- Diagnosis of a breast cancer during pregnancy requires:
- A complex treatment plan with multiple multidisciplinary providers:
- From both oncology and obstetrics:
- Coordinating the timing of cancer treatment and the delivery of a high-risk pregnancy
- From both oncology and obstetrics:
- A complex treatment plan with multiple multidisciplinary providers:
- Gestational or pregnancy-associated breast cancer:
- Refers to any breast cancer diagnosed:
- During pregnancy or within the first year after childbirth
- This is a rare diagnosis:
- Yet remains the most common cancer in pregnant women:
- Affecting approximately 15 to 35 per 100,000 deliveries:
- Approximately 0.05%
- Affecting approximately 15 to 35 per 100,000 deliveries:
- Yet remains the most common cancer in pregnant women:
- The majority of pregnancy-associated breast cancers are:
- Ductal in origin
- More likely to be poorly-differentiated
- ER negative or PR negative and HER2-positive compared to non-pregnant women
- Present at advanced stages
- Evaluation of a dominant breast mass should include:
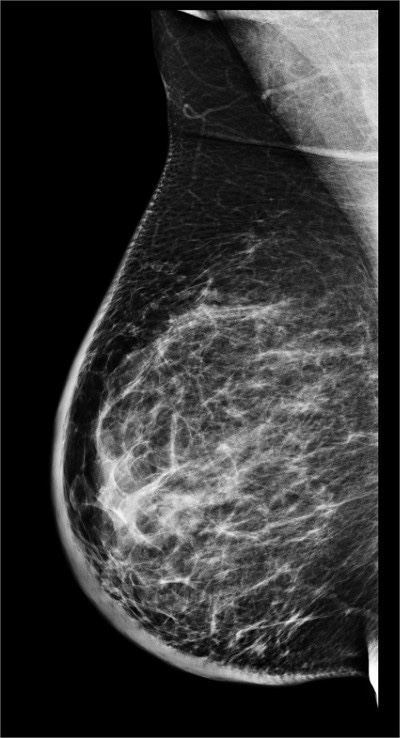
- Ultrasound and mammogram with fetal shielding, and core biopsy:
- Interpretation of mammography can be difficult in the highly dense tissue of pregnant women
- Use of MRI during pregnancy is both contraindicated and unhelpful:
- Gadolinium contrast may cause fetal harm during the first trimester and is typically avoided
- Although the majority (approximately 80%) of breast biopsies in pregnant women will be benign:
- It is critical that malignancy be ruled out
- Fine needle aspiration during pregnancy is associated with a higher rate of false positive and false negative results, without receipt of tumor markers:
- Therefore core biopsy is recommended
- Routine staging is not recommended:
- In an asymptomatic patient with early-stage breast cancer
- Routine staging if indicated:
- Should be performed per National Comprehensive Cancer Network guidelines:
- With consideration given to safety of both the patient and fetus
- CT scans are not recommended due to excessive fetal radiation exposure
- Should be performed per National Comprehensive Cancer Network guidelines:
- Ultrasound and mammogram with fetal shielding, and core biopsy:
- Refers to any breast cancer diagnosed:
Early termination of pregnancy, is not thought to improve outcome in these patients
- Staging should be performed in women with advanced disease, and in those with symptoms concerning for metastases:
- This should include:
- Chest radiograph with fetal shielding
- Liver ultrasound or MRI without contrast
- “Low-dose” radionuclide bone scans
- When possible, treatment of pregnancy-associated breast cancers:
- Should follow similar guidelines to non-pregnant patients, and if at all possible, the pregnancy should be carried to term
- Systemic treatment of breast cancer during pregnancy:
- Involves special consideration of both the mother and baby
- Surgery is safe at all stages
- Chemotherapy can be delivered:
- From 14 weeks of gestation following completion of organogenesis through 35 weeks,:
- When it should be stopped to avoid leukopenia in preparation for delivery
- From 14 weeks of gestation following completion of organogenesis through 35 weeks,:
- Data from a single-institution prospective study indicates that:
- FAC chemotherapy (5-FU, doxorubicin, and cyclophosphamide) is safe during the second and third trimesters:
- With fetal malformations approximating 1%
- FAC chemotherapy (5-FU, doxorubicin, and cyclophosphamide) is safe during the second and third trimesters:
- Experience with taxanes remained limited:
- There are insufficient safety data regarding the use of taxanes during pregnancy and as such they are not recommended for general use during pregnancy
- But the National Comprehensive Cancer Network (NCCN) guidelines:
- Recommend weekly paclitaxel if warranted
- But the National Comprehensive Cancer Network (NCCN) guidelines:
- There are insufficient safety data regarding the use of taxanes during pregnancy and as such they are not recommended for general use during pregnancy
- Methotrexates are contraindicated due to teratogenic side effects
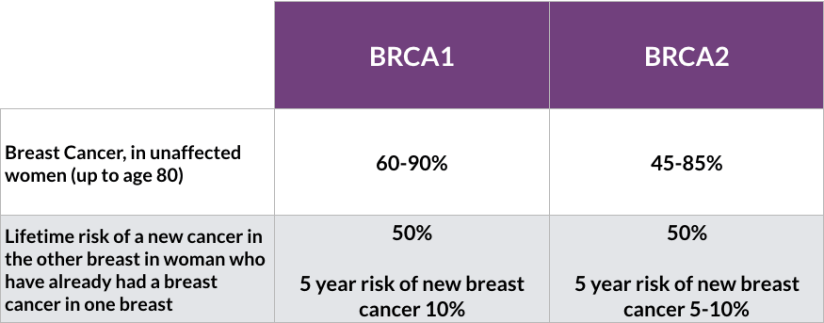
- Although 20% of pregnancy-associated breast cancers are HER2-positive:
- Anti-HER2 therapy including trastuzumab has not been proven safe during pregnancy and is best delivered in the adjuvant setting
- Anhydramnios has been reported with use of trastuzumab during pregnancy
- MotHER, a prospective U.S. registry:
- Is evaluating women exposed to trastuzumab +/- pertuzumab during pregnancy or within 6 months of conception and following pregnancy outcomes and infants for the first month of life:
- Current recommendations suggest that targeted anti-HER2 therapy be delayed until after delivery
- Is evaluating women exposed to trastuzumab +/- pertuzumab during pregnancy or within 6 months of conception and following pregnancy outcomes and infants for the first month of life:
- Breast conservation is not recommended:
- If radiation would be timed during pregnancy because this is contraindicated:
- However, it can be performed if radiation falls after delivery
- If radiation would be timed during pregnancy because this is contraindicated:
- Axillary lymph node dissection was previously recommended:
- But sentinel lymph node biopsy is feasible and should be offered
- Hormone therapy should be postponed until after childbirth
- This should include:
- Chemotherapy during pregnancy can cause concerns for the fetus which depend on the timing of drug administration:
- In the first trimester:
- Congenital malformations can occur
- In the second and third trimester the greatest concerns are :
- Prematurity
- Low birth weight
- Myelosuppression
- Among approved chemotherapy agents:
- Long-term outcomes of children with in-utero exposure demonstrate normal development, cognition, and school performance when prematurity is controlled for
- In the first trimester:
- References
- NCCN Guideline: Breast cancer during pregnancy. National Comprehensive Cancer Network website. https://www.nccn.org/professionals/physician_gls/default_nojava.aspx. Accessed September 21, 2019. Accessed September 21, 2019.
- Brent RL. The effect of embryonic and fetal exposure to x-ray, microwaves, and ultrasound: counseling the pregnant and nonpregnant patient about these risks. Sem Oncol. 1989;16(5):347-368.
- Meisel JL, Economy KE, Calvillo KZ, et al. Contemporary multidisciplinary treatment of pregnancy-associated breast cancer. Springerplus. 2013;2(1):297.
- Pant S, Landon MB, Blumenfeld M, Farrar W, Shapiro CL. Treatment of breast cancer with trastuzumab during pregnancy. J Clin Oncol. 2008;26(9):1567-1569.
- Yang WT, Dryden MJ, Gwyn K, Whitman GJ, Theriault R. Imaging of breast cancer diagnosed and treated with chemotherapy during pregnancy. Radiology. 2006;239(1):52-60.
- Berry DL, Theriault RL, Holmes FA, et al. Management of breast cancer during pregnancy using a standardized protocol. J Clin Oncol. 1999;17(3):855-861.
- Macdonald HR Pregnancy associated breast cancer. Breast J. 2020 Jan 14. doi: 10.1111/tbj.13714. [Epub ahead of print]
- Goidescu I, Nemeti G, Caracostea G, Eniu DT, Chiorean A, Pintican R, Cruciat G, Muresan D. The role of imaging techniques in the diagnosis, staging and choice of therapeutic conduct in pregnancy associated breast cancer. Med Ultrason. 2019 Aug 31;21(3):336-343. doi: 10.11152/mu-1958. Review.
- Alfasi A, et al. Breast Cancer during Pregnancy-Current Paradigms, Paths to Explore. Cancers. 2019; 11: 1669
- Gooch JC1,2, Chun J1, Kaplowitz E1, Guth A1, Axelrod D1, Shapiro R1, Roses D1, Schnabel F1. Pregnancy-associated breast cancer in a contemporary cohort of newly diagnosed women Breast J. 2019 Aug 25. doi: 10.1111/tbj.13510. [Epub ahead of print]

#Arrangoiz #BreastSurgeon #CancerSurgeon #SurgicalOncologist #BreastCancer #PregnancyAssociatedBreastCancer #Miami #CASO #CenterforAdvancedSurgicalOncology