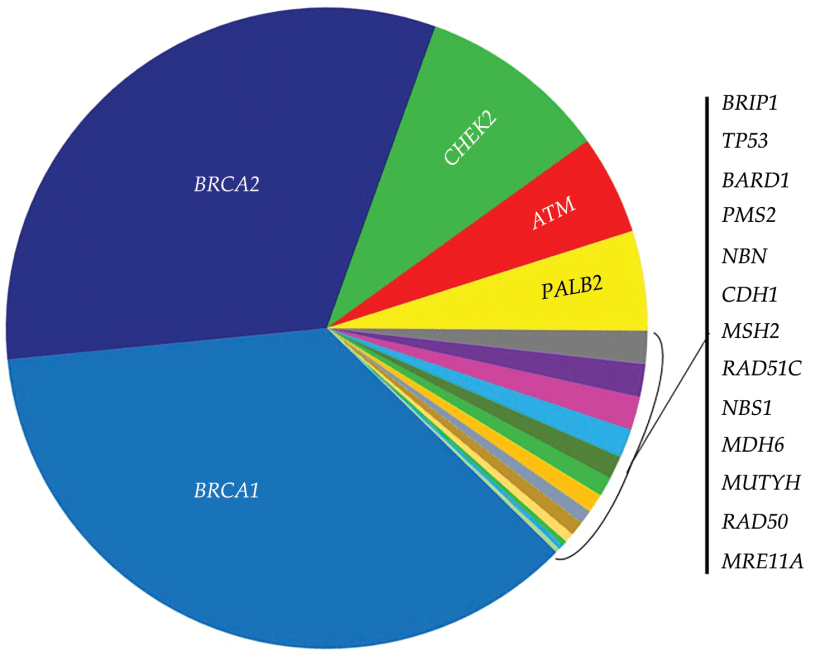
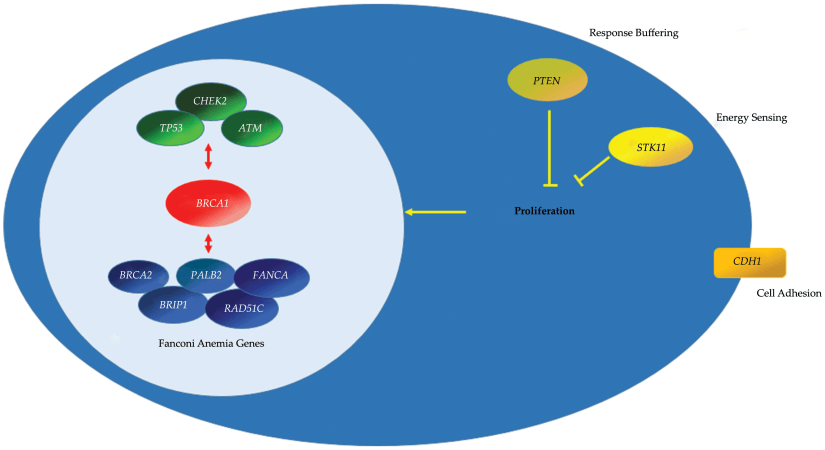
- For patients with BRCA mutations:
- Bilateral mastectomy is advised to reduce the risk of breast cancer
- Mastectomy has been shown to reduce the risk of breast cancer:
- In patients with hereditary cancer by 90%, according to both retrospective and prospective studies
- In many of the seminal studies investigating surgical prophylaxis of breast cancer:
- The type of surgery performed was a total (or simple) mastectomy:
- Which consists of removing all breast tissue, including overlying skin, as well as the nipple‐areolar complex while sparing the pectoral muscles and axillary content
- The type of surgery performed was a total (or simple) mastectomy:
- In the PROSE study:
- The only two women diagnosed with breast cancer after surgical prophylaxis:
- Had undergone skin‐sparing instead of total mastectomies:
- One of these patients was diagnosed with metastatic breast cancer at the time of surgery
- The other woman went on to develop breast cancer within the residual breast epithelium
- Had undergone skin‐sparing instead of total mastectomies:
- Therefore, total mastectomy:
- Is still the recommended risk‐reducing surgery in BRCA carriers
- The only two women diagnosed with breast cancer after surgical prophylaxis:
- Nipple‐sparing (NSM) and skin‐sparing (SSM) mastectomies:
- Require a less extensive reconstruction and often a better cosmetic result
- Since the PROSE study in 2004:
- A piece of mounting evidence demonstrates that NSM, as well as SSM:
- May be effective forms of surgical prophylaxis in BRCA carriers
- A piece of mounting evidence demonstrates that NSM, as well as SSM:
- In a recent retrospective study, patients with BRCA who underwent prophylactic NSM at nine institutions from 1968 to 2013 were reviewed
- In this cohort, all 346 BRCA mutation carriers who did not have incidental cancer on final pathology:
- Did not develop breast cancer after 36 months of follow up
- Using risk models for BRCA carriers:
- The authors approximated that 22 primary breast cancers were expected in this cohort without prophylactic NSM
- Nevertheless, this was a retrospective study and more evidence is needed for NSM to replace the currently recommended total mastectomy for surgical prophylaxis of breast cancer in BRCA patients
- In this cohort, all 346 BRCA mutation carriers who did not have incidental cancer on final pathology:
- References:
- Rebbeck TR, Friebel T, Lynch HT, et al. Bilateral prophylactic mastectomy reduces breast cancer risk in BRCA1 and BRCA2 mutation carriers: the PROSE Study Group. J Clin Oncol. 2004;22(6): 1055‐1062.
- Ludwig KK, Neuner J, Butler A, Geurts JL, Kong AL. Risk reduction and survival benefit of prophylactic surgery in BRCA mutation carriers, a systematic review. Am J Surg. 2016;212(4):660‐669.
- Jakub JW, Peled AW, Gray RJ, et al. Oncologic safety of prophylactic nipple‐sparing mastectomy in a population with BRCA mutations: a multi‐institutional study. JAMA Surg. 2018;153(2):123‐129.
- Yao K, Liederbach E, Tang R, et al. Nipple‐sparing mastectomy in BRCA1/2 mutation carriers: an interim analysis and review of the literature. Ann Surg Oncol. 2015;22(2):370‐376.
- Domchek SM, Friebel TM, Singer CF, et al. Association of risk‐ reducing surgery in BRCA1 or BRCA2 mutation carriers with cancer risk and mortality. JAMA. 2010;304(9):967‐975.

#Arrangoiz #BreastSurgeon #CancerSurgeon #SurgicalOncologist #CASO #CenterforAdvancedSurgicalOncology #Miami #RiskReducingSurgery #RiskReducingMastectomy