
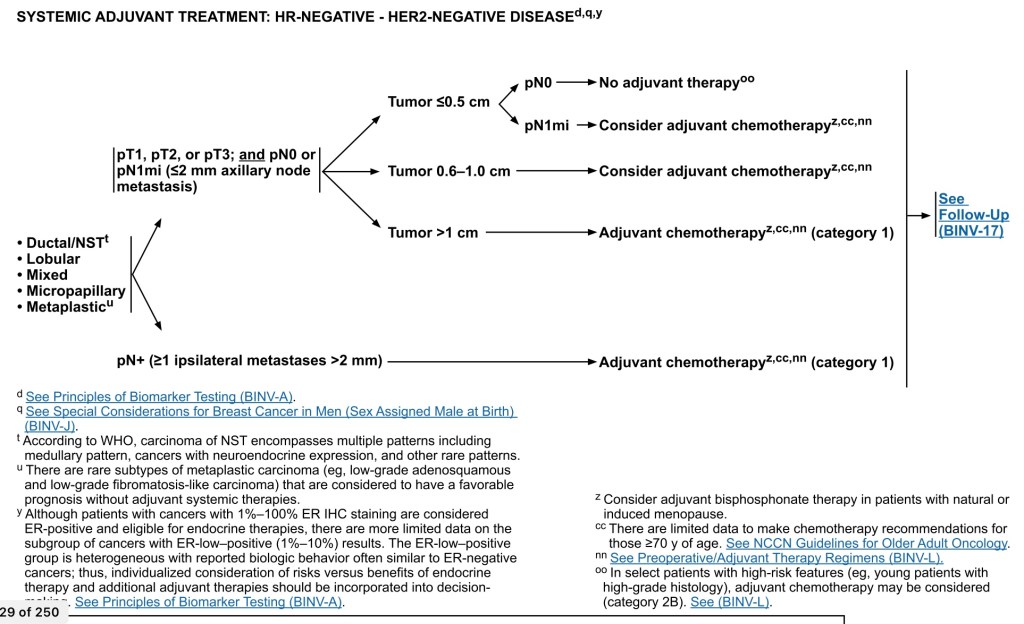
- Algorithm for Adjuvant Therapy (AC) in TNBC:
- For histologic types:
- Ductal
- Lobular
- Mixed
- Metaplastic
- For pT1, T2, or T3 and pN0 or pN1mi (≤ 2 mm axillary nodal metastasis), adjuvant systemic treatment depends on tumor size:
- For T1a (T ≤ 0.5 cm including microinvasive) and pN0:
- NO adjuvant therapy, however, if pN1mi:
- Adjuvant therapy should be considered
- NO adjuvant therapy, however, if pN1mi:
- For T1b (T > 0.5 cm and ≤ 1.0 cm), pN0 or pN1mi:
- Adjuvant therapy should be considered
- For tumors greater than 1 cm, N0 or N1mi:
- Chemotherapy should be given
- For node-positive disease (one or more metastases ≥ 2 mm) to one or more axillary lymph nodes:
- AC should be given
- For T1a (T ≤ 0.5 cm including microinvasive) and pN0:
- For histologic types:
- Standard Adjuvant Systemic Chemotherapy for TNBC:
- Current AC for TNBC does not differ according to subtype
- Preferred regimens are:
- Dose-dense AC (doxorubicin / cyclophosphamide) followed by paclitaxel every 2 weeks
- Dose-dense AC followed by weekly paclitaxel
- TC (docetaxel and cyclophosphamide)

- Other acceptable adjuvant therapy regimens as per NCCN guidelines are:
- Dose-dense AC (doxorubicin / cyclophosphamide)
- AC (doxorubicin / cyclophosphamide) every 3 weeks
- Cyclophosphamide / methotrexate / fluorouracil
- AC followed by docetaxel every 3 weeks
- AC followed by weekly paclitaxel
- Epirubicin / cyclophosphamide
- TAC (docetaxel / doxorubicin / cyclophosphamide)
- High-risk triple-negative patients are, in general:
- Treated with an anthracycline and a taxane-containing regimen
- Emerging concepts in systemic therapy for TNBC – Precision Medicine:
- The considerable overlap of the BRCA1 tumor phenotype and TNBC:
- Has led investigators to take advantage of TNBC BRCA-ness when considering chemotherapy regimens
- The DNA repair defects characteristic of BRCA1-deficient cells:
- Confer sensitivity to poly (adenosine diphosphate ribose) polymerase 1 (PARP) inhibition
- Platinum salts:
- Such as carboplatin and cisplatin:
- Cause DNA cross-link strand breaks and thus should be effective in BRCA-mutated calls and TNBC:
- Which are deficient in homologous DNA repair mechanisms
- Cause DNA cross-link strand breaks and thus should be effective in BRCA-mutated calls and TNBC:
- Such as carboplatin and cisplatin:
- The considerable overlap of the BRCA1 tumor phenotype and TNBC:
- Consideration should be given for neoadyuvant chemotherapy (NAC) for:
- Stages IIA, IIB, and IIIA and if the patient fulfills criteria for breast-conserving surgery apart from tumor size
- The NCCN guidelines recommend:
- The same NAC regimens for TNBC as for adjuvant treatment
- Pathologic complete response (pCR) rates:
- For TNBC are higher (22% to 44%) than for non-TNBC (6% to 11%)
- Residual disease following NAC:
- Portends a worse prognosis than a pCR
- Response to neoadjuvant therapy (adriamycin-, cytoxan-, taxol-containing regimens) differs according to intrinsic subtype:
- Masuda and colleagues noted a pCR of 28% overall:
- However, when looked at by subtype:
- BL1 subtype has a pCR of 52% versus:
- BL2 0%
- LAR 10%
- MSL (23%)
- BL1 subtype has a pCR of 52% versus:
- However, when looked at by subtype:
- Masuda and colleagues noted a pCR of 28% overall:
- TNBC subtype is an independent predictor of pCR following NAC:
- Tsai and colleagues determined that residual nodal disease post-NAC for TNBC was associated with an unfavorable outcome:
- They calculated the lymph node ratio (LNR) and the proportion of positive nodes over the number excised, following NAC according to molecular subtype:
- Patients with a low LNR (≤ 0.2) had a prolonged DFS compared with those with a high (> 0.65) LNR
- They calculated the lymph node ratio (LNR) and the proportion of positive nodes over the number excised, following NAC according to molecular subtype:
- Patients with TNBC:
- Have higher pCR rates in the axilla following NAC than patients with hormone receptor-positive cancers:
- 49.4% versus 21.1%
- Have higher pCR rates in the axilla following NAC than patients with hormone receptor-positive cancers:
- Further, a pCR determined by the sentinel node biopsy in the axilla may reduce the need for full axillary dissection in selected patients who achieve the pCR:
- Axillary downstaging to reduce the extent of axillary node dissection with NAC is an emerging paradigm
- Despite the relative chemosensitivity of TNBC:
- Early relapse is common:
- Leading to the term:
- Triple-negative paradox
- Leading to the term:
- TNBC tumors show increased rates of pCR to NAC:
- But have lower DFS and overall survival (OS) compared with other subtypes
- Early relapse is common:
- It has been postulated that the triple-negative paradox:
- Is due to differing pCR rates and higher likelihood of relapse in patients who do not achieve pCR
- Swisher and colleagues found that failure to achieve a pCR (in both the breast and axilla) following NAC:
- Was a significant adverse feature for patients with TNBC tumors
- With a median follow-up of 4.6 years:
- Those with a pCR the locoregional recurrence-free survival was:
- 98.6% versus 89.9% for those who did not achieve pCR (p = .007)
- Those with a pCR the locoregional recurrence-free survival was:
- For TNBC patients having residual disease following NAC:
- The use of AC following surgery has been associated with an improvement in survival
- The CREATE-X (Capecitabine for Residual Cancer as Adjuvant Therapy) trial:
- Was designed to study the effect of adjuvant capecitabine on patients with TNBC who did not achieve pCR following NAC
- Women in the capecitabine group:
- Had better 2-year:
- DFS:
- 82.8% versus 74.0%
- OS:
- 94% versus 89.2%
- DFS:
- Had better 2-year:
- In an ongoing clinical trial (SWOG 1418):
- The role of pembrolizumab (an immune checkpoint inhibitor) in patients with residual disease after NAC is being investigated
- Two landmark trials have shown the efficacy of platinum salts when added to NAC regimens for TNBC:
- The GeparSixto trial:
- Compared paclitaxel, doxorubicin, and bevacizumab (Avastin) with or without carboplatin
- In the TNBC subset:
- pCR improved from 37.9% to 58.7% with the addition of carboplatin
- In the Cancer and Leukemia Group B 40603 trial:
- Patients received paclitaxel 80 mg/m2 once per week for 12 weeks followed by doxorubicin plus cyclophosphamide once every 2 weeks for four cycles, and were randomly assigned to concurrent carboplatin (area under curve 6) once every 3 weeks for four cycles and / or bevacizumab 10 mg/kg once every 2 weeks for nine cycles
- The addition of either carboplatin (60% versus 44%; p = .0018) or bevacizumab (59% versus 48%; p = .0089) significantly increased pCR in the breast:
- Whereas only carboplatin (54% versus 41%; p = .0029) significantly raised pCR in both and axilla
- The GeparSixto trial:
- In a meta-analysis of eight platinum-based therapy trials for TNBC:
- Tian and colleagues concluded that both pCR and overall response rate:
- Were significantly higher for platinum-based regimens
- Tian and colleagues concluded that both pCR and overall response rate:
- In the I-SPY 2 trial:
- A phase 2, multicenter, adaptively randomized trial:
- Multiple experimental regimens in combination with standard NAC were screened
- Seventy-two patients with TNBC were randomly assigned to receive veliparib (a PARP inhibitor), carboplatin, and standard therapy, and 44 patients were concurrently assigned to receive control therapy
- The investigators found the estimated rates of pathologic complete response in the triple-negative population at the completion of chemotherapy were:
- 51% in the veliparib-carboplatin group versus 26% in the control group
- They concluded that veliparib-carboplatin had an 88% predicted probability of success in a phase 3 trial for TNBC
- A phase 2, multicenter, adaptively randomized trial:
#Arrangoiz #CancerSurgeon #BreastSurgeon #SurgicalOncologist #ComplexSurgicalOncology #BreastCancer #TripleNegativeBreastCancer #TNBC #Miami #Mexico #Teacher #Surgeon #MountSinaiMedicalCenter #MSMC
