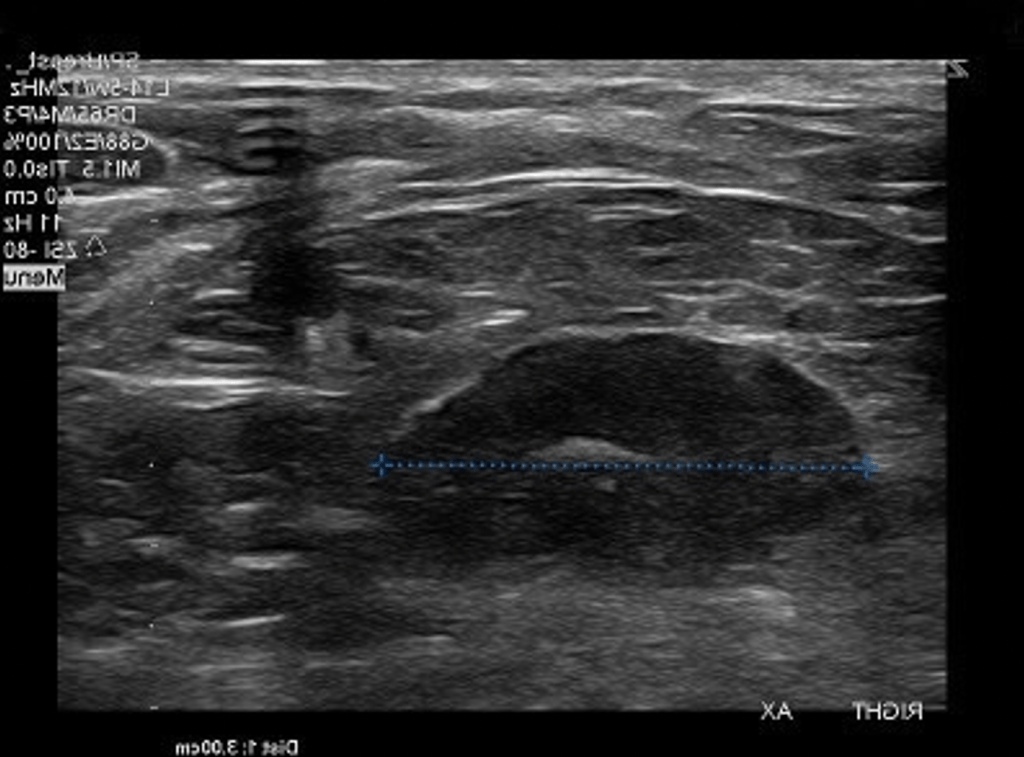
- On ultrasonograms:
- Fibroadenomas appear as:
- Circumscribed, homogeneous, oval, hypoechoic masses:
- That may have gentle lobulations
- A smooth, thin, echogenic capsule
- Variable acoustic enhancement; and homogeneity
- Circumscribed, homogeneous, oval, hypoechoic masses:
- On MRI:
- Fibroadenomas typically appear as smooth masses with high signal intensity on T2-weighted images and enhancement with the administration of gadolinium-based contrast agent
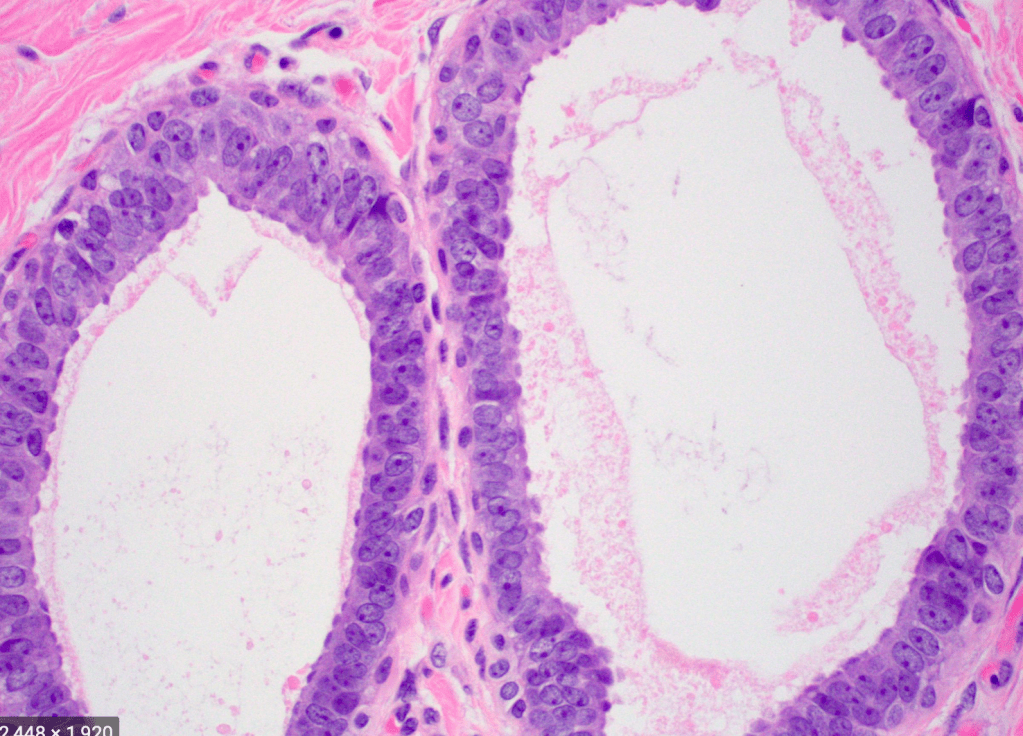
- Fibroadenoma:
- Is a common benign breast lesion:
- Results from the excess proliferation of connective tissue
- Fibroadenomas characteristically contain both:
- Stromal and epithelial cells
- Epidemiology:
- They usually occur in women:
- Between the ages of 10 and 40 years
- It is the most common breast mass:
- In the adolescent and young adult population :
- Their peak incidence is between:
- 25 and 40 years
- The incidence decreases after 40 years
- Clinical presentation:
- The typical presentation is in a woman of reproductive age:
- With a mobile palpable breast lump:
- Due to their hormonal sensitivity:
- Fibroadenomas commonly enlarge during pregnancy and involute at menopause:
- Hence, they rarely present after the age of 40 years
- Fibroadenomas commonly enlarge during pregnancy and involute at menopause:
- The lesions are well defined and well-circumscribed clinically and the overlying skin is normal
- The lesions are not fixed to the surrounding parenchyma and slip around under the palpating fingers:
- Hence the colloquial term a breast “mouse”
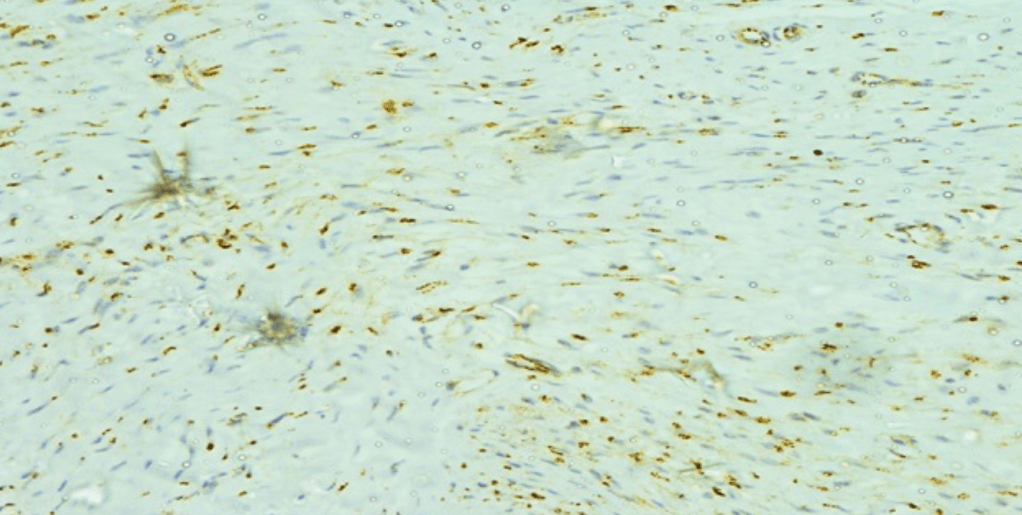
- Pathology:
- A fibroadenoma is a type of adenomatous breast lesion:
- It contains epithelium:
- Has minimal malignant potential
- It contains epithelium:
- Multiple fibroadenomas occur in:
- 10% to 15% of patients:
- Patients with multiple fibroadenomas:
- Tend to have a strong family history of these tumors
- They are assumed to be:
- Aberrations of normal breast development (ANDI) or the product of hyperplastic processes:
- Rather than true neoplasms
- 10% to 15% of patients:
- Fibroadenomas can be stimulated by estrogen and progesterone:
- Some fibroadenomas also have receptors and respond to:
- Growth hormone and epidermal growth factor
- Some fibroadenomas also have receptors and respond to:
- When found in an adolescent girl:
- The term juvenile fibroadenoma is more appropriate
- Location:
- Although they can be located anywhere in the breast:
- There may be a predilection for the upper outer quadrant
- Associations:
- Cyclosporin use o Cowden syndrome
- Radiographic features:
- Mammography:
- Fibroadenomas have a spectrum of features:
- Well-circumscribed discrete oval mass hypodense or isodense to the breast glandular tissue
- Mass with macro-lobulation or partially obscured margin
- Involuting fibroadenomas in older, typically postmenopausal patients may contain:
- Calcification:
- Often producing the classic, coarse popcorn calcification appearance
- In some cases the whole lesion is calcified
- Calcification may also present as crushed stone-like micro-calcification:
- Which makes differentiation from malignancy difficult
- Fibroadenomas have a spectrum of features:
- Breast ultrasound:
- Typically seen as a well-circumscribed, round to ovoid, or macro-lobulated mass with generally uniform hypoechogenicity
- Intralesional sonographically detectable calcification:
- May be seen in approximately 10% of cases
- Sometimes a thin echogenic rim (pseudo capsule) may be seen sonographically
- Breast MRI:
- T1: typically hypo intense or isointense compared with adjacent breast tissue
- T2: can be hypo- or hyper intense
- T1 C+ (Gd): can be variable but a majority will show slow initial contrast enhancement followed by a persistent delayed phase (type I enhancement curve)
- Non-enhancing internal septations may be seen
- Diagnosis:
- These lesions are easily biopsied under ultrasound guidance
- When a lesion has the typical features of a fibroadenoma on ultrasound and there are no clinical red flags:
- They can be safely followed clinically
- When lesions enlarge or have atypical imaging findings:
- Ultrasound-guided core biopsy is a minimally invasive outpatient procedure that will give a diagnosis with virtually no complications
- There may be a maximum diameter above which a biopsy should be done if no previous imaging is available:
- The reason for intervention based on size is that a phyllodes tumor may be indistinguishable from a fibroadenoma on ultrasound:
- A maximum diameter of 2.5 cm may be a useful benchmark for biopsy if you have no previous imaging
- Interval enlargement:
- Is an indication for biopsy
- Symptomatic, progressively enlarging masses or atypical presentations:
- May warrant surgical excision
- If a needle biopsy shows that a mass less than 2 centimeters in size is a fibroadenoma, with no other concerning features:
- It does not have to be surgically removed
- The patient’s core biopsy pathology demonstrating a fibroadenoma is consistent with the typical imaging findings of a smooth, round, hypoechoic mass:
- As the biopsy is concordant, no further intervention is needed:
- Follow-up for reassurance is acceptable
- Treatment and prognosis:
- They are benign lesions with minimal or no malignant potential
- The risk of malignant transformation is extremely low:
- Has been reported to range around 0.0125% to 0.3%
- Indications for biopsy include:
- Enlarging lesion
- Atypical findings on ultrasound
- A lesion above 2.5 cm and there are no previous studies for comparison
- Patient peace of mind:
- Some patients are simply not happy with a palpable mass in the breast without a histological diagnosis:
- This is a valid and reasonable indication for biopsy
- Some patients are simply not happy with a palpable mass in the breast without a histological diagnosis:
- References
- Tan BY, Tan PH A Diagnostic Approach to Fibroepithelial Breast Lesions. Surg Pathol Clin. 2018 Mar;11(1):17-42.
- American Society of Breast Surgeons – benign breast disease. January 8, 2018. Choosing Wisely website. http://www.choosingwisely.org/clinician-lists/asbrs-benign-breast-disease-biopsy-proven-fibroadenomas-smaller-than-2-cm/. Accessed October 17, 2019.
#Arrangoiz #CancerSurgeon #BreastSurgeon #SurgicalOncologist #BreastFibroadenoma #Breast Cancer