
Art Show Miami Florida


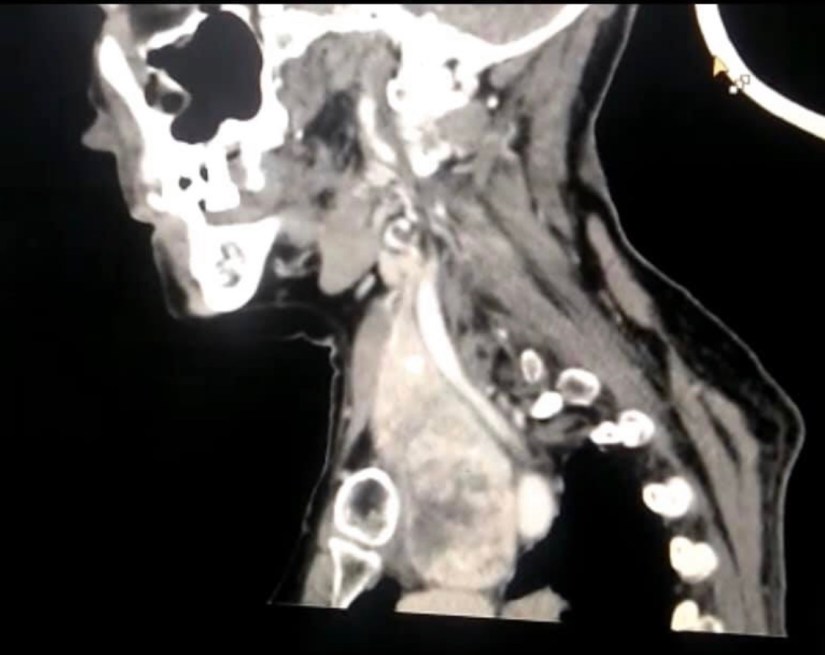
#Arrangoiz #ThyroidSurgeon #ThyroidExpert #Goiter #ThyroidSurgery #CancerSurgeon #HeadandNeckSurgeon #SurgicalOncologist #Teacher #Surgeon #Miami #Mexico #MountSinaiMedicalCenter
– Suspicion of malignancy
– Compression of the trachea
– Compression of the esophagus
– Quality of life
#Arrangoiz #ThyroidSurgeon #ThyroidExpert #HeadandNeckSurgeon #MultinodularGoiter #Goiter #Miami #MountSinaiMedicalCenter #Mexico
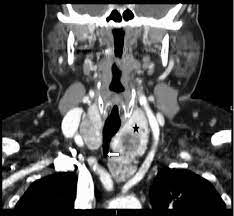
#Arrangoiz #ThyroidSurgeon #ThyroidExpert #MultinodularGoiter #Goiter #SubsternalGoiter #HeadandNeckSurgeon #MountSinaiMedicalCenter #Miami #Mexico
WHAT ARE THE SYMPTOMS OF A GOITER?
The term “goiter” simply refers to the abnormal enlargement of the thyroid gland. It is important to know that the presence of a goiter does not necessarily mean that the thyroid gland is malfunctioning. A goiter can occur in a gland that is producing too much hormone (hyperthyroidism), too little hormone (hypothyroidism), or the correct amount of hormone (euthyroidism). A goiter indicates there is a condition present which is causing the thyroid to grow abnormally
One of the most common causes of goiter formation worldwide is iodine deficiency. While this was a very frequent cause of goiter in the United States many years ago, it is no longer commonly observed. The primary activity of the thyroid gland is to concentrate iodine from the blood to make thyroid hormone. The gland cannot make enough thyroid hormone if it does not have enough iodine. Therefore, with iodine deficiency the individual will become hypothyroid. Consequently, the pituitary gland in the brain senses the thyroid hormone level is too low and sends a signal to the thyroid. This signal is called thyroid stimulating hormone (TSH). As the name implies, this hormone stimulates the thyroid to produce thyroid hormone and to grow in size. This abnormal growth in size produces what is termed a “goiter.” Thus, iodine deficiency is one cause of goiter development. Wherever iodine deficiency is common, goiter will be common. It remains a common cause of goiters in other parts of the world.
Hashimoto’s thyroiditis is a more common cause of goiter formation in the US. This is an autoimmune condition in which there is destruction of the thyroid gland by one’s own immune system. As the gland becomes more damaged, it is less able to make adequate supplies of thyroid hormone. The pituitary gland senses a low thyroid hormone level and secretes more TSH to stimulate the thyroid. This stimulation causes the thyroid to grow, which may produce a goiter.
Another common cause of goiter is Graves’ disease. In this case, one’s immune system produces a protein, called thyroid stimulating immunoglobulin (TSI). As with TSH, TSI stimulates the thyroid gland to enlarge producing a goiter. However, TSI also stimulates the thyroid to make too much thyroid hormone (causes hyperthyroidism). Since the pituitary senses too much thyroid hormone, it stops secreting TSH. In spite of this the thyroid gland continues to grow and make thyroid hormone. Therefore, Graves’ disease produces a goiter and hyperthyroidism.
Multinodular goiters are another common cause of goiters. Individuals with this disorder have one or more nodules within the gland which cause thyroid enlargement. This is often detected as a nodular feeling gland on physical exam. Patients can present with a single large nodule or with multiple smaller nodules in the gland when first detected. Thus, in early stages of a multinodular goiter with many small nodules, the overall size of the thyroid may not be enlarged yet. Unlike the other goiters discussed, the cause of this type of goiter is not well understood.
In addition to the common causes of goiter, there are many other less common causes. Some of these are due to genetic defects, others are related to injury or infections in the thyroid, and some are due to tumors (both cancerous and benign tumors).
As mentioned earlier, the diagnosis of a goiter is usually made at the time of a physical examination when an enlargement of the thyroid is found. However, the presence of a goiter indicates there is an abnormality of the thyroid gland. Therefore, it is important to determine the cause of the goiter. As a first step, you will likely have thyroid function tests to determine if your thyroid is underactive or overactive. Any subsequent tests performed will be dependent upon the results of the thyroid function tests. If the thyroid is diffusely enlarged and you are hyperthyroid, your doctor will likely proceed with tests to help diagnose Graves’ Disease.
If you are hypothyroid, you may have Hashimoto’s Thyroiditis and you may get additional blood tests to confirm this diagnosis. Other tests used to help diagnose the cause of the goiter may include a radioactive iodine scan, thyroid ultrasound, or a fine needle aspiration biopsy.
The treatment will depend upon the cause of the goiter. If the goiter was due to a deficiency of iodine in the diet (not common in the United States), you will be given iodine supplementation given in preparations to take by mouth. This will lead to a reduction in the size of the goiter, but often the goiter will not completely resolve.
If the goiter is due to Hashimoto’s Thyroiditis, and you are hypothyroid, you will be given thyroid hormone supplement as a daily pill. This treatment will restore your thyroid hormone levels to normal, but does not usually make the goiter go completely away. While the goiter may get smaller, sometimes there is too much scar tissue in the gland to allow it to get much smaller. However, thyroid hormone treatment will usually prevent it from getting any larger. Although appropriate in some individuals, surgery is usually not routine treatment of thyroiditis.
If the goiter is due to hyperthyroidism, the treatment will depend upon the cause of the hyperthyroidism. For some causes of hyperthyroidism, the treatment may lead to a disappearance of the goiter. For example, treatment of Graves’ disease with radioactive iodine usually leads to a decrease or disappearance of the goiter.
Many goiters, such as the multinodular goiter, are associated with normal levels of thyroid hormone in the blood. These goiters usually do not require any specific treatment after the appropriate diagnosis is made. If no specific treatment is suggested, you may be warned that you are at risk for becoming hypothyroid or hyperthyroid in the future. However, if there are problems associated with the size of the thyroid per se, such as the goiter getting so large that it constricts the airway, your doctor may suggest that the goiter be treated by surgical removal.
Whatever the cause, it is important to have regular (annual) monitoring when diagnosed with a goiter.

#Arrangoiz #ThyroidSurgeon #ThyroidExpert #HeadandNeckSurgeon #ThyroidCancer #Goiter #MountSinaiMedicalCenter #Miami #Mexico

#Arrangoiz #ParathyroidSurgeon #ParathyroidExpert #Hyperparathyroidism #PrimaryHyperparathyroidism #CancerSurgeon #EndocrineSurgery #Teacher #Surgeon #HeadandNeckSurgeon #SurgicalOncologist #ParathyroidAdenoma #Hypercalcemia #ElevatedCalciumLevels #Miami #MountSinaiMedicalCenter #MSMC #Mexico #Hialeah
#Arrangoiz #ParathyroidSurgeon #ParathyroidExpert #Hyperparathyroidism #PrimaryHyperparathyroidism #CancerSurgeon #EndocrineSurgery #Teacher #Surgeon #HeadandNeckSurgeon #SurgicalOncologist #ParathyroidAdenoma #Hypercalcemia #ElevatedCalciumLevels #Miami #MountSinaiMedicalCenter #MSMC #Mexico #Hialeah
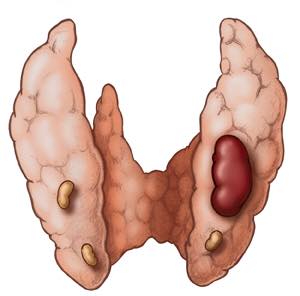
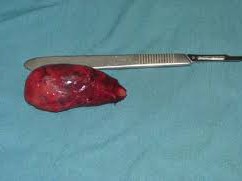
#Arrangoiz #ParathyroidSurgeon #ParathyroidExpert #Hyperparathyroidism #PrimaryHyperparathyroidism #CancerSurgeon #EndocrineSurgery #Teacher #Surgeon #HeadandNeckSurgeon #SurgicalOncologist #ParathyroidAdenoma #Hypercalcemia #ElevatedCalciumLevels #Miami #MountSinaiMedicalCenter #MSMC #Mexico #Hialeah