- The base of the adult breast extends:
- Vertically:
- From the 2nd to 6th ribs
- Horizontally:
- The breast extends from the lateral border of the sternum to the midaxillary line:
- In some individuals, into the axilla itself
- The breast extends from the lateral border of the sternum to the midaxillary line:
- Vertically:
- The adult breast consists of:
- Glandular and adipose tissue:
- Together with a system of connecting ligaments
- Glandular and adipose tissue:

- 1. Nipple:
- This is located at the apex of the breast and projects up to 1 cm
- Optimizing its positioning is of utmost importance in breast surgery
- In the average adult female the nipples lie in the midclavicular line:
- 19 cm to 21 cm from the sternal notch and 9 cm to 11 cm from the midline:
- But their position varies widely according to shape, size and age
- 19 cm to 21 cm from the sternal notch and 9 cm to 11 cm from the midline:
- 2. Areola:
- This is a circular area of skin that surrounds the nipple
- Its color darkens during pregnancy due to the deposition of melanin
- The areolar skin contains Montgomery glands:
- Which secrete a protective oily lubricant

- 3. Glandular tissue:
- The glandular tissue is the functional component of the lactating breast and the site of milk production, which is passed to the nipple via a system of ducts:
- Each breast, or mammary gland:
- Contains 15 to 20 lobes and each lobe is comprised of 20 to 40 terminal ductal lobular units (TDLU):
- The TDLU is the functional unit of the breast
- Contains 15 to 20 lobes and each lobe is comprised of 20 to 40 terminal ductal lobular units (TDLU):
- Each breast, or mammary gland:
- The breast mound is roughly hemispherical
- The bulk of the glandular tissue is found in the upper outer quadrant:
- Which is the commonest site of malignancy.
- The glandular tissue is the functional component of the lactating breast and the site of milk production, which is passed to the nipple via a system of ducts:
- 4. Adipose tissue:
- This forms up to 70% of the breast mass:
- It is the main determinant of breast size
- This forms up to 70% of the breast mass:
- 5. Ligaments:
- The structure and shape of the breast is maintained by fascial and ligamentous supports:
- As first described by Sir Astley Cooper in 1840
- Superficial fascial system:
- The breast is enveloped by the superficial and deep laminae of the superficial fascia:
- The superficial lamina is separated from the dermis by a thin layer of fatty tissue:
- But is often difficult to identify as a separate entity
- The superficial lamina is separated from the dermis by a thin layer of fatty tissue:
- The breast is enveloped by the superficial and deep laminae of the superficial fascia:
- Suspensory ligaments of Cooper:
- These fibrous strands extend through the breast parenchyma between the layers of the superficial and deep (pre-pectoral) fascia:
- They help to maintain a non-ptotic breast shape
- These fibrous strands extend through the breast parenchyma between the layers of the superficial and deep (pre-pectoral) fascia:
- The structure and shape of the breast is maintained by fascial and ligamentous supports:
- 6. Axillary tail (of Spence):
- There is a variable extension along the inferior edge of pectoralis major towards the axilla
- This usually lies within the subcutaneous fat but may penetrate the axillary fascia to lie adjacent to the lymph nodes
- Occasionally it is a separate entity with ducts that do not drain to the nipple.
- 7. Retromammary space:
- In reality this is not a space but a plane of loose connective tissue lying between the deep lamina of the superficial fascia and the deep pre-pectoral fascia
- Chassaignac bursa (also known as the retromammary bursa, submammary serous bursa or occasionally Chassaignac bag):
- Is the space behind the breast, lying between the pectoralis fascia posteriorly and deep layer of superficial fascia anteriorly
- This is the plane of dissection in which a subglandular pocket can be created for insertion of a prosthesis for breast augmentation
- 8. Muscle:
- The medial two-thirds of the base of the breast lie over the pectoralis major muscle
- The lateral one-third lies over serratus anterior and a small portion of the rectus abdominis and external oblique muscles
- The muscles are separated from the breast by the deep fascia
- 9. Rib cage:
- Deformities of the ribs, including those that are secondary to a spinal deformity can lead to an apparent asymmetry of breast position and/or shape

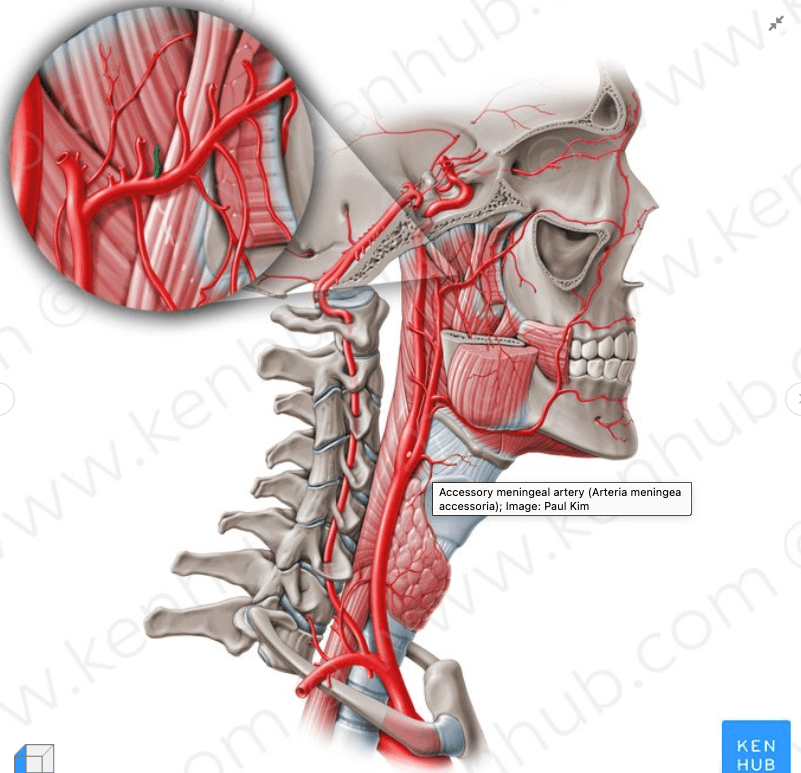
- Vascular Supply of the breast:
- The breast has a rich blood supply:
- Which permits safe division and excision of breast tissue:
- The viability of the nipple areolar complex is dependent on vessels that pass through the gland:
- Which must therefore be preserved
- The viability of the nipple areolar complex is dependent on vessels that pass through the gland:
- Which permits safe division and excision of breast tissue:
- The breast has a rich blood supply:
- There are three main arterial systems:
- Internal Thoracic (Mammary) Artery:
- Is responsible for roughly 60% of the vascular supply to the breast
- Arising directly from the subclavian artery, the internal thoracic artery passes posterior to the subclavian vein and runs along the edge of the sternum, deep to the costal cartilages
- Perforating branches of the internal thoracic artery pass through the 2nd to 6th intercostal spaces to supply the medial half of the breast:
- The 2nd and 3rd perforators are the predominant vessels and these are preferred for anastomosis when reconstructing the breast with a free tissue transfer
- Internal Thoracic (Mammary) Artery:
- Lateral Thoracic Artery:
- A branch of the second portion of the axillary artery:
- Supplies the upper outer quadrant of the breast
- The lateral thoracic artery runs along the lower border of the pectoralis minor muscle and curls around the lateral border of pectoralis major to enter the breast
- Other branches of the lateral thoracic artery perforate pectoralis major to supply the overlying breast tissue
- A branch of the second portion of the axillary artery:
- Posterior Intercostal Arteries:
- The lateral branch of the posterior intercostal arteries divides into posterior and anterior branches
- The anterior branches from the 3rd to 6th intercostal spaces supply the lateral portion of the breast and the overlying skin through their mammary branches
- Other Supply:
- The axillary artery also provides other branches to the breast, including the:
- Superior thoracic artery:
- A branch from the first part of the axillary artery)
- The pectoral branch of the thoracoacromial artery and the subscapular artery
- Superior thoracic artery:
- The axillary artery also provides other branches to the breast, including the:

- The venous drainage of the breast is via two venous systems:
- Superficial system:
- Which lies within the subdermal venous plexus:
- The pattern of drainage is highly variable
- Deep system:
- The deep venous system parallels the arterial supply:
- The medial half of the breast drains via veins that accompany the perforating branches of the internal mammary artery through the intercostal spaces, back to the internal mammary vein
- The lateral thoracic veins drain into the axillary vein
- The posterior intercostal veins drain into the azygous vein on the right and the hemiazygous vein on the left
- The deep venous system parallels the arterial supply:
- Innervation of the breast:
- The nerve supply to the breast consists of sensory fibres from the skin and sympathetic efferent fibres to the blood vessels, glandular tissue and smooth muscle cells in the skin and nipple
- The sensory nerve supply is derived from cutaneous branches of the intercostal nerves:
- Medially:
- Anterior branches of the 1st to 6th intercostal nerves
- Medially:
- Laterally:
- Lateral branches of the 2nd to 6th intercostal nerves
- Nipple areola complex:
- Supplied by the anterior branch of the 4th intercostal nerve
- There is an extensive nerve plexus within the nipple
- The skin of the nipple areola complex contains free nerve endings, Meissner’s corpuscles and Merkel disc endings