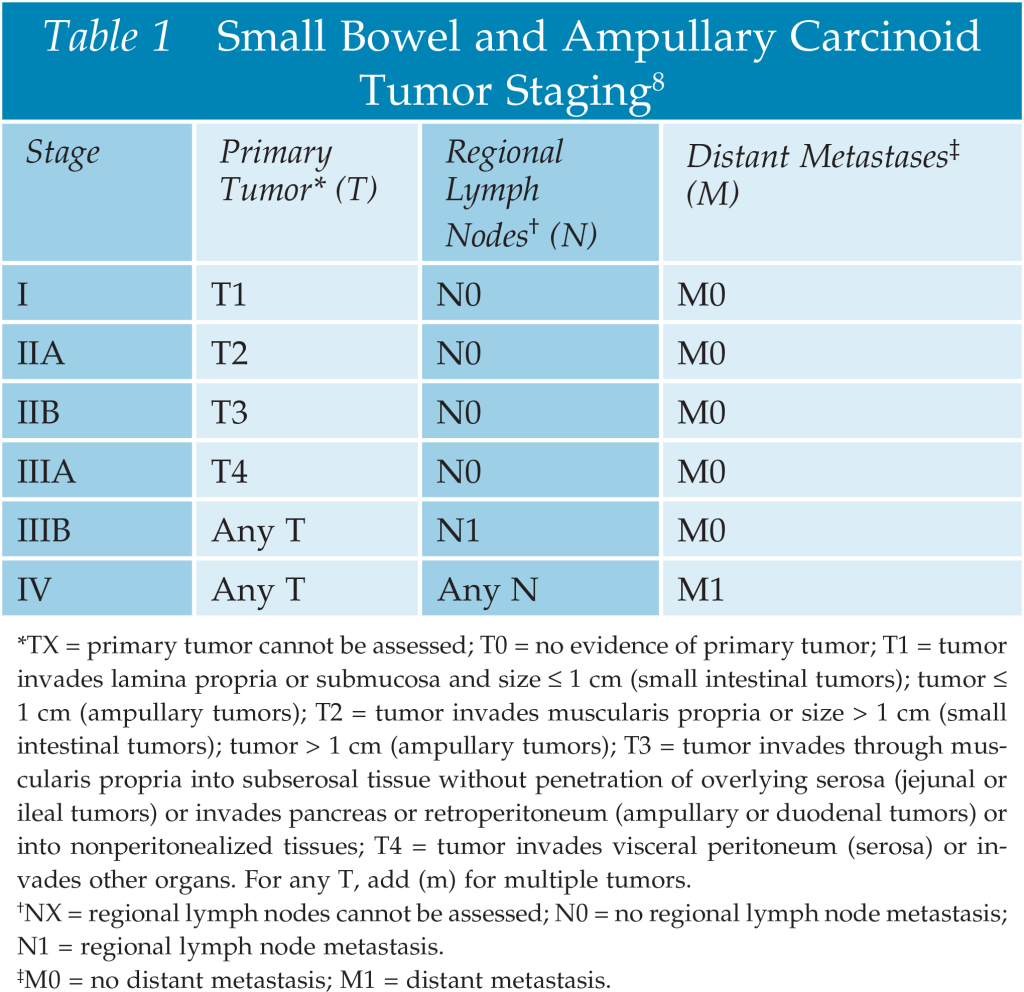
- Randomized Clinical Trials for DCIS
- Irene L. Wapnir, James J. Dignam, Bernard Fisher, Eleftherios P. Mamounas, Stewart J. Anderson, Thomas B. Julian, Stephanie R. Land, Richard G. Margolese, Sandra M. Swain, Joseph P. Costantino, Norman Wolmark
- Background:
- Ipsilateral breast tumor recurrence (IBTR):
- Is the most common failure event after lumpectomy for ductal carcinoma in situ (DCIS)
- Wapnir and colleagues evaluated invasive IBTR (I-IBTR) and its influence on survival:
- Among participants in two National Surgical Adjuvant Breast and Bowel Project (NSABP) randomized trials for DCIS:
- NSABP B-17
- NSABP B-24
- Among participants in two National Surgical Adjuvant Breast and Bowel Project (NSABP) randomized trials for DCIS:
- Ipsilateral breast tumor recurrence (IBTR):
- Methods:
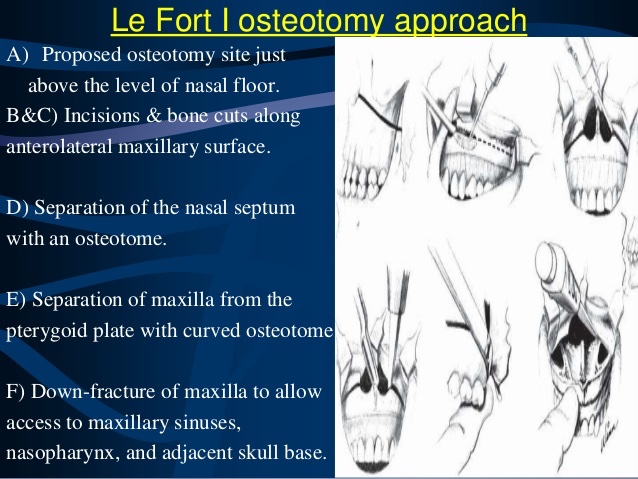
- In the NSABP B-17 trial:
- Accrual period:
- October 1, 1985, to December 31, 1990
- Accrual period:
- Patients with localized DCIS were randomly assigned to:
- The lumpectomy only (LO, n = 403) group or
- To the lumpectomy followed by radiotherapy (LRT, n = 410) group
- In the NSABP B-24 trial:
- Double-blinded, placebo-controlled trial
- Accrual period:
- May 9, 1991, to April 13, 1994
- All accrued patients were randomly assigned to:
- LRT+ placebo, (n=900) or
- LRT + tamoxifen (LRT + TAM, n = 899)
- Endpoints included:
- I-IBTR
- DCIS-IBTR
- Contralateral breast cancers (CBC)
- Over-all and breast cancer–specific survival
- Survival after I-IBTR
- Median follow-up was:
- 207 months for the B-17 trial:
- N = 813 patients
- 163 months for the B-24 trial:
- N = 1799 patients
- 207 months for the B-17 trial:
- In the NSABP B-17 trial:
- Results:
- Of 490 IBTR events:
- 263 (53.7%) were invasive
- Radiation reduced I-IBTR by:
- 52% in the LRT group compared with LO:
- NSABP B-17:
- Hazard ratio [HR] of risk of I-IBTR:
- 0.48, 95% confidence interval [CI] = 0.33 to 0.69, P < .001
- Hazard ratio [HR] of risk of I-IBTR:
- NSAPB B-24:
- LRT + TAM reduced I-IBTR:
- By 32% compared with LRT + placebo:
- HR of risk of I-IBTR:
- 0.68, 95% CI = 0.49 to 0.95, P = .025)
- HR of risk of I-IBTR:
- By 32% compared with LRT + placebo:
- LRT + TAM reduced I-IBTR:
- NSABP B-17:
- 52% in the LRT group compared with LO:
- The 15-year cumulative incidence of I-IBTR was:
- 19.4% for LO
- 8.9% for LRT (B-17)
- 10.0% for LRT + placebo (B-24)
- 8.5% for LRT + TAM
- The 15-year cumulative incidence of all contralateral breast cancers was:
- 10.3% for LO
- 10.2% for LRT (B-17)
- 10.8% for LRT + placebo (B-24)
- 7.3% for LRT + TAM
- I-IBTR:
- Was associated with increased mortality risk:
- HR of death = 1.75, 95% CI = 1.45 to 2.96, P < .001
- Whereas recurrence of DCIS was not
- Twenty-two of 39 deaths after I-IBTR were attributed to breast cancer
- Was associated with increased mortality risk:
- Among all patients (with or without I-IBTR), the 15-year cumulative incidence of breast cancer death was:
- 3.1% for LO
- 4.7% for LRT (B-17)
- 2.7% for LRT + placebo (B-24)
- 2.3% for LRT + TAM
- Of 490 IBTR events:

#Arrangoiz #BreastSurgeon #CancerSurgeon #BreastCancer #Teacher #Surgeon