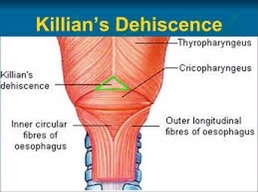
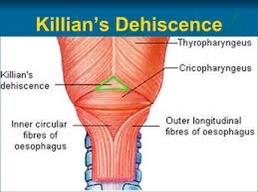
- I went to medical school at the Anahuac University in Mexico City, which is one of the most prestigious medical schools in Mexico:
- I graduated Suma Cum Laude from this medical school and was the president of the student medical council.
- I trained in general surgery at Michigan State University where I was named chief resident during my fifth year of residency which was a great honor.
- My complex surgical oncology fellowship which included a head and neck training was performed at the Fox Chase Cancer Center in Philadelphia, Pennsylvania.
- At the same time, I undertook a Masters in Science (Clinical Research for Health Care Professionals) at Drexel University in Philadelphia, Pennsylvania.
- I also performed a two-year global online fellowship in Head and Neck Surgery and Oncology with the International Federations of Head and Neck Societies / Memorial Sloan Kettering Cancer Center.
- I encountered patients with very complex problems, and the greatest lesson I learned was there are always treatment options, utilizing all different types of techniques including radiation, chemotherapy and surgery.
- This comprehensive training has provided me with an extensive understanding of the multidisciplinary approach to treating patients with cancer.
- I encountered patients with very complex problems, and the greatest lesson I learned was there are always treatment options, utilizing all different types of techniques including radiation, chemotherapy and surgery.
- I have developed a particularly strong interest in the surgical and multimodal treatment of patients with breast cancer, head and neck cancer (including thyroid and parathyroid cancer), and endocrine diseases (benign and malignant thyroid and parathyroid diseases), using traditional surgery, regional therapies, and minimally invasive techniques.
- I am an expert in the treatment of thyroid cancer including; active surveillance for early, small papillary thyroid cancers, minimally invasive thyroid surgery, selective and comprehensive neck dissections.
- For the management of parathyroid disease, I offer a minimally invasive radio-guided technique called MIRP (minimally invasive radio-guided parathyroidectomy) through a 2 cm incision which will allow the patient to have a great cosmetic result and quick return to normal life after the operation.
- I am extremely aware of the impact that a breast cancer diagnosis has on a patient. I do my best to promote a positive atmosphere in which to start my patients’ course of treatment and take the time to explain the pros and cons of each treatment option, so that they can make an informed decision.
- My management philosophy also includes, not just an emphasis on successful treatment, but also preserving a good cosmetic outcome. I feel fortunate to be a fellowship trained, very highly specialized clinician, because this combination of factors allows me, and our treatment team to focus on one thing all day, every day, and do it well: curing cancer. I think there is nothing more rewarding that I could do as a clinician.
- I hold my patients as my number one priority. I will spend as much time as necessary educating, answering questions and providing guidance for each individual patient to help them throughout each stage of their management. I believe in honest discussions, where both the patients and family’s goals and expectations are openly communicated. We will work together as a team to put together an evidence based personalized treatment plan. My personal goal is to treat and care for every patient with the same compassion and honesty as if they were a friend or family member.

👉Will join the Center for Advanced Surgical Oncology at Palmetto General Hospital as a breast / thyroid / parathyroid / head and neck surgeon in July, 2020
#Arrangoiz #Surgeon #CancerSurgeon #HeadandNeckSurgeon #BreastSurgeon #SurgicalOncologist #PalmettoGeneralHospital #CenterforAdvancedSurgicalOncology