- Tumor Size:
- Low risk:
- Tumors less than 2 cm in greatest dimension
- High Risk:
- Tumors equal or greater than 2 cm in greater dimension
- For cSCC tumors with diameter less than 2 cm:
- Risk stratification by location and size:
- Is largely based on extrapolation from older data in BCC:
- This 27-year retrospective review of 5755 BCCs showed that high-risk sites correspond roughly to the:
- Mask areas of the face, and that recurrences after standard excision or curettage and electrodesiccation (C&E) were significantly more common:
- When tumors in high-risk locations:
- Were 6 mm or more in diameter
- When tumors in moderate-risk locations:
- Were 10 mm or more in diameter
- When tumors in high-risk locations:
- Therefore, for the purpose of determining which tumors should be removed with Mohs micrographic surgery (MMS) or excision with complete circumferential peripheral and deep margin assessment (CCPDMA) rather than standard excision or C&E, the NCCN panel has defined the following as high risk:
- Tumors in area L (low-risk region) that are ≥ 20 mm
- Tumors in area M (moderate-risk region) that are ≥ 10 mm
- Tumors in area H (high-risk region) of any size
- Mask areas of the face, and that recurrences after standard excision or curettage and electrodesiccation (C&E) were significantly more common:
- This 27-year retrospective review of 5755 BCCs showed that high-risk sites correspond roughly to the:
- Is largely based on extrapolation from older data in BCC:
- Risk stratification by location and size:
- Low risk:
- Location:
- Low risk:
- Trunk
- Extremities
- High risk:
- Mucosal surfaces
- Genitalia
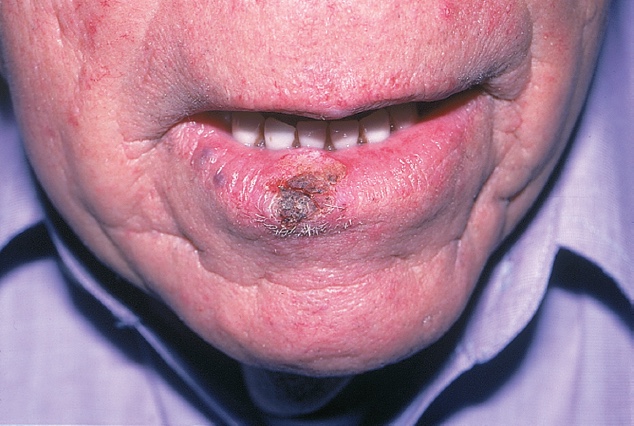
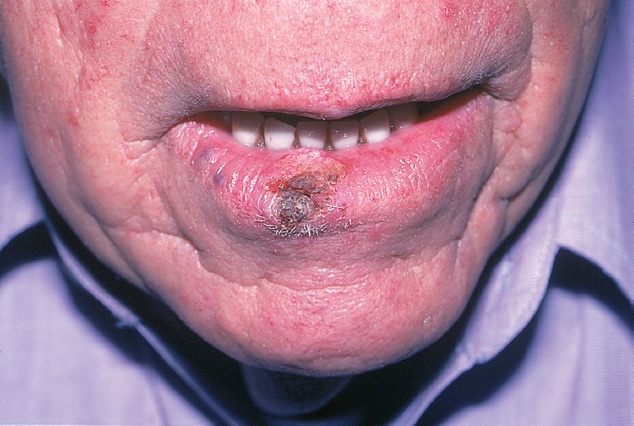
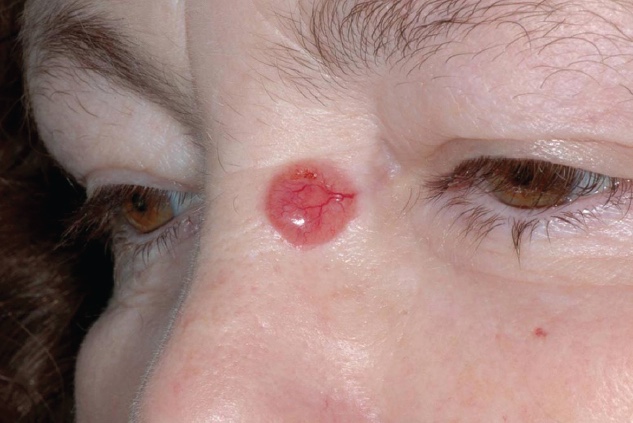
- Mask areas of face including:
- Eyelids
- Periorbital
- Nose
- Lips, chin
- Preauricular areas
- Postauricular areas
- Ear
- Prior burn scars
- Irradiated areas
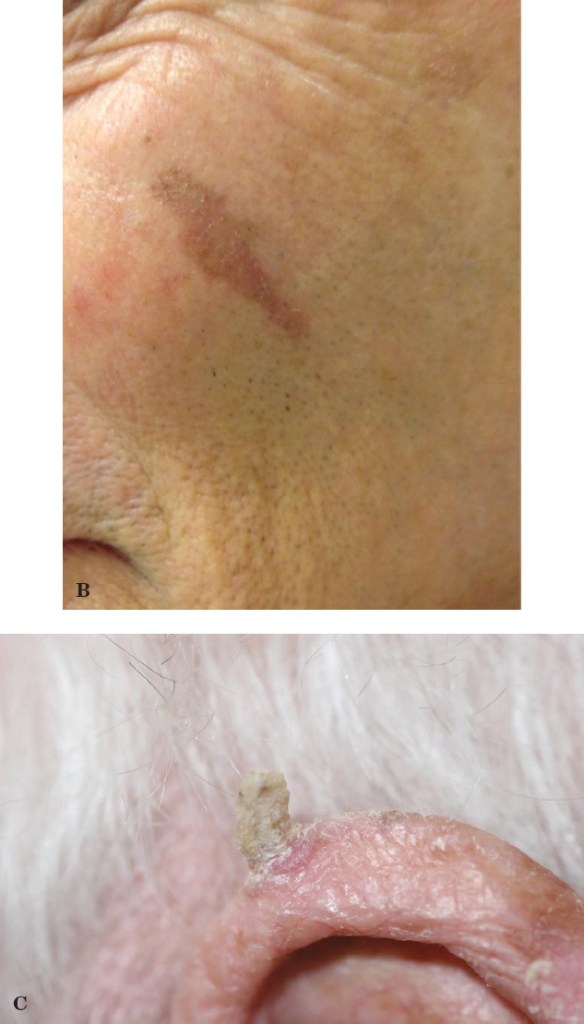
- In general, cSCCs that develop in the head and neck area:
- Are more likely to recur than those developing on the trunk and extremities
- SCCs that develop on the genitalia, mucosal surfaces, and ears:
- Are also at greater risk of metastasizing
- The concept of a so-called high-risk “mask area of the face” dates back at least to 1983
- Low risk:
- Borders:
- Low risk:
- Well defined
- High risk:
- Ill defined
- Low risk:
- Arising from scar or inflammation:
- Low risk:
- No
- High risk:
- Yes
- Low risk:
- Prior irradiation
- Low risk:
- No
- High risk:
- Yes
- Tumors developing in sites of prior radiotherapy refer to:
- Primary cSCCs arising in areas previously irradiated for unrelated conditions
- All recurrent tumors:
- Irrespective of prior therapy:
- Are defined as high risk
- Irrespective of prior therapy:
- Data from older studies and one more recent study support that prior radiotherapy for unrelated (frequently benign) conditions:
- Is a risk factor for NMSC recurrence or metastasis
- Low risk:
- History of inmunosuppression:
- Low risk:
- No
- High risk:
- Yes
- In addition to increasing the risk of cSCC development:
- Immunosuppression has been shown to be associated with:
- Poorer outcomes in large meta-analyses, and prospective, and retrospective studies:
- Each of these studies showed that immunosuppression was associated with at least one measure of poor outcome (recurrence, metastasis, or death), but results are inconsistent regarding which of these outcomes are effected
- These studies cover a broad range of extent of disease and treatment approaches:
- Suggesting that immunosuppression is associated with poor prognosis regardless of treatment approach used or the stage of disease at time of treatment
- A few studies from the organ transplant literature have evaluated other risk factors that might be linked to the higher rates of SCC recurrence and metastasis among transplant patients:
- A retrospective review of 307 patients with cSCC confirmed that those who received organ transplants had more aggressive disease than those who did not, and that SCCs in transplant patients were more likely to have deep tissue spread and perineural and lymphatic invasion at presentation
- Poorer outcomes in large meta-analyses, and prospective, and retrospective studies:
- Immunosuppression has been shown to be associated with:
- Low risk:
- Primary vs recurrent:
- Low risk:
- Primary
- High risk:
- Recurrent
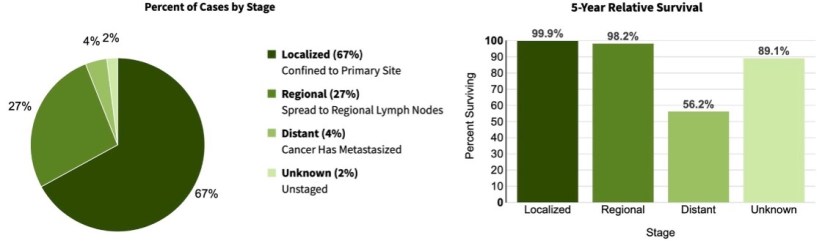
- The higher risk of recurrence and metastasis for recurrent cSCC versus primary cSCC disease has been extensively documented in the literature
- Low risk:
- Degree of differentiation:
- Low risk:
- Well differentiated
- High risk:
- Poorly differentiated
- In their extensive meta-analysis of risk factors for local recurrence and metastasis of cSCC:
- Rowe and colleagues found that patients with well-differentiated tumors fared significantly better than those patients with poorly differentiated lesions
- Another cohort study of 315 patients also associated differentiation grade with overall survival:
- Erglo and colleagues colleagues reported differentiation to be a significant risk factor of recurrence in an analysis of 1039 patients
- Many other studies, including some very large retrospective studies (n > 1000) provide
supporting evidence that:- Poor differentiation is correlated with cSCC recurrence, metastasis, disease-specific survival, and overall survival
- Another high-risk histologic feature reported in the literature is:
- The presence of desmoplasia:
- In studies from Germany, desmoplastic cSCC was shown to pose a greatly increased risk of both recurrence and metastasis
- A retrospective study using the PALGA national registry of the Netherlands reported significantly higher rates of metastasis for desmoplastic versus non-desmoplastic cSCCs:
- 89% versus 21% (P < .001)
- The significance of desmoplasia as a risk factor was confirmed by multivariable analysis
- A more recent review of 72 patients with desmoplastic SCC:
- Reported a high rate of recurrence of 80%
- The presence of desmoplasia:
- Low risk:
- Perineural invasion:
- Low risk:
- Not present
- Hight risk:
- Present
- Perineural involvement is uncommon in any NMSC (2% to 6% of the cases):
- But develops more frequently and is more aggressive in cSCC versus BCC
- cSCC with perineural involvement poses a greatly increased risk of:
- Recurrence
- Metastasis (nodal and distant)
- Death
- It is more common in recurrent versus primary tumors:
- It is associated with other risk factors, including:
- Larger lesion size
- Poor differentiation
- Adenosquamous (mucin producing)
- Desmoplastic
- Metaplastic (carcinosarcomatous)
- It is associated with other risk factors, including:
- In a prospective cohort study of 315 patients with cSCC of the head and neck, Kyrgidis and colleagues identified perineural involvement as a factor associated with lower overall survival and recurrence-free survival
- Even among patients with other risk factors:
- PNI is an independent risk factor for lymph node metastasis:
- As demonstrated by multivariate analysis of a prospective study of high-risk patients
- PNI is an independent risk factor for lymph node metastasis:
- If large nerve involvement is suspected:
- MRI should be considered to evaluate extent and / or rule out skull involvement in those with head and neck tumors
- cSCC involving unnamed small nerves (< 0.1 mm in caliber):
- May have a low risk of poor outcomes in the absence of other risk factors
- Low risk:
- Lymphovascular invasion:
- Low risk:
- Not present
- Hight risk:
- Present
- Two prospective studies of patients with local or regional cSCC have shown significant association between lymphovascular invasion (LVI) and lymph node metastasis
- Retrospective studies corroborate this finding and also show that LVI is:
- Prognostic for parotid involvement
- Retrospective studies corroborate this finding and also show that LVI is:
- One retrospective study of 114 patients with cSCC and PNI or neurotropism showed by multivariate analysis that in this high-risk population:
- LVI was significantly associated with disease- specific death and all-cause death
- The presence of LVI from the primary tumor is important to note:
- Especially in patients with other risk factors, and should increase the suspicion of subclinical nodal disease
- Low risk:
- Depth of invasion
- Low risk:
- Less than 2 mm
- Less than Clark level IV
- High risk:
- Equal or greater than 2 mm
- Clark level IV / V
- Data from many large studies support that risk of recurrence and metastasis:
- Increases with increasing lesion depth
- cSCC lesion depth can be quantified as thickness in mm or by anatomic layer(s) invaded:
- A standard Breslow measurement per the College of American Pathologists (CAP) 2013 protocol:
- Would be the distance from the deepest point of tumor invasion to the granular layer or base of ulcer if present, excluding parakeratosis or scale crust
- Brantsch and colleagues prospectively examined potential risk factors for metastasis and local recurrence of cSCC in 615 patients over a 20- year period:
- With a median follow-up of 43 months, metastasis occurred in:
- 0% of tumors 2.0 mm in thickness
- 4% of tumors 2.1 mm to 6.0 mm in thickness
- 16% of tumors thicker than 6.0 mm
- With a median follow-up of 43 months, metastasis occurred in:
- Thicker lesions also had a higher risk of local recurrence:
- A large retrospective analysis and a very large meta-analysis (n = 17,248) provide data supporting that risk of recurrence and metastasis is significantly higher:
- For lesions with thickness > 2 mm
- A large retrospective analysis and a very large meta-analysis (n = 17,248) provide data supporting that risk of recurrence and metastasis is significantly higher:
- Meta-analyses have shown that 4-mm and 6-mm thickness cutoffs:
- Are also prognostic for recurrence and metastasis, and one retrospective study showed by multivariate analysis that risk for recurrence and metastasis:
- Increases significantly for every 1-mm increase in tumor depth
- Retrospective studies and meta-analyses support that anatomic level of invasion is significantly correlated with cSCC recurrence and metastasis:
- Some studies showed significantly higher risk of recurrence or metastasis for cSCC lesions with Clark levels IV-V:
- Corresponding to invasion of the deep reticular dermis or subcutaneous fat,
respectives
- Corresponding to invasion of the deep reticular dermis or subcutaneous fat,
- Other studies have shown that lesions with invasion
into the subcutaneous fat:- Significantly increases rates of recurrence and metastasis
- Some studies showed significantly higher risk of recurrence or metastasis for cSCC lesions with Clark levels IV-V:
- Are also prognostic for recurrence and metastasis, and one retrospective study showed by multivariate analysis that risk for recurrence and metastasis:
- A standard Breslow measurement per the College of American Pathologists (CAP) 2013 protocol:
- Low risk:
- Subtype:
- Low risk:
- N/A
- High risk:
- Bowen disease
- Desmoplastic SCC
- Acantholytic SCC
- Adenosquamous SCC
- Basosquamous SCC
- The histologic subtypes of acantholytic (adenoid), adenosquamous (or mucin-producing), and metaplastic (carcinosarcomatous) SCC are rare histologic subtypes:
- Only case reports and case series document the outcomes of patients with these subtypes, and thus their prognostic significance is debated:
- However, because these tumors may have a high risk of recurrence and likely would not be included in the high-risk category on the basis of their degree of differentiation, the panel decided to list them as separate risk factors
- Only case reports and case series document the outcomes of patients with these subtypes, and thus their prognostic significance is debated:
- Low risk:
#Arrangoiz #CancerSurgeon #HeadandNeckSurgeon #SurgicalOncologist #CASO #SkinCancer #CenterforAdvancedSurgicalOncology #BCC #SCC