- Nipple discharge is a relatively frequent event in females:
- Being the third most common breast symptom prompting medical care:
- After breast pain and breast palpable mass
- Being the third most common breast symptom prompting medical care:
- Over 80% of females will develop an episode of nipple discharge during their fertile life:
- Which can be categorized as:
- Lactational
- Physiological
- Pathological
- According to the clinical history and the characteristics of the discharge
- Which can be categorized as:
- Lactational nipple discharge:
- Is considered as a normal milk production
- It is expected during pregnancy and lactation:
- May persist for up to one-year post-partum or after cessation of breastfeeding
- When a milky nipple discharge:
- Occurs in females without recent history of pregnancy or lactation:
- It is called galactorrhea:
- Commonly involves bilateral multiple ducts
- This is the result of an inappropriate increase in prolactin release:
- Usually supported by a prolactinoma:
- A prolactin-producing benign tumor of pituitary gland
- Usually supported by a prolactinoma:
- It is called galactorrhea:
- Occurs in females without recent history of pregnancy or lactation:
- Physiological nipple discharge:
- Is a benign entity:
- Usually bilateral
- White, green, or yellow in color
- It involves multiple ducts
- Is associated with nipple squeezing
- Some causes of physiological nipple discharge are:
- Hypothyroidism
- Medication side-effects
- Is a benign entity:
- Pathologic nipple discharge (PND):
- Is defined as a clear, serous, or bloody nipple secretion (not green or milky):
- It is spontaneous
- Discharging from a single duct and unilateral
- It is frequently caused by:
- A benign lesion, such as:
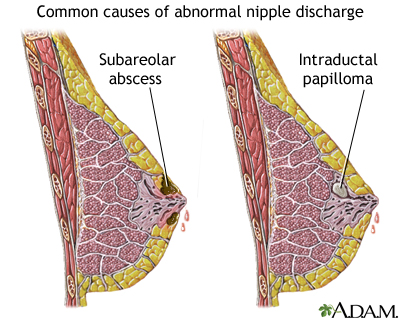
- Intraductal papilloma(s):
- 35% to 56% of the cases
- Ductal ectasia:
- 6% to 59% of the cases
- Intraductal papilloma(s):
- An underlying malignancy can be present in a percentage of cases:
- Reported to be variable from 5% to 33%
- A benign lesion, such as:
- Because to differentiate between a benign from a malignant etiology of a PND based on clinical and diagnostic assessment is not easy:
- Surgical excision has been considered the main way for getting both definitive diagnosis and eliminating the symptom
- Is defined as a clear, serous, or bloody nipple secretion (not green or milky):
- Clinical History and Physical Examination:
- Clinical history plays an important role:
- For evaluating the probability of malignancy
- Predicting factors for malignancy in the presence of PND are:
- BRCA 1 / 2 mutations
- History of ipsilateral cancer
- Previous breast biopsy with diagnosis of atypia
- Age over 50 years:
- In a study including 318 patients with nipple discharge (any fluid from the nipple, spontaneous discharge or observed during breast examination):
- Seltzer has reported a higher incidence of breast cancer:
- Equal to 9% in females over 50 (95 patients and 9 cancers):
- While the incidence was of only 1.3% in younger patients (223 patients and 3 cancers)
- Equal to 9% in females over 50 (95 patients and 9 cancers):
- Seltzer has reported a higher incidence of breast cancer:
- In a study including 318 patients with nipple discharge (any fluid from the nipple, spontaneous discharge or observed during breast examination):
- Physical examination:
- Has the aim of distinguishing between benign and pathological discharge and of verifying the presence of palpable mass or other associated findings
- It usually includes:
- A complete breast evaluation:
- With inspection and palpation
- Followed by a focused inspection of the nipple area:
- Using a magnifying lamp
- A complete breast evaluation:
- The physical examination is essential to investigate the:
- Color of discharge
- The number of ducts involved
- The frequency of discharge (persistent or intermittent)
- If it is unilateral or bilateral
- A spontaneous single-pore bloody and clear discharge:
- Is suspect for pathological discharge
- Clinical history plays an important role:
- Mammography:
- Represents the first conventional imaging technique to investigate nipple discharge:
- At least after 39 years old
- For patients with PND, aged between 30 and 40 years old with high-family risk:
- Mammography could be appropriated in order to exclude the presence of microcalcifications
- As well as for females younger than 30 of age:
- When initial ultrasound shows suspicious findings
- The protocol includes:
- The standard cranio-caudal and mediolateral oblique views
- Mammography findings that are suspect to be associated to an occult malignancy can range from:
- Microcalcifications
- Masses
- Focal density asymmetry
- Architectural distortion or ductal ectasia
- Otherwise no abnormality can be identified
- Mammography has low sensitivity and limited accuracy:
- In the detection of retroareolar lesions that are often small, intraductal, and without calcifications
- Ductal ectasia:
- May occur as a general increase in density of the retroareolar region and in order to better visualize the area:
- Spot compression views could be performed
- May occur as a general increase in density of the retroareolar region and in order to better visualize the area:
- In order to improve spatial resolution:
- Magnification mammography can be performed:
- To identify microcalcifications and to distinguish between benign or malignant duct disease
- Magnification mammography can be performed:
- Microcalcifications with:
- Branching or linear pattern, variable density, or distributed in a segmental way:
- Are all highly suspicious of malignancy
- Whereas round or rod-like calcifications:
- Suggest for benign disease
- Branching or linear pattern, variable density, or distributed in a segmental way:
- Bahl et al studied 252 patients with at least one pathological feature of nipple discharge (unilateral, clear or bloody, or spontaneous discharge) who underwent surgical excision or a 2-year follow-up:
- Of 20 cancers diagnosed:
- Only three were revealed by mammography:
- With a 15% (3/20) sensitivity
- Only three were revealed by mammography:
- Of 20 cancers diagnosed:
- In other studies, the sensitivity of mammography:
- Ranged from 7% to 26%.
- Represents the first conventional imaging technique to investigate nipple discharge:
- Ultrasound:
- Offers a better performance than mammography:
- For detecting intraductal lesions
- Ductal ectasia:
- Defined by a duct caliber greater than 3 mm
- Is one of the most common findings seen on ultrasound:
- It appears as dilated retroareolar ducts containing anechoid fluid or hypoecoic debris
- Is one of the most common findings seen on ultrasound:
- Defined by a duct caliber greater than 3 mm
- An intraductal papilloma appears as:
- A hypoechoic nodule with a central vascular pedicle on color Doppler:
- Doppler ultrasound is helpful in differentiating:
- Intraductal viscous secretion versus intraductal nodule with vascular sign
- Doppler ultrasound is helpful in differentiating:
- A hypoechoic nodule with a central vascular pedicle on color Doppler:
- Ultrasound malignant features are:
- Irregular duct margins
- Wall thickening
- Hypoechoic intraductal mass with acoustic shadowing
- In a study by Park et al:
- The detection rate of malignant lesions occult on mammography and ultrasound-detected:
- Was reported to be 8 of 53 females with PND examined (15%)
- The detection rate of malignant lesions occult on mammography and ultrasound-detected:
- Yoon et al:
- Have also reported that adding ultrasound to mammography in the pre-operative setting of PND:
- Led to the detection of malignancies in 26% of patients (ultrasound detected fivebreast cancers in addition to the 19 breast cancers found by mammography)
- Have also reported that adding ultrasound to mammography in the pre-operative setting of PND:
- The role of ultrasound elastography is disputable in predicting malignancy in patients with PND:
- Guo et al have evaluated the diagnostic accuracy of elastography in patients with PND:
- Affirming that it is a useful tool for predicting malignancy:
- With sensitivity for malignancy of 90% and that it could be used as a helpful test before more invasive examination (such as ductoscopy or duct excision):
- However, it is only a preliminary study and further studies are needed to verify the diagnostic perfor- mance of elastography
- With sensitivity for malignancy of 90% and that it could be used as a helpful test before more invasive examination (such as ductoscopy or duct excision):
- Affirming that it is a useful tool for predicting malignancy:
- Guo et al have evaluated the diagnostic accuracy of elastography in patients with PND:
- Offers a better performance than mammography:
- Nipple discharge cytology:
- Is performed by squeezing the nipple with a gentle compression of the areola area and spreading the secretion onto a glass slide:
- After smearing, the slides are immediately fixed by spray fixation or by immersion in 95% ethyl alcohol:
- Then stained with the Papanicolaou stain
- After smearing, the slides are immediately fixed by spray fixation or by immersion in 95% ethyl alcohol:
- It is a simple and fast examination, easy to perform and painless:
- But strongly limited by a low sensitivity for cancer:
- With a false negative rate over 50%
- Moreover, it can be technically impossible when discharge is not present on the moment of the examination
- According to the American College of Radiology:
- This examination has not proven to be effective in differentiating benign from malignant lesions:
- Therefore, discharge cytology is not routinely recommended
- This examination has not proven to be effective in differentiating benign from malignant lesions:
- But strongly limited by a low sensitivity for cancer:
- Nipple discharge smears are classified as abnormal if they contained:
- Papillary, atypical, suspicious, or malignant cells:
- Malignant nipple discharge cytology is correlated with higher specificity values
- Papillary, atypical, suspicious, or malignant cells:
- Is performed by squeezing the nipple with a gentle compression of the areola area and spreading the secretion onto a glass slide:
#Arrangoiz #BreastSurgeon #CancerSurgeon #SurgicalOncologist #Teacher #BreastCancer #NippleDischarge #PathologicNippleDischarge #PhysiologicNippleDischarge #CASO #Miami #CenterforAdvancedSurgicalOncology #IntraductalPapilloma #DuctalEctasia