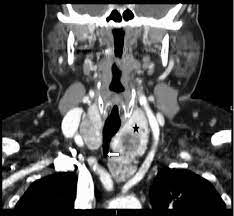
- Although rare, thyroid glands within the mediastinum may exist without connection to the normal cervical orthotopic gland:
- Such purely isolated mediastinal goiters represent only 0.2% to 3% of all goiters requiring surgical treatment
- Such lesions are important to recognize because unlike all other types of substernal goiters:
- Blood supply of the isolated mediastinal goiter may be through purely mediastinal arteries (including the aorta, subclavian, internal mammary, thyrocervical trunk, and innominate) and veins:
- This is extremely important in planning their surgical resection
- Blood supply of the isolated mediastinal goiter may be through purely mediastinal arteries (including the aorta, subclavian, internal mammary, thyrocervical trunk, and innominate) and veins:
- This entity is best termed isolated mediastinal goiter:
- Other terms have been used, including aberrant mediastinal and ectopic mediastinal goiter
- Three explanations exist for isolated mediastinal goiter:
- Embryologic fragmentation of the thyroid anlagen with hyperdescent, likely associated with cardiac and great vessel descent, may explain some cases of isolated mediastinal goiter
- Alternatively, isolated mediastinal goiter may form as an exophytic nodule, through progressive attenuation of the nodule-thyroid stalk
- Finally, the isolated mediastinal goiter may form as a parasitic nodule representing a thyroid tissue fragment implant in the upper mediastinum from past goiter surgery:
- I have seen such implants also within the peri-thyroid area and posterior to the upper cervical segment of the carotid artery
#Arrangoiz #ThyroidSurgeon #ThyroidExpert #HeadandNeckSurgeon #CancerSurgeon #HeadandNeckSurgeon #MultinodularGoiter #SubsternalGoiter #CASO #CenterforAdvancedSurgicalOncology