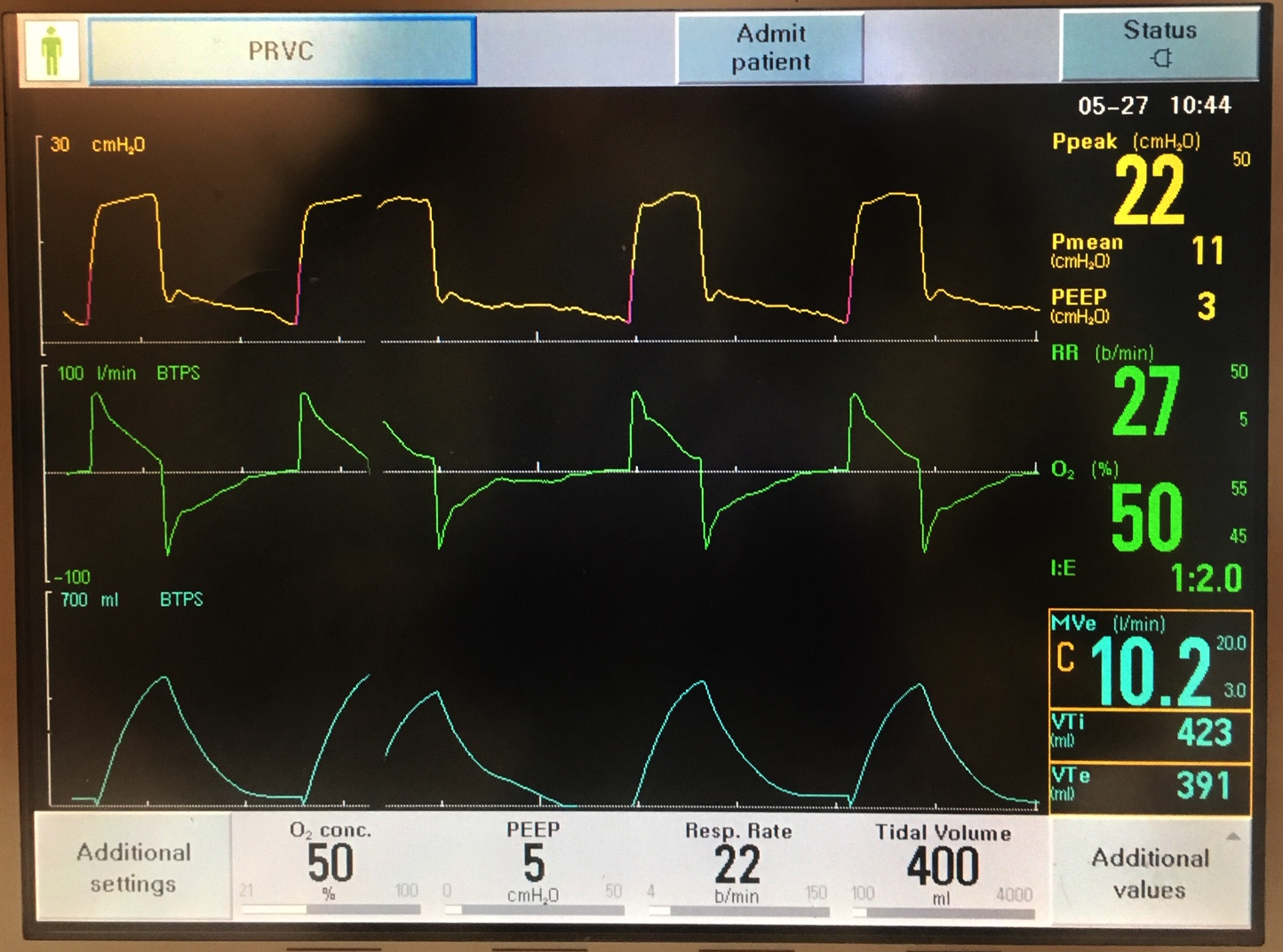
- In the NSABP B-27 trial:
- 2411 women with operable breast cancer were randomly assigned to receive:
- Preoperative:
- Group 1:
- AC followed by surgery
- Group 2:
- AC followed by docetaxel then surgery
- Group 3:
- AC followed by surgery and then docetaxel
- Group 1:
- Tamoxifen:
- Was initiated concurrently with chemotherapy
- Preoperative:
- The addition of docetaxel:
- Preoperatively or postoperatively:
- Did not significantly improve OS or DFS
- The sample size of the study was deemed insufficient to yield significance for the moderate improved DFS:
- However, in the subset of patients with a clinical partial response to AC:
- The addition of preoperative docetaxel (but not postoperative docetaxel):
- Resulted in a significant increase in DFS compared with AC alone:
- Hazard ratio, 0.71; 95% confidence interval, 0.55–0.91; P=.007
- Resulted in a significant increase in DFS compared with AC alone:
- There was a significant decrease in the cumulative incidence of all local recurrence as first events in the two groups treated with docetaxel:
- Approximately half of which was accounted for by ipsilateral breast tumor recurrences in women treated with breast-conserving therapy
- The addition of preoperative docetaxel (but not postoperative docetaxel):
- However, in the subset of patients with a clinical partial response to AC:
- Preoperatively or postoperatively:
- 2411 women with operable breast cancer were randomly assigned to receive:
- For protocol B-27:
- Mean tumor size was 4.5 cm:
- This and other key characteristics were evenly balanced among the three treatment arms
- The addition of docetaxel preoperatively resulted:
- In significant increases in cCR and pCR at the time of surgery compared with AC alone:
- 63.6% versus 40.1% and 26.1% versus 13.7%, respectively
- Despite this:
- Addition of docetaxel to AC:
- Did not significantly impact:
- On survival in this cohort of patients
- There was a trend toward improved DFS in group II patients who received preoperative docetaxel (T):
- But this was not statistically significant:
- 72% versus 67% DFS at 5 years; HR = 0.86, P = 0.10
- But this was not statistically significant:
- In an analysis of relapse-free survival (RFS):
- Which did not include second primary cancers:
- Group II had a significantly better outcome compared with group I:
- 74% versus 69% RFS at 5 years; HR = 0.81, P = 0.03).
- Group III RFS was not significantly different from group I:
- 71% at 5 years; HR = 0.91, P = 0.32
- Group II had a significantly better outcome compared with group I:
- Which did not include second primary cancers:
- Addition of docetaxel:
- Significantly reduced the incidence of local recurrences as first events:
- Including IBTR in patients treated with breast conservation
- Significantly reduced the incidence of local recurrences as first events:
- Did not significantly impact:
- Addition of docetaxel to AC:
- There were no significant interactions between treatment and estrogen receptor status, age, tumor size, or clinical nodal status
- An exploratory analysis of treatment effects in subsets of patients according to clinical response to AC suggests that:
- Preoperative T, but not postoperative T:
- Significantly increased DFS in patients who had a partial clinical response after four cycles of AC:
- 63%, 74%, 65% at 5 years for groups I, II, and III
- HR = 0.68 for group II versus group I, P = 0.003
- 63%, 74%, 65% at 5 years for groups I, II, and III
- Significantly increased DFS in patients who had a partial clinical response after four cycles of AC:
- Addition of T:
- Did not appear to be beneficial in patients:
- Who were nonresponders after AC nor in those patients who had a cCR after AC
- Did not appear to be beneficial in patients:
- Pathologic complete response:
- Was a highly significant predictor of DFS and OS in all treatment groups (HR = 0.45, P < 0.0001, and HR = 0.33, P < 0.0001, respectively)
- In addition, pathologic nodal status after chemotherapy was a significant prognostic factor for survival:
- Independent of pathologic response in the breast
- Preoperative T, but not postoperative T:
- In significant increases in cCR and pCR at the time of surgery compared with AC alone:
- Mean tumor size was 4.5 cm:
REFERENCES
- Bear HD, Anderson S, Smith RE. Sequential preoperative or postoperative docetaxel added to preoperative doxorubicin plus cyclophosphamide for operable breast cancer: National Surgical Adjuvant Breast and Bowel Project Protocol B-27. J Clin Oncol. 2006;24:2019-2027.
- NSABP Clinical Trials Overview. Protocol B-27. A Randomized Trial Comparing Preoperative Doxorubicin (Adriamycin) Cyclophosphamide (AC) to Preoperative AC Followed by Preoperative Docetaxel (Taxotere) and to Preoperative AC followed by Postoperative Docetaxel in Patients with Operable Carcinoma of the Breast. http://www.nsabp.pitt.edu/B-27.asp. Accessed December 18, 2016.
#Arrangoiz #Surgeon #BreastSurgeon #CancerSurgeon #BreastCancer