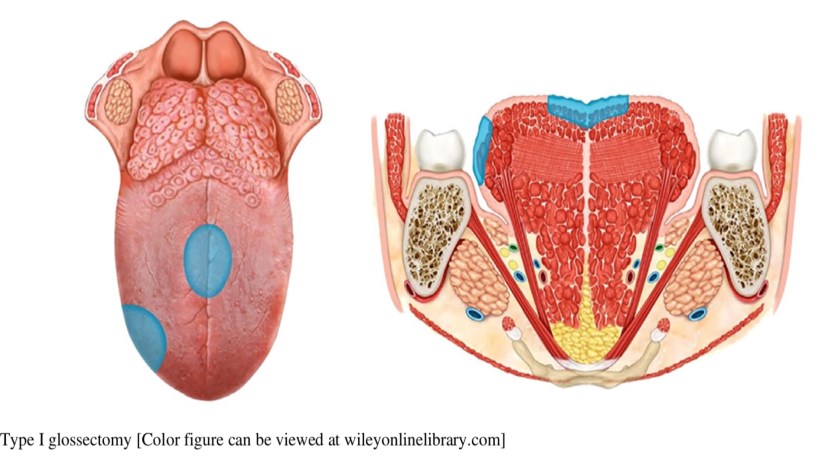
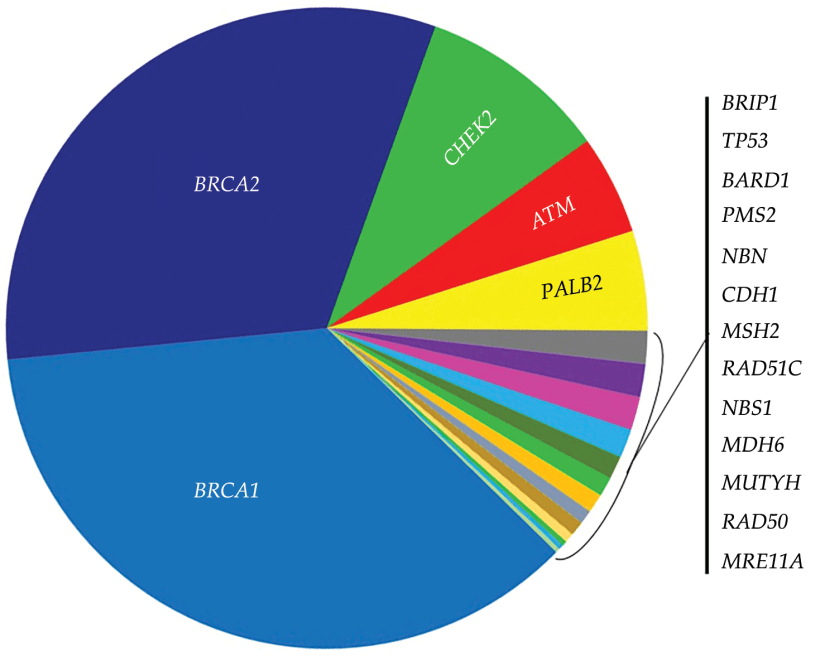
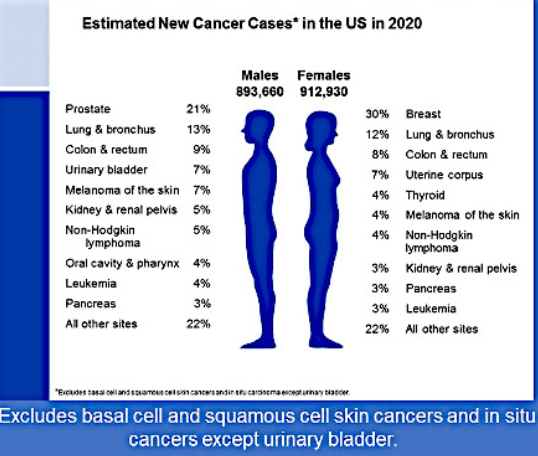
👉Expert Panel on Radiation Oncology – Head & Neck Cancer: Joseph K. Salama, MD; Nabil Saba, MD; Harry Quon, MD, MS; Jonathan J. Beitler, MD; Madhur Kumar Garg, MD; Joshua Lawson, MD; Mark W. McDonald, MD7; John A. Ridge, MD, PhD; Richard V. Smith, MD; Anamaria Reyna Yeung, MD1; Sue S. Yom, MD.
👉Introduction
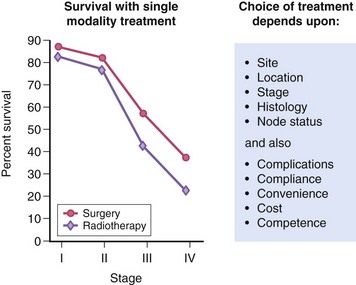
1. The standard definitive treatment of locoregionally advanced squamous cell cancer of the head and neck (SCCHN) is either concurrent chemoradiotherapy (CRT) or appropriate surgical resection followed by adjuvant therapy directed by pathologic risk factors.
2. However, local or regional recurrences and distant metastases remain frequent after surgical treatment of stage III or IV disease.
3. Radiation therapy (RT) is added to surgery to decrease locoregional failure.
4. Adjuvant RT has been shown to improve locoregional control compared to neoadjuvant RT in a multi-institutional randomized trial.
5. Postoperative RT (PORT) has traditionally been given for most patients with potentially resectable stages III, IVa, and IVb SCCHN with the goal of maximizing cure and maintaining organ function.
6. Following PORT, locoregional control is 69% to 72%, and 5-year survival rates approach 30% to 40%.
7. This has prompted interest in adding chemotherapy to surgery and RT to improve outcome.
8. The addition of concurrent chemotherapy to adjuvant RT has been shown to result in improved locoregional control, disease-free survival (DFS), and/or overall survival (OS) rates for selected SCCHN patients.
B. Role of Postoperative Radiation Therapy
1. The addition of PORT became an accepted treatment following the publication of results from the MD Anderson Cancer Center (MDACC) which demonstrated that adjuvant RT decreased recurrence.
2. The Radiation Therapy Oncology Group (RTOG) 73-03 trial randomized 320 (T2 to T4, N any) head and neck cancer patients to 50 Gy preoperatively versus 60 Gy postoperatively.
– The 10-year locoregional control was significantly improved in the postoperatively treated patients (70% vs 58%).
– OS was not different between the groups due to deaths from distant metastases and second primary cancers.
3. Further evidence supporting the role of adjuvant RT includes an analysis by the Surveillance, Epidemiology and End Results (SEER) program of node-positive head and neck cancer patients reporting an absolute 10% overall and cause-specific survival benefit at 5 years.
– All nodal stages and primary sites (except oral cavity) had improved 5-year OS rates following PORT compared to patients who did not receive PORT.
– Additionally, within a randomized study of postoperative patients, a prospective cohort of nonrandomized low-pathologic-risk postoperative patients was observed.
= Compared to patients with intermediate pathologic risk treated with PORT, the low-risk group had comparable, if not worse locoregional control.
= Finally, a small randomized study of buccal mucosa patients demonstrated a DFS benefit to PORT compared to surgical resection alone.
C. High-risk pathologic features were initially defined by two large analyses.
1. A post-hoc analysis of a randomized study of SCCHN patients treated at the MDACC demonstrated that extranodal extension was an independent predictor of recurrence, and that two or more involved lymph nodes trended toward worse locoregional recurrence.
2. These factors (ENE and two or more positive lymph nodes) were validated in a combined analysis of postoperative RTOG studies:
– RTOG 85-03 testing the addition of cisplatin prior to PORT and RTOG 88-24 testing the addition of concurrent cisplatin to PORT were reanalyzed to determine the importance of different pathologic features on risk of recurrence.
= Patients treated on the RTOG 85-03 were stratified into three post-hoc groups:
= Group 1 included those with no more than two pathologically involved lymph nodes, no extracapsular spread (ECS), and uninvolved surgical margins
= Group 2 included patients with two or more pathologically involved lymph nodes or extranodal extension and surgically uninvolved margins
= Group 3 included patients with microscopically involved surgical margins.
= These risk stratifications resulted in significant differences in 5-year locoregional recurrence rates: 17%, 27%, and 67% in groups 1, 2, and 3, respectively.
= Furthermore, median survival times were different among the three groups, being 5.6 years in group 1, 2.6 years in group 2, and 1.5 years in group 3.
D. Timing of PORT:
1. When PORT is delivered without chemotherapy, the combination of surgical resection and PORT should be considered as a treatment package.
2. Completion of the treatment package in as short a time as possible has been associated with improved locoregional control and survival rates, which are likely related to tumor repopulation effects.
3. The strongest evidence favoring completion of surgery and PORT within a tight schedule comes from a trial at the MDACC randomizing 213 patients to a tailored therapy based on their respective risk factors:
– Patients with no pathologic risk features received no PORT
– Those in the intermediate-risk group (n = 31) received 57.6 Gy over 6.5 weeks
– Those in the high-risk group were randomized to receive either 63 Gy over 5 weeks (n = 76) or 7 weeks (n = 75).
– For high-risk patients, completion of the entire treatment package in less than 11 weeks was associated with improved actuarial 5-year locoregional control (76%) compared to packages of 11 to 13 weeks duration (62%) and greater than 13 weeks duration (38%) (P=0.002).
– Although a statistically significant OS benefit defined as a P value < 0.05 was not seen, patients receiving accelerated PORT over 5 weeks trended towards improved survival (P=0.08) and locoregional control (P=0.11).
= Some single institution retrospective analyses support this finding while others do not.
= Confounding these analyses is the fact that potentially larger tumors with high-risk features required larger more complex operations with potentially longer recovery times, which may influence the initiation of PORT.
4. When PORT is delivered with concurrent chemotherapy, the impact of time to completion of treatment is presently unknown.
– In general, the treatment should be completed in as short a time frame as possible when patients have adequately healed from their surgical wounds.
– However, when the RTOG examined the addition of postoperative paclitaxel followed by radiation concurrent with paclitaxel and cisplatin, there was a noted improvement in locoregional control rates and survival when compared to historical controls.
= Therefore, whether the timing of PORT affects outcomes in the setting of concurrent chemotherapy is not clear.
American College of Radiology ACR Appropriateness Criteria ADJUVANT THERAPY FOR RESECTED SQUAMOUS CELL CARCINOMA OF THE HEAD AND NECK
Expert Panel on Radiation Oncology – Head & Neck Cancer: Joseph K. Salama, MD; Nabil Saba, MD; Harry Quon, MD, MS; Jonathan J. Beitler, MD; Madhur Kumar Garg, MD; Joshua Lawson, MD; Mark W. McDonald, MD7; John A. Ridge, MD, PhD; Richard V. Smith, MD; Anamaria Reyna Yeung, MD1; Sue S. Yom, MD.
A. Introduction:
1. The standard definitive treatment of locoregionally advanced squamous cell cancer of the head and neck (SCCHN) is either concurrent chemoradiotherapy (CRT) or appropriate surgical resection followed by adjuvant therapy directed by pathologic risk factors.
2. However, local or regional recurrences and distant metastases remain frequent after surgical treatment of stage III or IV disease.
3. Radiation therapy (RT) is added to surgery to decrease locoregional failure.
4. Adjuvant RT has been shown to improve locoregional control compared to neoadjuvant RT in a multi-institutional randomized trial.
5. Postoperative RT (PORT) has traditionally been given for most patients with potentially resectable stages III, IVa, and IVb SCCHN with the goal of maximizing cure and maintaining organ function.
6. Following PORT, locoregional control is 69% to 72%, and 5-year survival rates approach 30% to 40%.
7. This has prompted interest in adding chemotherapy to surgery and RT to improve outcome.
8. The addition of concurrent chemotherapy to adjuvant RT has been shown to result in improved locoregional control, disease-free survival (DFS), and/or overall survival (OS) rates for selected SCCHN patients.
B. Role of Postoperative Radiation Therapy
1. The addition of PORT became an accepted treatment following the publication of results from the MD Anderson Cancer Center (MDACC) which demonstrated that adjuvant RT decreased recurrence.
2. The Radiation Therapy Oncology Group (RTOG) 73-03 trial randomized 320 (T2 to T4, N any) head and neck cancer patients to 50 Gy preoperatively versus 60 Gy postoperatively.
– The 10-year locoregional control was significantly improved in the postoperatively treated patients (70% vs 58%).
– OS was not different between the groups due to deaths from distant metastases and second primary cancers.
3. Further evidence supporting the role of adjuvant RT includes an analysis by the Surveillance, Epidemiology and End Results (SEER) program of node-positive head and neck cancer patients reporting an absolute 10% overall and cause-specific survival benefit at 5 years.
– All nodal stages and primary sites (except oral cavity) had improved 5-year OS rates following PORT compared to patients who did not receive PORT.
– Additionally, within a randomized study of postoperative patients, a prospective cohort of nonrandomized low-pathologic-risk postoperative patients was observed.
= Compared to patients with intermediate pathologic risk treated with PORT, the low-risk group had comparable, if not worse locoregional control.
= Finally, a small randomized study of buccal mucosa patients demonstrated a DFS benefit to PORT compared to surgical resection alone.
C. High-risk pathologic features were initially defined by two large analyses.
1. A post-hoc analysis of a randomized study of SCCHN patients treated at the MDACC demonstrated that extranodal extension was an independent predictor of recurrence, and that two or more involved lymph nodes trended toward worse locoregional recurrence.
2. These factors (ENE and two or more positive lymph nodes) were validated in a combined analysis of postoperative RTOG studies:
– RTOG 85-03 testing the addition of cisplatin prior to PORT and RTOG 88-24 testing the addition of concurrent cisplatin to PORT were reanalyzed to determine the importance of different pathologic features on risk of recurrence.
= Patients treated on the RTOG 85-03 were stratified into three post-hoc groups:
= Group 1 included those with no more than two pathologically involved lymph nodes, no extracapsular spread (ECS), and uninvolved surgical margins
= Group 2 included patients with two or more pathologically involved lymph nodes or extranodal extension and surgically uninvolved margins
= Group 3 included patients with microscopically involved surgical margins.
= These risk stratifications resulted in significant differences in 5-year locoregional recurrence rates: 17%, 27%, and 67% in groups 1, 2, and 3, respectively.
= Furthermore, median survival times were different among the three groups, being 5.6 years in group 1, 2.6 years in group 2, and 1.5 years in group 3.
D. Timing of PORT:
1. When PORT is delivered without chemotherapy, the combination of surgical resection and PORT should be considered as a treatment package.
2. Completion of the treatment package in as short a time as possible has been associated with improved locoregional control and survival rates, which are likely related to tumor repopulation effects.
3. The strongest evidence favoring completion of surgery and PORT within a tight schedule comes from a trial at the MDACC randomizing 213 patients to a tailored therapy based on their respective risk factors:
– Patients with no pathologic risk features received no PORT
– Those in the intermediate-risk group (n = 31) received 57.6 Gy over 6.5 weeks
– Those in the high-risk group were randomized to receive either 63 Gy over 5 weeks (n = 76) or 7 weeks (n = 75).
– For high-risk patients, completion of the entire treatment package in less than 11 weeks was associated with improved actuarial 5-year locoregional control (76%) compared to packages of 11 to 13 weeks duration (62%) and greater than 13 weeks duration (38%) (P=0.002).
– Although a statistically significant OS benefit defined as a P value < 0.05 was not seen, patients receiving accelerated PORT over 5 weeks trended towards improved survival (P=0.08) and locoregional control (P=0.11).
= Some single institution retrospective analyses support this finding while others do not.
= Confounding these analyses is the fact that potentially larger tumors with high-risk features required larger more complex operations with potentially longer recovery times, which may influence the initiation of PORT.
4. When PORT is delivered with concurrent chemotherapy, the impact of time to completion of treatment is presently unknown.
– In general, the treatment should be completed in as short a time frame as possible when patients have adequately healed from their surgical wounds.
– However, when the RTOG examined the addition of postoperative paclitaxel followed by radiation concurrent with paclitaxel and cisplatin, there was a noted improvement in locoregional control rates and survival when compared to historical controls.
= Therefore, whether the timing of PORT affects outcomes in the setting of concurrent chemotherapy is not clear.