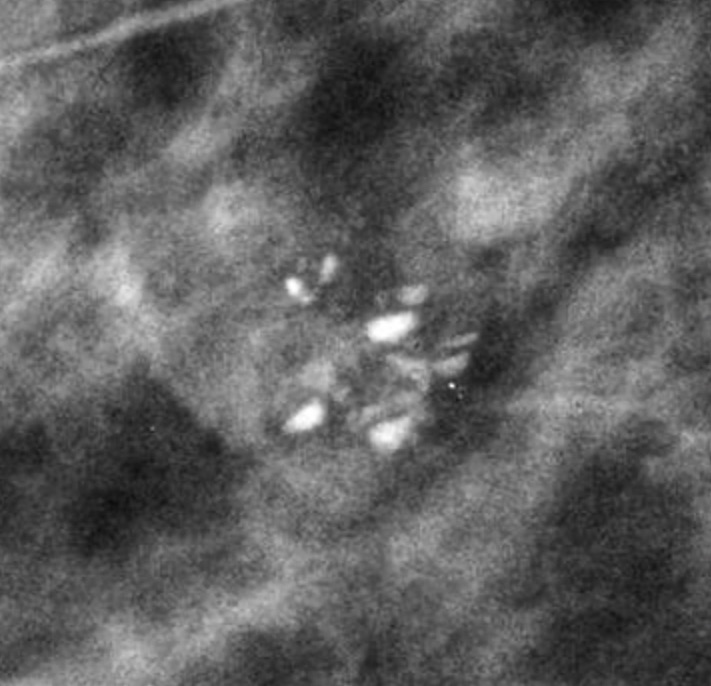
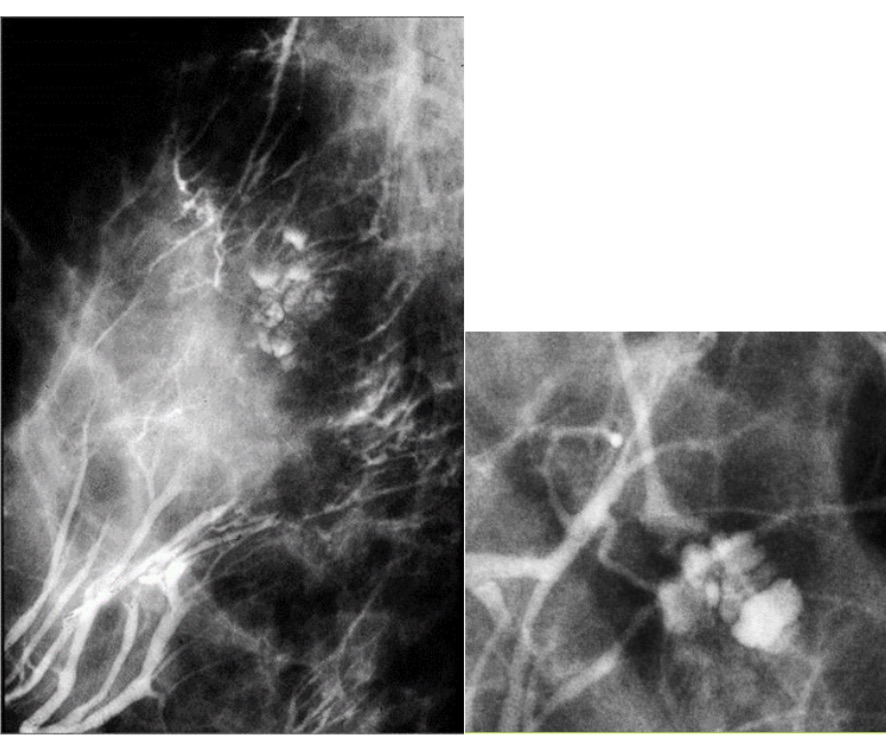
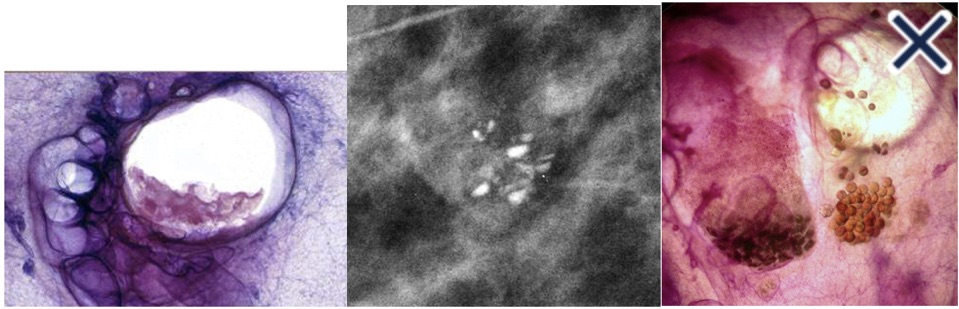
- Tissue harmonic imaging:
- Creates images derived solely from higher frequencies
- The ultrasound beam:
- Is transmitted centered at 1 frequency, e.g., 6 MHz
- Received centered at a multiple of the transmitted frequency, e.g., 12 MHz
- Different techniques can be used to process the received signals so that only the returning high-frequency harmonic signal is used to produce the image:
- Whereas echoes from the fundamental / lower frequencies are rejected
- THI increases signal-to-noise ratio:
- Resulting in better tissue contrast
- THI reduces reverberation, clutter, and speckle artifacts:
- Improving contrast resolution
- It accentuates real echoes in addition to suppressing artifactual echoes
- The suppression of speckle artifact by coded harmonics makes solid nodules more hypoechoic and conspicuous than they are with fundamental imaging (Images a and b)
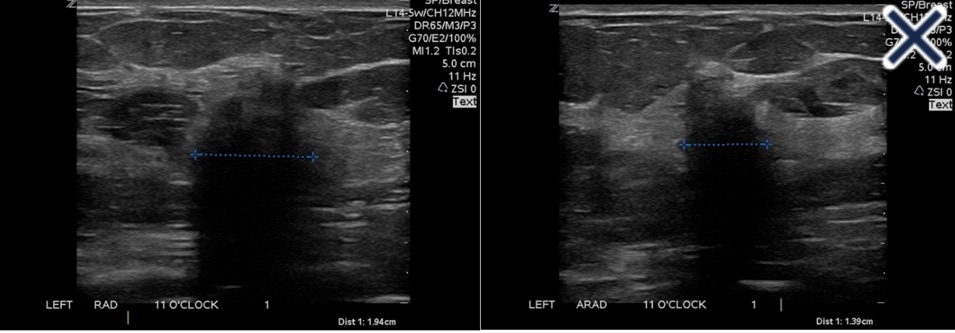
- It makes the thin, echogenic capsule that surrounds most benign lesions appear to be thinner, more echogenic and more complete than with fundamental imaging
- THI cannot be combined with simultaneous color Doppler imaging because the resulting frame rate would be unacceptable:
- When Doppler is required, the image that is interlaced with Doppler must be constructed at fundamental rather than harmonic frequencies
- THI is of limited value in differentiating benign from malignant lesions.
- Another method of reducing artifactual echoes, improving contrast, and making the thin, echogenic capsule more conspicuous is real time spatial compounding of images:
- In conventional imaging, each frame is created by a single sweep of the beam at a 90 degree angle to the long axis of the transducer
- In compound imaging, there are multiple sweeps of the beam from different angles, creating a spatially and temporally compounded image from multiple angles over time
- Among other things, the lateral borders of lesions can be seen better with compound imagin
- References
- Mesurolle B, Helou T, El-Khoury M, Edwardes M, Sutton EJ, Kao E. Tissue harmonic imaging, frequency compound imaging, and conventional imaging use and benefit in breast sonography. J Ultrasound Med. 2007;26(8):1041-1051.
- Cha JH, Moon WK, Cho N, Kim SM, Park SH, Han BK, et al. Characterization of benign and malignant solid breast masses: comparison of conventional US and tissue harmonic imaging. Radiology. 2007;242(1):63-69.
- Stavros AT. Breast ultrasound equipment requirements. In: Stavros AT. Breast Ultrasound. Philadelphia, PA: Lippincott Williams & Wilkins; 2004:16-41.