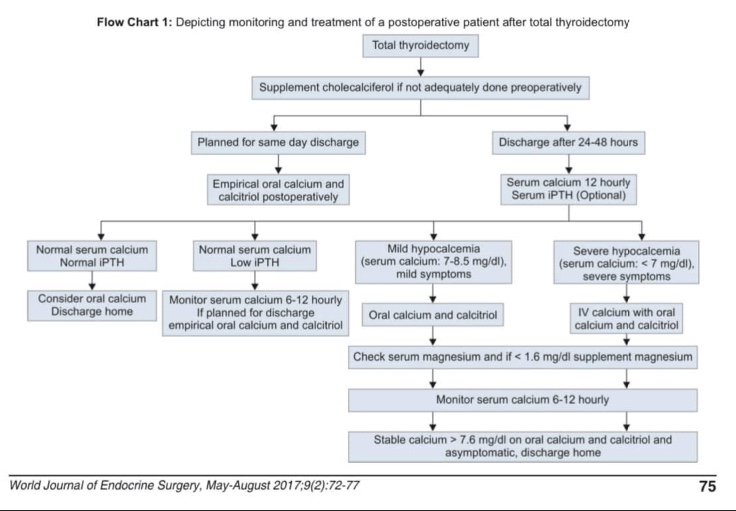
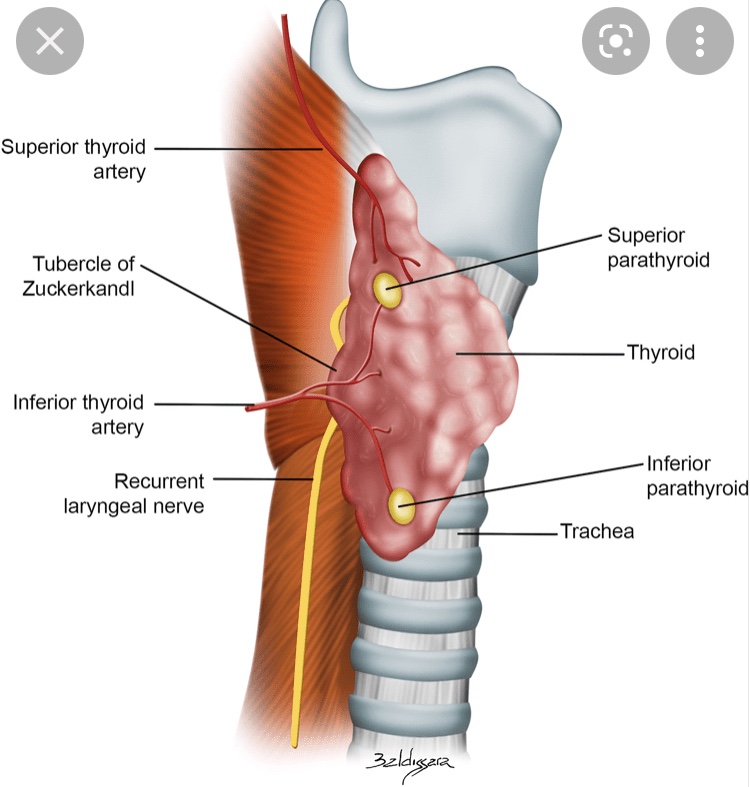
- Unlike many cancers, the risk of recurrence does not parallel the risk of mortality in differentiated thyroid cancer
- In most patients, the risk of recurrence far exceeds the risk of disease-specific mortality:
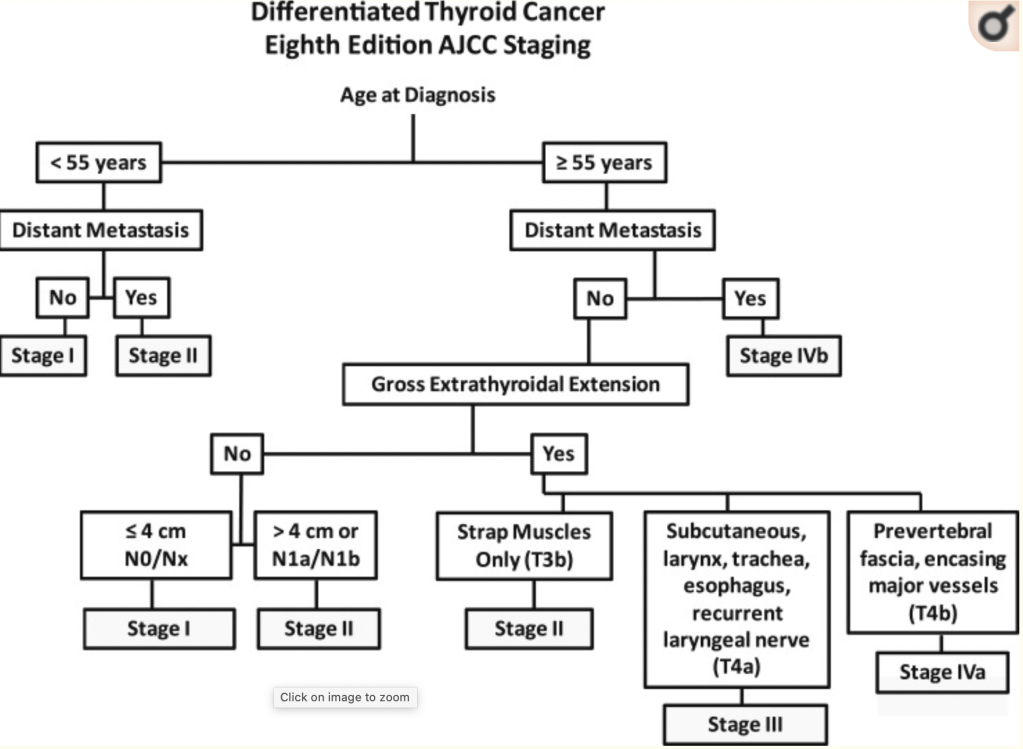
- Thus staging systems designed to predict mortality in thyroid cancer:
- Would not be anticipated to be predictive of disease recurrence
- Thus staging systems designed to predict mortality in thyroid cancer:
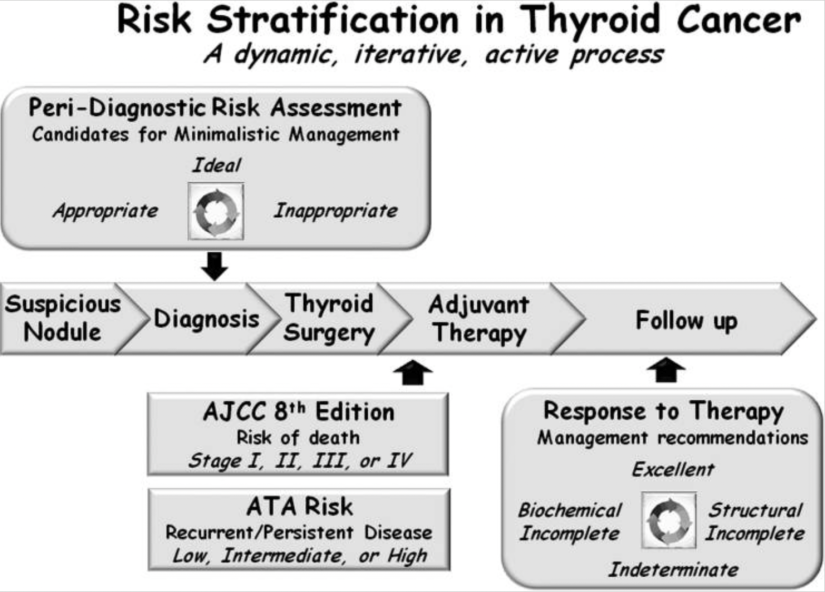
- To address this issue, a risk-stratification system:
- Was developed and validated to predict the risk of structural disease recurrence:
- Based on information obtained around the time of initial therapy
- A modified version of this original risk-stratification system was endorsed in the 2009 ATA guidelines and subsequently modified in the 2015 ATA guidelines
- Was developed and validated to predict the risk of structural disease recurrence:
- Whereas initially conceived as a three-category model of risk assessment:
- Low, intermediate, or high risk
- The ATA risk-stratification system:
- Is now visualized as a continuum of risk:
- Ranging from very low to very high risk of structural disease recurrence
- Is now visualized as a continuum of risk:

- The three-category model was proven to be very useful and reproducible across multiple studies
- The 2015 ATA guideline:
- Expanded the low-risk category to include not only intra-thyroidal papillary thyroid cancer but also patients with:
- Very small-volume lymph node micro-metastases:
- Less than 0.2 cm in largest dimension
- Intra-thyroidal well-differentiated follicular thyroid cancer:
- With capsular invasion and fewer than four foci of vascular invasion
- Intra-thyroidal encapsulated follicular variant of papillary thyroid carcinoma:
- Now known as noninvasive follicular thyroid neoplasm with papillary-like nuclear features
- Either unifocal or multifocal intra-thyroidal papillary micro-carcinoma:
- Even if they have known BRAFV600E mutations
- Very small-volume lymph node micro-metastases:
- The high-risk category was also expanded to include:
- Follicular cancer with more than four foci of vascular invasion and
- Pathologic lymph node metastasis:
- With any metastatic lymph node ≥ 3 cm in largest dimension
- The remaining tumors were classified as intermediate risk based on the data available at the time the guidelines were written
- Expanded the low-risk category to include not only intra-thyroidal papillary thyroid cancer but also patients with:

- The last several years have seen an abundance of published data confirming the association among specific molecular alterations, histological subtypes, and clinical outcomes in follicular cell-derived thyroid cancer
- Point mutations in BRAFV600E:
- Are associated with:
- Increased risk of recurrence
- Radioactive iodine refractoriness
- Extrathyroidal extension
- Lymph node metastases
- Disease-specific mortality
- Are associated with:
- Likewise, oncogenic genetic alterations in TERT promoter, TP53, EIF1AX, and β-catenin:
- Are associated with more aggressive tumor behavior and poorer clinical outcomes
- Furthermore, mutational combinations (such as BRAFV600E + TERT promoter mutations or RAS + TERT promoter mutations):
- Are associated with significantly increased risk beyond that associated with either mutation in isolation
- As shown in the ATA continuum of risk figure:
- Appropriate molecular risk stratification requires integration of the genetic abnormality into the proper clinical context:
- As the presence of a specific mutation does not always portend a poor prognosis:
- e.g., BRAFV600E mutations are found in > 50% of papillary micro-carcinomas:
- Which usually display an indolent clinical course
- e.g., BRAFV600E mutations are found in > 50% of papillary micro-carcinomas:
- As the presence of a specific mutation does not always portend a poor prognosis:
- Although not yet proven:
- It seems reasonable to consider either:
- More careful follow-up or potentially more aggressive therapies for tumors with the highest risk mutational profiles:
- Particularly those with mutational combinations associated with the poorest clinical outcome
- More careful follow-up or potentially more aggressive therapies for tumors with the highest risk mutational profiles:
- It seems reasonable to consider either:
- It is important to remember that there is no guarantee that more aggressive surgery, radioactive iodine therapy, thyroid-stimulating hormone suppression, or other systemic therapies:
- Will necessarily provide therapeutic benefit simply because we can identify a patient at high risk for poorer outcomes on the basis of clinic-pathological presentation or molecular profiling
- Prospective studies evaluating the impact of more aggressive surgical and systemic therapies in the setting of high-risk mutational profiles are needed
- Appropriate molecular risk stratification requires integration of the genetic abnormality into the proper clinical context:
- Point mutations in BRAFV600E:
- The ATA risk-stratification system performs well in clinical practice:
- With low-risk patients:
- Demonstrating no evidence of disease 80% to 90% of the time
- Biochemical incomplete responses 15% of the time
- Structural incomplete responses 3% to 5% of the time
- Intermediate-risk patients achieve:
- Excellent response ∼60% of the time
- Have a biochemical incomplete response ∼15% to 20% of the time,
- Structural incomplete response ∼20% of the time
- High-risk patients achieve:
- No evidence of disease status in less than 30% of the cases
- Structural incomplete response 50% to 75% of the cases
- Biochemical incomplete response 10% to 15% of the cases
- The studies contributing to these approximations are extensively reviewed in the ATA guidelines
- Interestingly:
- Age is a major determinant of response to therapy:
- In ATA high-risk patients:
- The proportion of excellent responders was found to be significantly higher among younger patients (age < 55 years) than among older patients (age ≥ 55 years; 40.3% vs 27.5%, P = 0.02)
- The proportion of structural incomplete responders was significantly larger among older patients than among younger patients (53% vs 33%, P = 0.002)
- Moreover, ATA high-risk younger patients with a structural incomplete response to therapy had a significantly better DSS than older patients (74% vs 12%, respectively, P < 0.001)
- In ATA high-risk patients:
- Age is a major determinant of response to therapy:
- With low-risk patients:
#Arrangoiz #ThyroidSurgeon #ThyroidExpert #ThyroidCancer #EndocrineSurgery #HeadandNeckSurgery #CancerSurgeon #SurgicalOncologist