- Clin Thyroidol 2021;33:184–186.
- Background
- Disease-specific survival for differentiated thyroid cancer (DTC) is lengthy and most profoundly impacted by:
- Patient age (55 years)
- Distant metastases
- Extent of gross extrathyroidal extension (ETE)
- Nodal status
- Microscopic margin positivity (R1 margin):
- Has shown no impact on local recurrence, but it has not been studied as:
- A marker of completeness of resection
- Has shown no impact on local recurrence, but it has not been studied as:
- This National Cancer Database (NCDB) study evaluated the impact of R1 margin on overall survival in DTC to determine the presence of modifiable factors such as institutional annual thyroidectomy volume and hospital setting of thyroidectomy
- Disease-specific survival for differentiated thyroid cancer (DTC) is lengthy and most profoundly impacted by:
- Methods
- The NCDB was queried for adults (ages 18 to 90) who underwent surgery from 2004 to 2016 for treatment of well- and moderately-differentiated thyroid carcinoma with a primary tumor size of 1 to 4 cm
- Patients with histologically aggressive variants were excluded
- Patients with gross ETE into the strap muscles (T3b); subcutaneous soft tissues, larynx, trachea, esophagus, or recurrent laryngeal nerve (T4a); or invading prevertebral fascia, encasing carotid or mediastinal vessels (T4b), or nodal disease were included if primary tumor size was less than 4 cm
- Binary logistic-regression analyses evaluated factors affecting margin positivity after adjusting for confounders including age, gender, comorbidity score (modified Charlson–Deyo), extent of thyroid operation (lobectomy or total), margin status, lymphovascular invasion, multifocality, institutional annual case volume for thyroidectomy, and institu- tion type (community, comprehensive community, academic, or integrated network
- Results
- This study included 14,471 patients with DTC (median age, 47.52 years [range, 18–90]; 74.7% female)
- After correcting for confounders, disease-specific (nonmodifiable) factors impacting survival included:
- ETE:
- 25.2% of patients; OR, 1.47; P = 0.001), lateral neck nodal disease (12.6%; OR, 1.73; P = 0.001
- Lymphovascular invasion:
- 15.7%; OR, 1.32; P = 0.044
- R1 margin:
- 15.3%; OR, 1.46; P = 0.038
- ETE:
- Modifiable factors impacting survival included:
- Institutional case volume and type of facility:
- Overall, institutions had a mean case volume of 30.3 thyroidectomies per year (range, 4 to 485):
- When evaluating R1 margin status as a modifiable risk factor for survival:
- Treatment in academic / research facilities (OR, 0.623; 95% CI, 0.527–0.738; P<0.001), integrated networks (OR, 0.782; 95% CI, 0.654–0.934; P = 0.009), or facilities with higher case volumes (OR, 0.979; 95% CI, 0.978–0.982; P = 0.004):
- Was associated with lower odds of R1 margin
- Treatment in academic / research facilities (OR, 0.623; 95% CI, 0.527–0.738; P<0.001), integrated networks (OR, 0.782; 95% CI, 0.654–0.934; P = 0.009), or facilities with higher case volumes (OR, 0.979; 95% CI, 0.978–0.982; P = 0.004):
- When evaluating R1 margin status as a modifiable risk factor for survival:
- Overall, institutions had a mean case volume of 30.3 thyroidectomies per year (range, 4 to 485):
- Institutional case volume and type of facility:
- This study was sufficiently powered to detect a small impact of R1 margin on 8-year survival:
- OR, 1.464; 95% CI, 1.039–2.121; P = 0.038
- Conclusions
- In this large database study:
- R1 margin in DTC impacted overall survival
- Modifiable risk factors associated with decreased risk of R1 margin were:
- Treatment in an academic / research facility or integrated network and higher institutional annual case volume
- In this large database study:
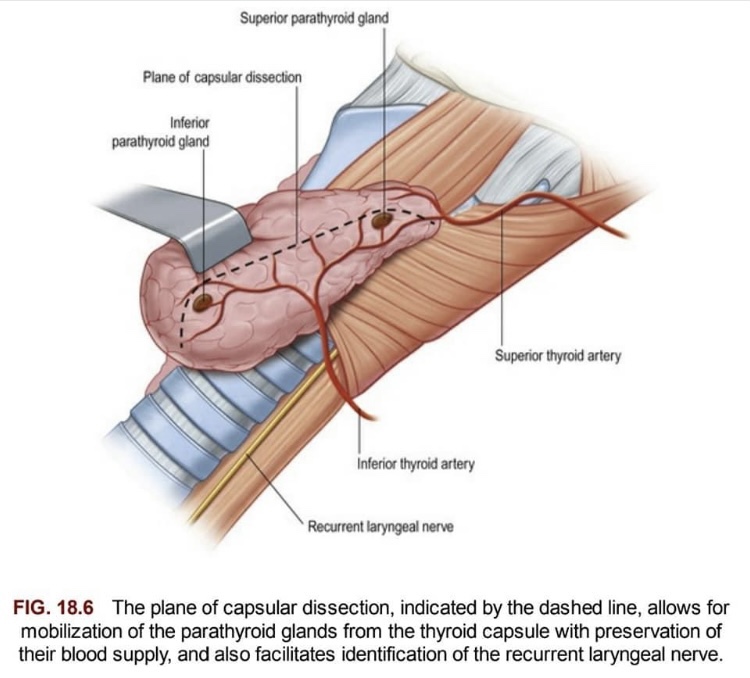
- As the thyroid gland has an incomplete capsule:
- Interpreting an R1 margin as a prognostic indicator in DTC has been the source of some confusion
- A meta-analysis of patients with DTC:
- Found no association between R1 margin and local recurrence
- This study aimed to evaluate the impact of negative versus R1 margins in a more homogeneous population of patients with smaller DTCs
- The major modifiable factor in this study was care setting:
- Higher-volume thyroid surgeons are more likely to operate in academic settings
- Worldwide, higher individual surgeon volume for thyroidectomy (30 to 100 cases / year) is protective against complications and yields better oncologic outcomes
- A similar NCDB study with a more heteroge- neous thyroid cancer population confirmed that treatment in an academic center was associated with a lower probability of R1 margin and higher overall survival
- Rather than contradicting prior studies about the seeming irrelevance of R1 margin in DTC:
- This study highlights how R1 margin may be a marker of incomplete resection when thyroidectomy is performed in low-volume centers
- When I receive pathologic “positive margins,” assuming no ETE was present intraoperatively:
- I generally reassure the patient that all tumor was appropriately resected:
- However, this study raises important questions about how to interpret pathology reports documenting R1 margin from lower-volume centers
- I generally reassure the patient that all tumor was appropriately resected:
- In contrast to this study’s findings of improvement in mean overall survival with negative margins:
- Prior retrospective studies have not found that R1 margin impacts overall survival
- Although this study corrected for confounders:
- ETE was the variable most strongly correlated with an R1 margin (OR, 2.8):
- ETE decreases survival in thyroid cancer, and it makes sense that ETE would be a natural continuation of R1 margins
- However, further research is needed to determine the prognostic significance of thyroid cancers with positive inked margins without a finding of gross ETE
- ETE decreases survival in thyroid cancer, and it makes sense that ETE would be a natural continuation of R1 margins
- ETE was the variable most strongly correlated with an R1 margin (OR, 2.8):

#Arrangoiz #CancerSurgeon #SurgicalOncologist #HeandandNeckSurgeon #ThyroidSurgeon #EndocrineSurgery #ThyroidExpert #ThyroidCancer #CASO #CenterforAdvancedSurgicalOncology