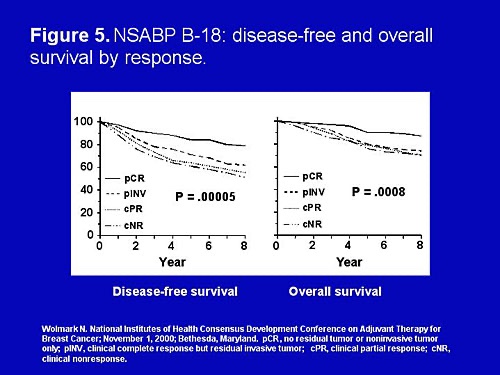
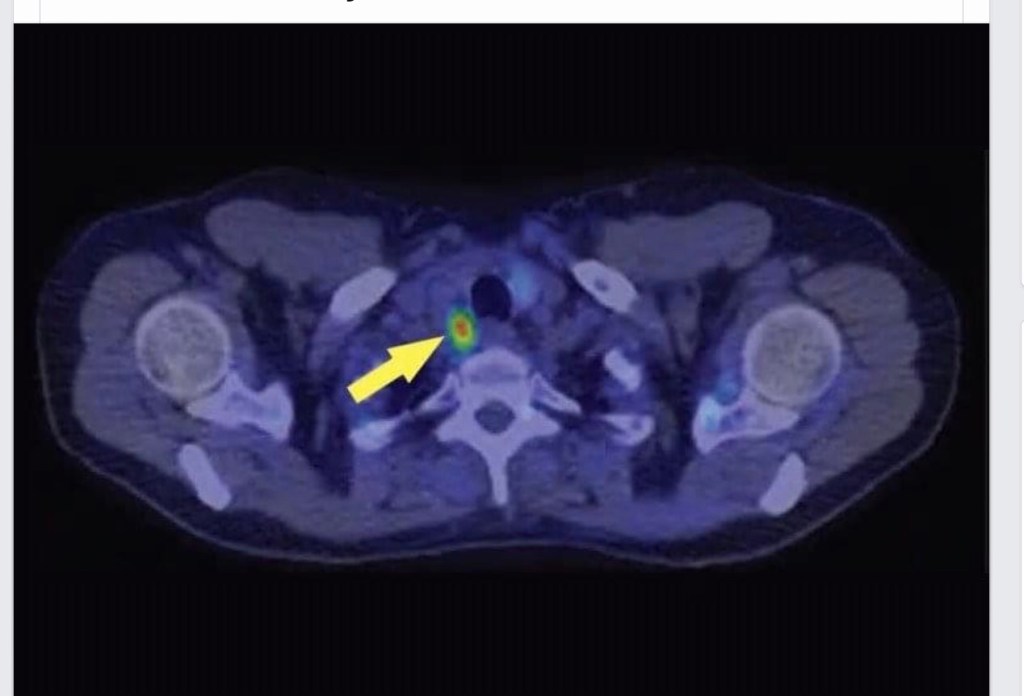
- Whole-breast irradiation (WBI) can be administered following delivery:
- Uterine shielding can reduce the dose but in non-life threatening situations:
- Radiation should be held until after delivery
- Uterine shielding can reduce the dose but in non-life threatening situations:
- Risks of radiation while pregnant can include:
- Toxicity to the fetus
- Increased risk of second malignancy in the child
- Although hypofractionated WBI has been studied and found to be comparable to standard WBI:
- Few patients enrolled were younger than age 50 years
- Current American Society for Radiation Oncology guidelines:
- Do not recommend:
- Hypofractionated WBI for patients younger than age 50 years
- Do not recommend:
- Accelerated partial breast irradiation:
- Has been shown to provide equivalent rates of local recurrence compared to standard WBI:
- However, due to a paucity of young patients enrolled in such trials:
- It is not currently recommended for patients younger age 50 years
- However, due to a paucity of young patients enrolled in such trials:
- Has been shown to provide equivalent rates of local recurrence compared to standard WBI:
- REFERENCES
- Fisher B, Anderson S, Bryant J, et al. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med. 2002;347:1233-1241.
- Haviland JS, Owen JR, Dewar JA, et al; START Trialists’ Group. The UK Standardisation of Breast Radiotherapy (START) trials of radiotherapy hypofractionation for treatment of early stage breast cancer: 10-year follow-up results of two randomised controlled trials. Lancet Oncol. 2013;14:1086-1094.
- Luis SA, Christie DR, Kaminski A, Kenny L, Peres MH. Pregnancy and radiotherapy: management options for minimizing risk, case series, and comprehensive literature review. J Med Imaging Radiat Oncol. 2009;53:559-568.
- Smith BD, Arthur DW, Buchholz TA, et al. Accelerated partial breast irradiation consensus statement from the American Society for Radiation Oncology (ASTRO). Int J Radiat Oncol Biol Phys. 2009;74:987-1001.
- Smith BD, Bentzen SM, Correa CR, et al. Fractionation for whole breast irradiation: an American Society for Radiation Oncology (ASTRO) evidence-based guideline. Int J Radiat Oncol Biol Phys. 2011;81:59-68.
- Strnad V, Ott OJ, Hildebrandt G, et al; Groupe Européen de Curiethérapie of European Society for Radiotherapy and Oncology (GEC-ESTRO). 5-year results of accelerated partial breast irradiation using sole interstitial multicatheter brachytherapy versus whole-breast irradiation with boost after breast-conserving surgery for low-risk invasive and in-situ carcinoma of the female breast: a randomised, phase 3, non-inferiority trial. Lancet. 2016;387:229-238.
- Whelan TJ, Pignol JP, Levine MN, et al. Long-term results of hypofractionated radiation therapy for breast cancer. N Engl J Med. 2010;362:513-520.
#Arrangoiz #BreastSurgeon #CancerSurgeon #SurgicalOncologist #BreastCancer