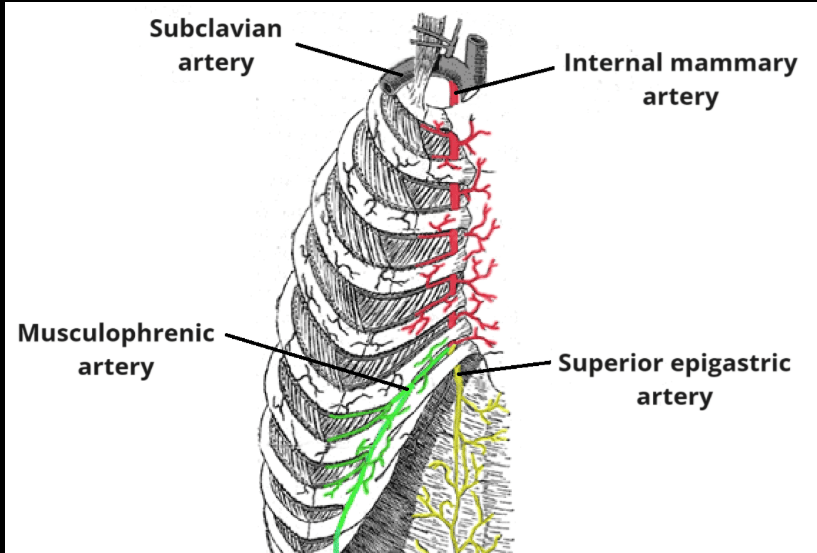
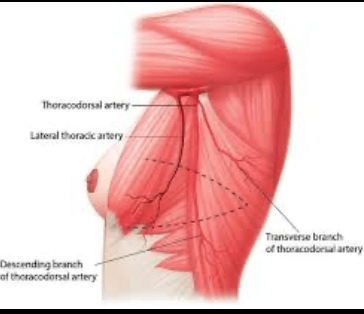
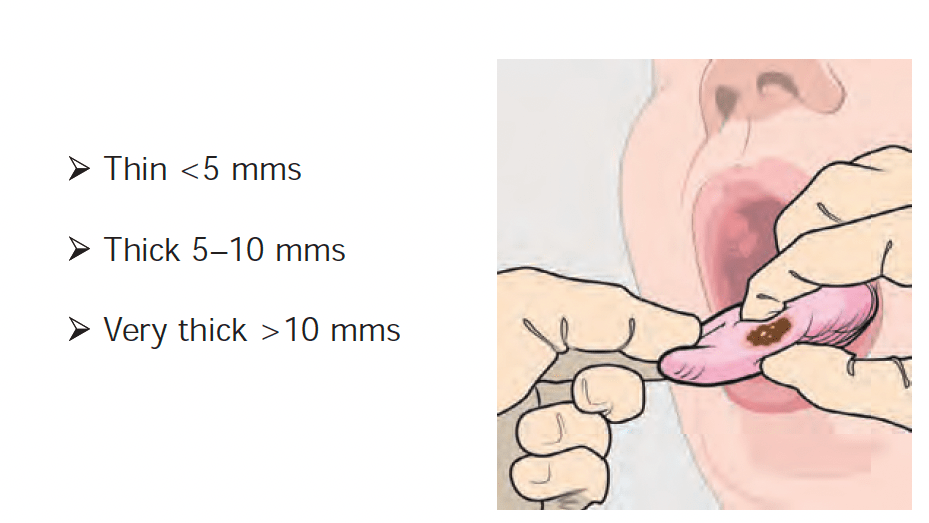
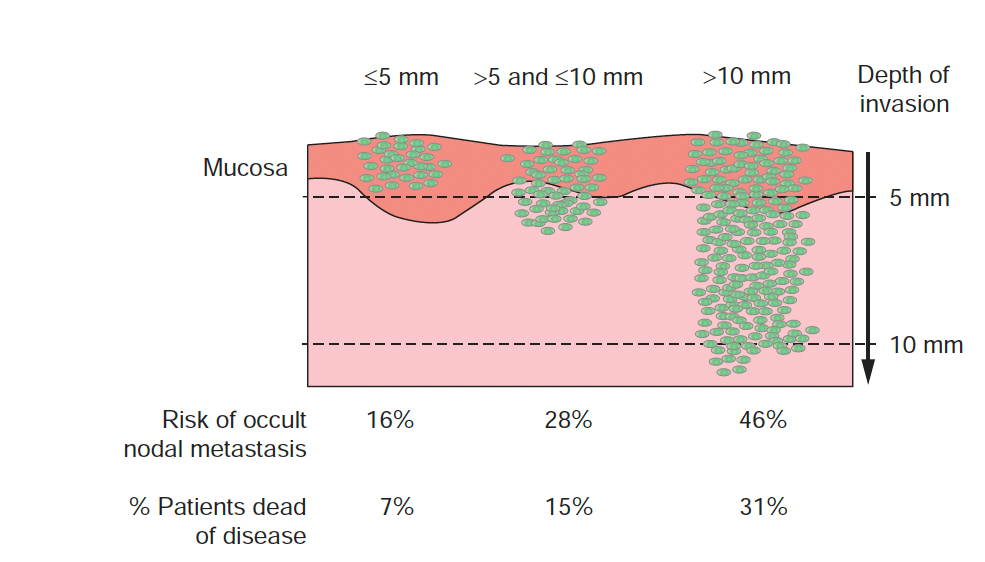
- Surgical excision of an area of atypical duct hyperplasia (ADH) found on core needle biopsy:
- Is recommended to rule out underlying occult breast cancer:
- Which can be found in 15% to 30% of patients
- Is recommended to rule out underlying occult breast cancer:
- Studies consistently show higher rates of upgrade to DCIS (2/3 of the cases) compared to invasive carcinoma (1/3 of the cases)
- A multivariable model assessing predictors for risk of upgrade at the time of excision of ADH found that:
- Lesions that were less than 50% removed by core biopsy, compared to those with greater than 90% removed:
- Had a significantly higher risk of upgrade (OR 3.8)
- Similarly, ADH with individual cell necrosis (OR 4.3) and with multiple foci of atypia on core biopsy (OR 2-3 foci 2.1; OR >3 foci 3.6 compared to 1 foci) were more likely to have a subsequent upgrade
- Lesions that were less than 50% removed by core biopsy, compared to those with greater than 90% removed:
- ADH is associated with an increased risk of future development of breast cancer when identified on a core needle biopsy or at time of surgery:
- With a relative risk of approximately 4
- Increasing number of foci of atypia:
- Has also been reported to be associated with increasing future breast cancer risk
- A study by Degnim and colleagues combined outcomes for women with a history of atypical hyperplasia from the Mayo Clinic and the Nashville Cohort:
- In the combined analysis, among women with ADH, the relative risk of breast cancer was 2.65 with 1 foci, 5.19 with 2 foci, and 8.94 with >3 foci, p<.001
- As the vast majority of subsequent cancers in this population are estrogen positive:
- Patients with ADH may benefit from chemoprevention as demonstrated in the NSABP P-1 study:
- Which demonstrated a 49% reduction in the development of invasive breast cancer in high-risk patients with the use of tamoxifen compared to placebo (p<0.00001), with the greatest benefit seen in women with atypical hyperplasia or lobular carcinoma in situ:
- However, tamoxifen did not effect overall survival
- Which demonstrated a 49% reduction in the development of invasive breast cancer in high-risk patients with the use of tamoxifen compared to placebo (p<0.00001), with the greatest benefit seen in women with atypical hyperplasia or lobular carcinoma in situ:
- Patients with ADH may benefit from chemoprevention as demonstrated in the NSABP P-1 study:
- References
- Mooney K, Bassett LW, Apple SK. Upgrade rates of high-risk breast lesions diagnosed on core needle biopsy: a single-institution and literature review. Modern Pathol. 2016;29(12):1471-1484.
- Pena A, Shah SS, Fazzio RT, Hoskin TL, Brahmbhatt RD1, Hieken TJ, at al. Multivariate model to identify women at low risk of cancer upgrade after a core needle biopsy diagnosis of atypical ductal hyperplasia. Breast Cancer Res Treat.2017;164(2):295-304.
- Hartmann LC, Degnim AC, Santen RJ, Dupont WD, Ghosh K. Atypical hyperplasia of the breast – risk assessment and management options. NEJM. 2015;372(1):78-89.
- Degnim AC, Dupont WD, Radisky DC, et al. Extent of atypical hyperplasia stratifies breast cancer risk in 2 independent cohorts of women. Cancer. 2016;122(19):2971-2978.


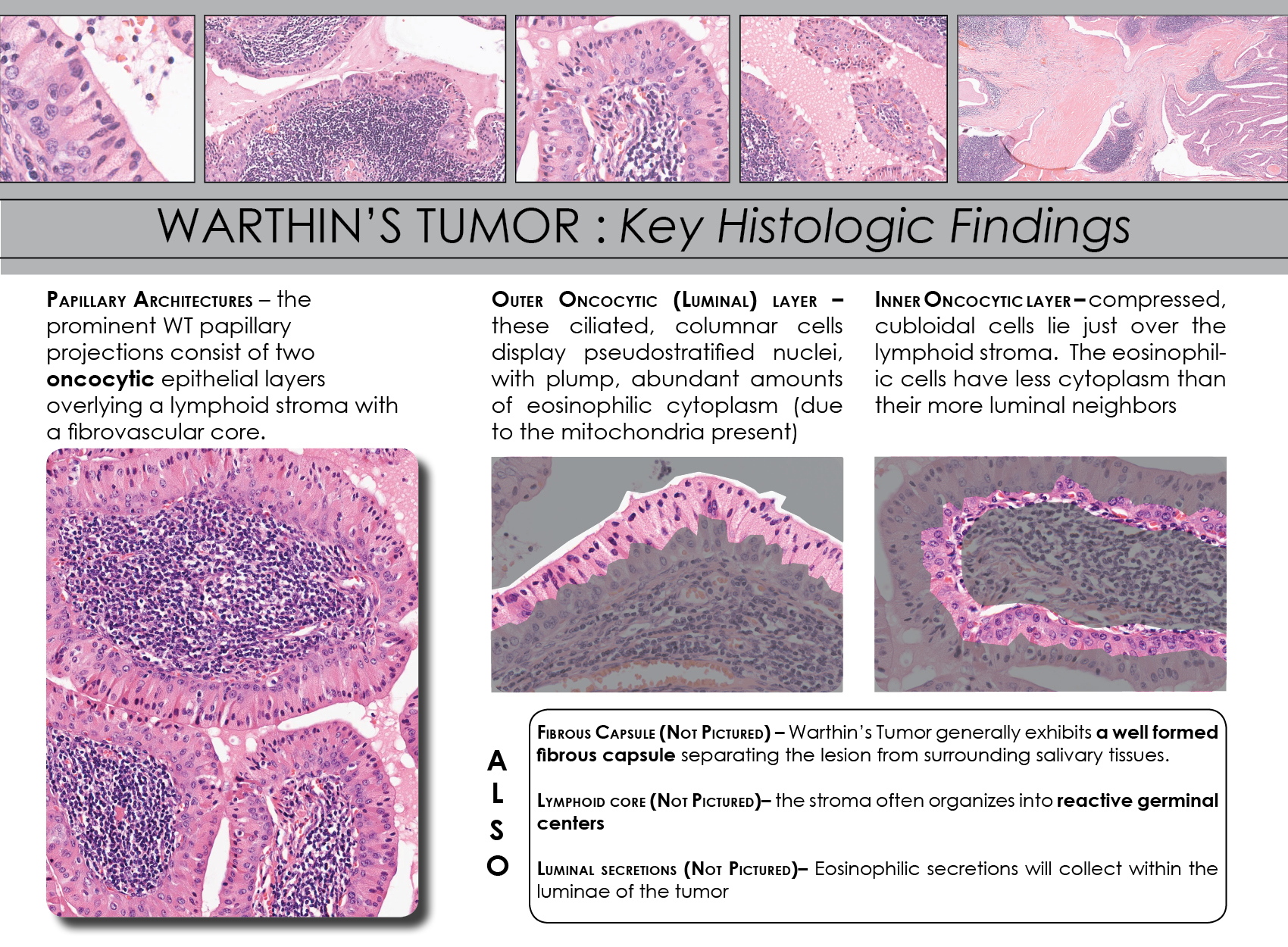
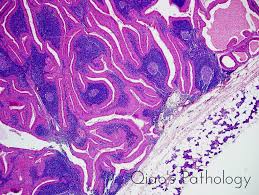
![warthin-tumor-parotid-[1-pa003-1]](https://arrangoizmd.com/wp-content/uploads/2019/01/warthin-tumor-parotid-1-pa003-1.jpeg)