Treatment of Obstructive or Substernal Goiter
• Goiter refers to:
• Abnormal growth of the thyroid gland
• Patients with longstanding goiters (cervical or substernal):
• May develop symptoms of obstruction due to:
• Progressive compression of the trachea or
• Sudden enlargement (usually accompanied by pain):
• Secondary to hemorrhage into a nodule
• The most common obstructive symptom is:
• Exertional dyspnea:
• Which is present in 30% to 60% of the cases:
• Usually occurs when the tracheal diameter:
• Is under 8 mm
• Substernal goiter:
• May be detected incidentally on chest radiograph or computed tomography (CT) scan or
• Found because of obstructive symptoms such as:
• Dyspnea, wheezing, or cough
• Anatomic Relationships:
• Enlarging thyroid lobes usually grow outward:
• Because of their location in the anterior neck in front of the trachea, covered only by thin muscles, subcutaneous tissue, and skin
• In patients with substantial enlargement of one lobe or asymmetric enlargement of both lobes:
• The trachea, esophagus, or blood vessels may be displaced or, less often, compressed
• Bilateral lobar enlargement:
• Especially if the goiter extends posterior to the trachea;
• May cause either:
• Compression or concentric narrowing of the trachea or
• Compression of the esophagus or jugular veins
• With some goiters, there is growth of one or both lobes through the thoracic inlet into the thoracic cavity;
• Which can result in obstruction of any of the structures in the inlet:
• Such goiters are referred to as substernal
• The goals of therapy:
• Once goiter is detected (on physical exam or incidentally during a radiologic procedure performed for other purposes):
• An evaluation is performed to assess:
• Thyroid function and to identify the underlying cause
• Presence of obstructive symptoms
• Presence of suspicious sonographic features in nodules within the goiter:
• These factors determine management
• For patients with obstructive goiter (cervical or substernal):
• The goal of management is to relieve the obstructive symptoms by resecting or ablating the thyroid gland
• For patients with asymptomatic substernal goiter:
• The goal is to prevent growth and subsequent development of obstructive symptoms
• Obstructive symptoms:
• Patients with obstructive symptoms from a cervical or substernal goiter:
• Require removal or ablation of the thyroid
• Once obstructive symptoms are present:
• There is risk of further thyroid growth and progressive tracheal compression:
• Which in some instances (eg, hemorrhage) may be rapid and fatal
• For the treatment of obstructive goiter:
• Surgery rather than radioiodine ablation is preferred:
• For patients with obstructive symptoms who are unable or unwilling to go undergo surgery:
• Radioiodine therapy is an alternative option:
• However, the reduction in thyroid volume with radioiodine is only moderate, and there are theoretical concerns that radioiodine could acutely worsen obstruction or a missed malignancy
• Most obstructive cervical or substernal goiters are benign:
• However, patients with nodules within a goiter that are malignant or suspicious for malignancy on fine-needle aspiration (FNA) biopsy require surgery
• Asymptomatic substernal goiter:
• The main treatment options for patients with asymptomatic substernal goiter are:
• Surgery or observation with monitoring:
• The choice of therapy depends upon the extent of substernal extension and patient characteristics:
• Someauthors suggest surgery for most patients with asymptomatic substernal goiters:
• That extend below the level of the brachiocephalic vein:
• However, this is an area of controversy, and other experts prefer to monitor such patients
• The arguments for removing a substernal goiter in a patient who has no obstructive symptoms include:
• Some goiters continue to enlarge and become more difficult to remove if obstructive symptoms do develop
• Suppressive therapy is relatively ineffective and is associated with significant morbidity in older patients
• Forty-two percent of patients with evidence of upper airway obstruction on flow-volume loops are asymptomatic
• As patients age, surgical complications are more common and severe
• The substernal component could contain a cancer that cannot be palpated or biopsied:
• Range of reported cancer risk 3% to 22%
• There is a small risk of hemorrhage into the goiter that could result in acute airway obstruction
• Observation rather than surgery is recommended by some authors for:
• Asymptomatic patients with normal flow-volume loops whose goiters end at the level of the brachiocephalic vein or higher:
• These patients are monitored with serial computed tomography (CT) scans:
• Initially after one year and, if stable, at increasing intervals (eg, two years later, then three years, then five years)
• Other asymptomatic patients with substernal goiter who may be followed with serial CT scans, rather than undergo surgery, include the following:
• Older patients who are poor operative candidates
• Patients without thyroid enlargement whose glands extend slightly substernally due to kyphosis
• Patients whose goiters extend only slightly substernally on a CT scan obtained without neck extension may not be substernal when the neck is extended:
• Their glands are also usually accessible for FNA biopsy if indicated
• Patients who, in retrospect, have serial CT scans showing long-term stability of a substernal goiter, especially if the inferior extent is above the level of the brachiocephalic vein:
• However, if growth is detected on serial CT scans;
• Surgery is recommended if possible, for these patients also
• Levothyroxine may have a limited role in patients with asymptomatic substernal goiters:
• In a randomized trial, thyroid-stimulating hormone (TSH)-suppressive doses of levothyroxine:
• Reduced goiter size in approximately two-thirds of patients with sporadic, multinodular goiters:
• However, most large goiters do not shrink sufficiently to alleviate symptoms, any reduction in size is not rapid, and levothyroxine is not effective in those patients who already have low serum TSH concentrations (ie, subclinical hyperthyroidism)
• Additionally, growth of the goiter may resume as soon as levothyroxine treatment is stopped
• For the treatment of obstructive goiter:
• Surgery rather than radioiodine is recommended
• In addition, patients with nodules within a goiter that are malignant or suspicious for malignancy on fine-needle aspiration (FNA) biopsy require surgery
• Some authors also suggest surgery for most patients with asymptomatic, substernal goiters:
• That extend below the level of the brachiocephalic vein:
• Assuming they are good surgical candidates
• Patients with substernal goiter should be referred to an experienced thyroid surgeon:
• As complication rates appear to be lower in high-volume centers
• Patients who may require sternotomy:
• Should be referred to an experienced thoracic surgeon
• Preoperative assessment:
• The initial evaluation of substernal goiter includes:
• Serum TSH;
• To evaluate for subclinical or overt hyperthyroidism
• Imaging studies (noncontrast computed tomography [CT] or magnetic resonance imaging [MRI]):
• To evaluate the extent of the goiter and its effect upon surrounding structures
• A flow-volume loop study:
• If there is uncertainty if the goiter is causing upper airway obstruction
• FNA biopsy if malignancy is suspected:
• Most nodules within goiters have benign thyroid aspirates
• The decision to proceed with surgery is usually made on:
• The basis of symptoms
• Anatomic studies
• The results of a flow-volume loop study
• Once the decision to proceed with surgery has been made, other preoperative assessment of substernal goiter should include:
• Laryngoscopy:
• To assess the tracheal lumen and vocal cord function
• If the patient is hyperthyroid and surgery is elective:
• An antithyroid drug and, if not contraindicated, a beta blocker should be given for several weeks before surgery:
• Patients with subclinical hyperthyroidism do not need to be prepared with an antithyroid drug
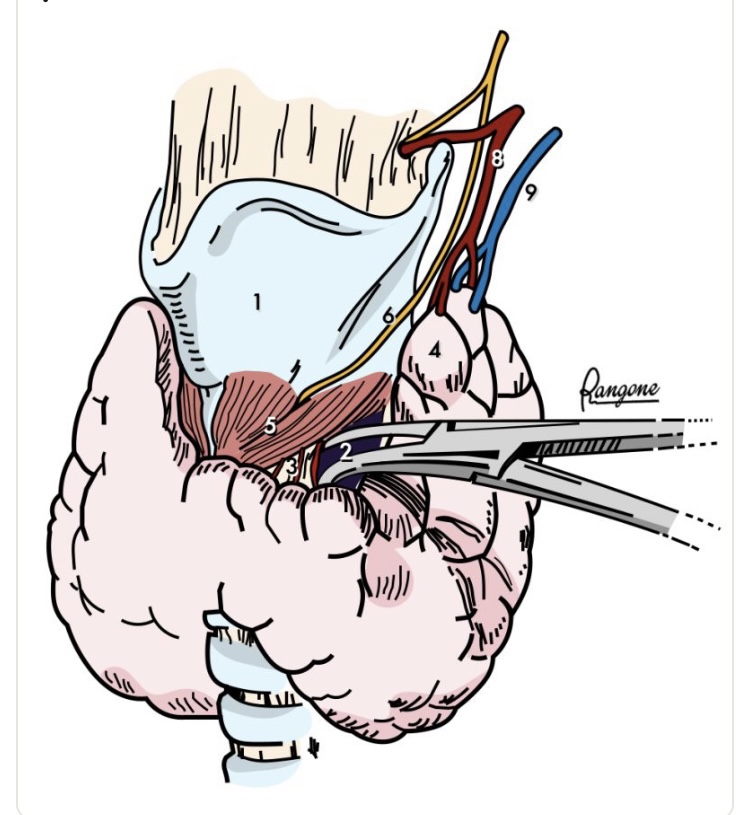
• Surgical approach:
• The majority of obstructive and substernal goiters can be excised through a standard cervical incision:
• While partial or complete sternotomy or even thoracotomy may be required in patients with:
• Previous cervical thyroidectomy
• Very large substernal goiters
• Invasive cancer
• In an Italian study of 19,662 patients undergoing total thyroidectomy at six centers, 1055 had substernal goiters and only 69 (6.5%) required sternotomy:
• Patients requiring sternotomy were more likely to have a malignancy: 36% compared with 22% of those excised through a cervical incision
• Similar results were noted in two single-center studies and in a systematic review
• Recurrent or ectopic goiter in addition to malignancy:
• Were associated with the need for sternotomy
• The extent of surgery for benign goiter:
• Depends upon the expertise of the surgeon
• The extent of the goiter
• Whether the patient has obstructive symptoms and signs
• Because of the risk of recurrent goiter:
• A total or near-total thyroidectomy should be performed unless during the procedure the surgeon feels that a less extensive operation is prudent because of an increased risk of recurrent laryngeal nerve injury or hypoparathyroidism due to anatomic considerations
• If a more limited operation is done, we suggest:
• Patients with large, relatively symmetric goiters:
• Should have a bilateral subtotal thyroidectomy
• Patients with asymmetric goiters:
• Should have a hemithyroidectomy on the more involved side and a subtotal thyroidectomy on the contralateral side
• If the patient already has vocal cord paralysis on the one side and the contralateral thyroid lobe is not very large:
• We suggest doing only a hemithyroidectomy and isthmusectomy
• In patients with chronic autoimmune thyroiditis who have concentric tracheal compression:
• Excision of the isthmus alone may be sufficient to alleviate the compression
• These fibrous glands may be difficult to dissect free from nerves and parathyroid glands, and more extensive surgery may result in complications
• Complications:
• The major complications of surgery for large goiters and substernal goiters are:
• Injury to the recurrent laryngeal nerves
• Trachea
• Parathyroid glands
• Surgery for substernal goiter appears to be associated with higher complication rates than surgery for cervical goiter:
• As illustrated by a statewide database of cervical (n = 32,777) and substernal thyroidectomies (n = 1153) performed between 1998 and 2004:
• Patients who underwent substernal thyroidectomy were more likely to be:
• Older
• Have a comorbid condition
• Be uninsured
• Be undergoing total thyroidectomy
• Have surgery at a low-volume center
• After adjusting for these variables:
• Patients undergoing substernal thyroidectomy were still at higher risk for the following:
• Recurrent laryngeal nerve injury (odds ratio [OR] 2.4, 95% CI 1.5-3.8)
• Postoperative bleeding (OR 1.9, 95% CI 1.2-2.9)
• Deep venous thrombosis (OR 5.9, 95% CI 2.4-15.2)
• Respiratory failure (OR 4.2, 95% CI 2.8-6.2)
• Red blood cell transfusion (OR 5.7, 95% CI 3.8-8.5)
• Mortality (OR 8.3, 95% CI 4.2-16.3)
• In the same database, complication rates were significantly lower at hospitals that performed a high volume of substernal thyroidectomies
• Of note, this study did not report how many patients with substernal goiter required sternotomy, a procedure that may be associated with higher surgical complication rates
• In the Italian study of 1055 patients with substernal goiter, 69 patients who required a sternotomy were compared with 986 patients whose goiters were excised through a cervical incision:
• Only phrenic nerve palsy was more common in the sternotomy group
• In a study from the National Surgical Quality Improvement Program database of 2716 patients with substernal goiter:
• The 14% who required a sternal split or transthoracic approach had a higher incidence of unplanned intubation (OR 2.70, 95% CI 1.17-6.25) and bleeding (OR 5.56, 95% CI 2.38-13.0); a higher incidence of death (1.9 versus 0.3%) was not statistically significant
• Together, these studies suggest that surgery for substernal goiter (using a cervical approach or sternotomy):
• Is associated with higher complication rates than surgery for cervical goiter
• Recurrent laryngeal nerve injury:
• Transient recurrent laryngeal nerve injury has been reported to occur in 2% to 9% of patients undergoing surgery for substernal goiter
• Permanent nerve injury occurs less commonly:
• 0 and 0.03% in the two largest studies [6,18] and 3% in two other reports
• Patients with bilateral nerve injury and therefore bilateral vocal cord paralysis require tracheostomy to provide an adequate airway
• Hypocalcemia:
• Hypocalcemia due to hypoparathyroidism:
• Is the most frequent complication of near-total thyroidectomy
• Is more common when the goiter is extensive and anatomic landmarks are displaced and obscured
• Hypocalcemia may be transient or permanent:
• Transient hypoparathyroidism occurred in 12 of 170 patients (7%) in one series
• Current requirements for short hospital stays argue for early treatment of hypocalcemia
• Tracheomalacia:
• In patients who have tracheomalacia due to pressure-induced destruction of tracheal rings by the goiter:
• The airway may collapse during the postoperative period
• In one study, 10% of patients could not be immediately extubated, although all were successfully extubated by 10 days:
• These patients were older, had larger goiters, and were more likely to have tracheal compression
• If recognized at the time of surgery:
• Tracheomalacia may in some cases be treated by partial tracheal resection and reconstruction:
• Otherwise, tracheostomy is necessary
• In a systematic review, the presence of a substernal goiter for more than five years causing tracheal compression:
• Was a risk factor for tracheomalacia and tracheostomy:
• However, in this review, tracheomalacia was an infrequent occurrence (3%):
• Was managed without tracheostomy in approximately 50% of cases
• Levothyroxine after surgery:
• Patients who undergo total thyroidectomy for benign disease:
• Should start a replacement dose of levothyroxine daily at an approximate dose of 1.6 mcg/kg body weight
• Patients over 65 years should be started at a 10% to 15% lower dose
• Serum TSH should be tested approximately six weeks after starting the replacement therapy
• The TSH should be kept in a normal range
• Treatment with higher doses of levothyroxine to suppress serum TSH:
• To prevent goiter recurrence in patients who have already had surgery for obstructive goiter (versus no thyroid hormone therapy or replacement therapy if needed to normalize the serum TSH concentration) is controversial:
• In one study with 10 years of follow-up, the recurrence rate was lower in the patients treated with levothyroxine (5% versus 42% in the untreated group had recurrent goiter):
• However, this benefit was not confirmed in a report with 30 years of follow-up:
• The recurrence rates were similar (41% and 45%) in treated and untreated patients
• Poor operative candidates:
• For patients with obstructive symptoms who are unable or unwilling to undergo surgery:
• Radioiodine therapy is an alternative option:
• Radioiodine is a reasonable option for patients who cannot or do not want to undergo surgery:
• Particularly if the substernal or obstructive goitrous tissue is functional on thyroid radionuclide imaging
• In patients with nonobstructive multinodular goiter:
• Radioiodine therapy results in goiter volume reductions:
• In the range of 30% to 60%
• Pretreatment with recombinant human thyroid-stimulating hormone (rhTSH, thyrotropin alfa):
• Allows treatment with lower doses of radioiodine for thyroid volume reduction:
• But may result in development of mild hyperthyroidism and transient goiter enlargement, or rarely induce Graves’ disease
• Methimazole may also be used to increase the radioiodine uptake and reduce the dose needed to reduce thyroid volume
• Results of radioiodine therapy for obstructive goiter include the following:
• In one series of 14 patients with large multinodular goiters, eight of whom had respiratory symptoms and eight of whom had substernal extension:
• All improved after treatment with 200 to 400 microcuries/g of radioiodine, and no patient had an acute exacerbation of obstructive symptoms
• Similar improvements in obstructive symptoms were seen in a series of 19 older patients treated with 100 microcuries/g tissue (average total dose 70 millicuries):
• Goiter volume decreased by 40%
• Volume reduction of the cervical and substernal components is similar (30%)
• Despite the reasonable results with radioiodine, surgery is still the treatment of choice because of concerns that radiation thyroiditis might result in worsening of airway obstruction and the need to rule out the diagnosis of carcinoma if the goiter is mostly substernal
• Recombinant human TSH:
• Similar to results seen with smaller multinodular goiters:
• The addition of rhTSH to radioiodine in patients with obstructive goiters:
• Results in a greater reduction in thyroid volume than radioiodine alone:
• However, rhTSH is not currently US Food and Drug Administration (FDA) approved for this indication, and it is not recommended, pending additional clinical trials demonstrating safety and efficacy
• Methimazole;
• Pretreatment of nontoxic nodular goiter with methimazole has also been used to increase the radioiodine uptake and reduce radioiodine dose or frequency of repeat radioiodine treatment:
• This is not currently an FDA-approved indication for this drug, but unlike rhTSH, it does not cause hyperthyroidism (it might cause transient hypothyroidism):
• The use methimazole to increase the radioiodine uptake in select patients with low radioiodine uptake who prefer radioiodine over surgery or who are poor surgical candidates is an option
SUMMARY AND RECOMMENDATIONS
• The most common symptom in patients with obstructive cervical or substernal goiter is:
• Exertional dyspnea:
• Which is present in 30% to 60% of cases:
• This symptom usually occurs when the tracheal diameter is under 8 mm
• Bilateral thyroid lobe enlargement:
• Especially if the goiter extends posterior to the trachea:
• May cause either:
• Compression or concentric narrowing of the trachea or
• Compression of the esophagus or jugular veins
• With some goiters, there is growth of one or both lobes through the thoracic inlet into the thoracic cavity:
• Which can result in obstruction of any of the structures in the inlet:
• Such goiters are referred to as substernal
• Patients with obstructive symptoms from a cervical or substernal goiter:
• Require removal or ablation of the thyroid
• For patients with obstructive goiter:
• The recommendation is surgery rather than radioiodine
• The reduction in thyroid volume with radioiodine is only moderate, and there are theoretical concerns that radioiodine could acutely worsen obstruction or a missed malignancy:
• However, radioiodine therapy is an option for patients with obstructive symptoms who are poor surgical candidates
• For asymptomatic patients with normal flow-volume loops whose goiters end at the level of the brachiocephalic vein or higher:
• Observation rather than surgery
• Monitoring of such patients with serial computed tomography (CT) scans:
• Initially after one year and, if stable, at increasing intervals (eg, two years later, then three years, then five years)
• Recommendations for asymptomatic patients with goiters that extend below the level of the brachiocephalic vein are controversial:
• Some authors suggests surgical excision for most patients:
• Except older patients and those who are poor operative candidates
• Other experts suggest observation of asymptomatic patients
• All patients requiring surgery for substernal goiter:
• Should be referred to experienced thyroid surgeons in high-volume centers to minimize complication rates
• For most patients undergoing surgery:
• A total or near-total thyroidectomy, rather than subtotal thyroidectomy, to minimize the risk of recurrent goiter
• Patients who undergo total thyroidectomy for benign disease:
• Should start a replacement dose of levothyroxine daily at an approximate dose of 1.6 mcg/kg body weight:
• Patients over 65 years should be started at a 10% to 15% lower dose
• Its is usually not recommend to prescribe suppressive doses of levothyroxine to prevent recurrence:
REFERENCES
1. Katlic MR, Grillo HC, Wang CA. Substernal goiter. Analysis of 80 patients from Massachusetts General Hospital. Am J Surg 1985; 149:283.
2. Allo MD, Thompson NW. Rationale for the operative management of substernal goiters. Surgery 1983; 94:969.
3. Miller MR, Pincock AC, Oates GD, et al. Upper airway obstruction due to goitre: detection, prevalence and results of surgical management. Q J Med 1990; 74:177.
4. Hedayati N, McHenry CR. The clinical presentation and operative management of nodular and diffuse substernal thyroid disease. Am Surg 2002; 68:245.
5. Newman E, Shaha AR. Substernal goiter. J Surg Oncol 1995; 60:207.
6. Erbil Y, Bozbora A, Barbaros U, et al. Surgical management of substernal goiters: clinical experience of 170 cases. Surg Today 2004; 34:732.
7. Gittoes NJ, Miller MR, Daykin J, et al. Upper airways obstruction in 153 consecutive patients presenting with thyroid enlargement. BMJ 1996; 312:484.
8. Pieracci FM, Fahey TJ 3rd. Substernal thyroidectomy is associated with increased morbidity and mortality as compared with conventional cervical thyroidectomy. J Am Coll Surg 2007; 205:1.
9. White ML, Doherty GM, Gauger PG. Evidence-based surgical management of substernal goiter. World J Surg 2008; 32:1285.
10. Berghout A, Wiersinga WM, Drexhage HA, et al. Comparison of placebo with L-thyroxine alone or with carbimazole for treatment of sporadic non-toxic goitre. Lancet 1990; 336:193.
11. Shimaoka K, Sokal JE. Suppressive therapy of nontoxic goiter. Am J Med 1974; 57:576.
12. Pieracci FM, Fahey TJ 3rd. Effect of hospital volume of thyroidectomies on outcomes following substernal thyroidectomy. World J Surg 2008; 32:740.
13. Ríos A, Rodríguez JM, Canteras M, et al. Surgical management of multinodular goiter with compression symptoms. Arch Surg 2005; 140:49.
14. Sancho JJ, Kraimps JL, Sanchez-Blanco JM, et al. Increased mortality and morbidity associated with thyroidectomy for intrathoracic goiters reaching the carina tracheae. Arch Surg 2006; 141:82.
15. Chen AY, Bernet VJ, Carty SE, et al. American Thyroid Association statement on optimal surgical management of goiter. Thyroid 2014; 24:181.
16. Testini M, Gurrado A, Avenia N, et al. Does mediastinal extension of the goiter increase morbidity of total thyroidectomy? A multicenter study of 19,662 patients. Ann Surg Oncol 2011; 18:2251.
17. de Perrot M, Fadel E, Mercier O, et al. Surgical management of mediastinal goiters: when is a sternotomy required? Thorac Cardiovasc Surg 2007; 55:39.
18. Zambudio AR, Rodríguez J, Riquelme J, et al. Prospective study of postoperative complications after total thyroidectomy for multinodular goiters by surgeons with experience in endocrine surgery. Ann Surg 2004; 240:18.
19. Moten AS, Thibault DP, Willis AW, Willis AI. Demographics, disparities, and outcomes in substernal goiters in the United States. Am J Surg 2016; 211:703.
20. Khan MN, Goljo E, Owen R, et al. Retrosternal Goiter: 30-Day Morbidity and Mortality in the Transcervical and Transthoracic Approaches. Otolaryngol Head Neck Surg 2016; 155:568.
21. Shen WT, Kebebew E, Duh QY, Clark OH. Predictors of airway complications after thyroidectomy for substernal goiter. Arch Surg 2004; 139:656.
22. Anderson PE, Hurley PR, Rosswick P. Conservative treatment and long term prophylactic thyroxine in the prevention of recurrence of multinodular goiter. Surg Gynecol Obstet 1990; 171:309.
23. Röjdmark J, Järhult J. High long term recurrence rate after subtotal thyroidectomy for nodular goitre. Eur J Surg 1995; 161:725.
24. Kay TW, d’Emden MC, Andrews JT, Martin FI. Treatment of non-toxic multinodular goiter with radioactive iodine. Am J Med 1988; 84:19.
25. Huysmans DA, Hermus AR, Corstens FH, et al. Large, compressive goiters treated with radioiodine. Ann Intern Med 1994; 121:757.
26. Bonnema SJ, Knudsen DU, Bertelsen H, et al. Does radioiodine therapy have an equal effect on substernal and cervical goiter volumes? Evaluation by magnetic resonance imaging. Thyroid 2002; 12:313.
27. Szumowski P, Abdelrazek S, Sykała M, et al. Enhancing the efficacy of 131I therapy in non-toxic multinodular goitre with appropriate use of methimazole: an analysis of randomized controlled study. Endocrine 2020; 67:136.
#Arrangoiz #ThryoidSurgeon #ThyroidExpert #ThyroidCancer #CancerSurgeon #Head