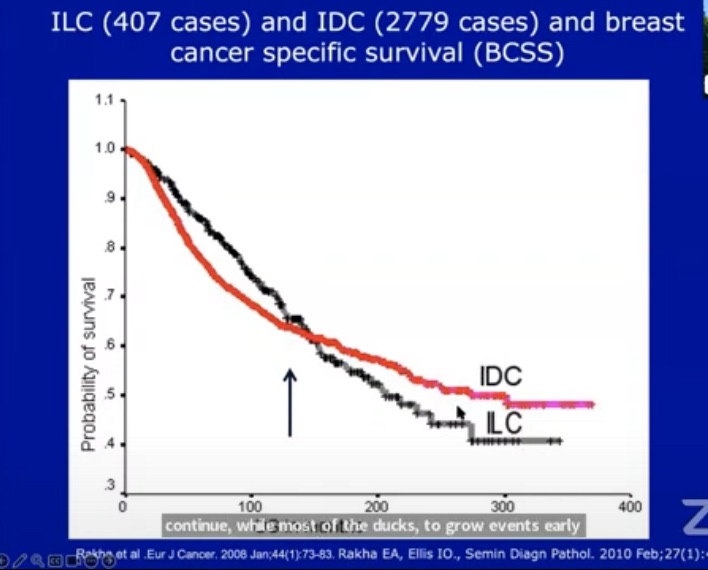
Paper summary (Eur Arch Otorhinolaryngol, 2026) — “The impact of drains on surgical outcomes in thyroid surgery”
This is a meta-analysis of randomized controlled trials comparing drain vs no drain after adult thyroid surgery (search Jan 1995–Aug 2025). It included 10 RCTs (n=1,078) and assessed haematoma/seroma (primary) plus SSI, return-to-theatre, pain, and length of stay.
Key findings
No significant difference with drains for: Haematoma (p=0.15) Seroma (p=0.64) Return-to-theatre (p=0.22) Drains were associated with worse outcomes: Higher SSI (4.2% vs 0.5%, p=0.01) Longer LOS (≈ +1.2 days, p<0.0001) More pain (MD ≈ +2.2, p=0.001)
Conclusion of the authors: routine drains don’t reduce clinically important collections/bleeding outcomes and should be selective/patient-specific.
Additional high-yield evidence on the same question
Systematic reviews
2017 meta-analysis (14 studies, n=1,927): drains increased infection and length of stay, with no significant differences in haematoma/seroma or RLN palsy/hypoparathyroidism. Cochrane review: highlights the key limitation of drains—they can block with clot and do not replace meticulous haemostasis / re-exploration when bleeding occurs; overall evidence did not support routine use.
Randomized trials (examples)
2013 RCT (Uganda, n=68): no-drain group had shorter LOS and less pain, with no signal that drains prevented important complications. 2023 RCT (lobectomy + central neck dissection, n=104): no routine drain needed; no-drain group had shorter LOS and better comfort metrics.
Evidence-based recommendation (practical)
1) Default position
For uncomplicated thyroidectomy/hemithyroidectomy, the best available RCT/meta-analysis evidence supports NO routine drain because it does not reduce haematoma/seroma and does increase SSI, pain, and LOS.
2) When a drain may be reasonable (selective use)
Consider a drain selectively when you believe a drain will meaningfully manage expected ongoing output or permit monitoring in a high-risk scenario, e.g.:
Extensive dissection / large dead space (e.g., combined procedures, broad flap elevation) Significant intraoperative oozing despite optimization (coagulopathy, difficult hemostasis) Reoperative thyroid surgery Very large goiter/substernal component (case-dependent) Neck dissection / lateral compartment work (many surgeons drain these by default; note: classic drain trials often exclude lateral neck dissections)
(Even in these settings, it’s worth emphasizing: drains don’t “prevent” a dangerous post-thyroidectomy hematoma—rapid recognition and evacuation remain key, and drains may clot off.)
3) What to do instead of routine drains (high-impact steps)
Meticulous hemostasis + Valsalva before closure Layered closure / dead-space minimization Standardized post-op neck checks and early warning protocol (swelling, tightness, voice change, stridor) Clear hematoma pathway (immediate bedside opening vs OR depending on severity/resources)