👉Clinical validation of ThyroSeq v3 performed in the recently completed prospective double-blind multicenter international study (ClinicalTrials.gov identifier NCT02352766).
👉Patients were enrolled in the study at 10 medical centers, 9 located in the U.S. and 1 in Singapore.
👉Totally, 782 patients with one or more thyroid nodules sampled by FNA (1013 samples) were enrolled.
👉Of those, 257 FNA samples from 234 patients had Bethesda III-V cytology, underwent surgery, and had FNA samples sufficient for molecular analysis.
👉These 257 FNA samples comprised a final validation set that determined ThyroSeq v3 performance.
👉The study had no post-unblinding sample exclusion.
👉Performance of ThyroSeq v3 in the prospective double-blind multicenter study was recently published in JAMA Oncology. (Steward, DL et al. JAMA Oncol. 2018.)
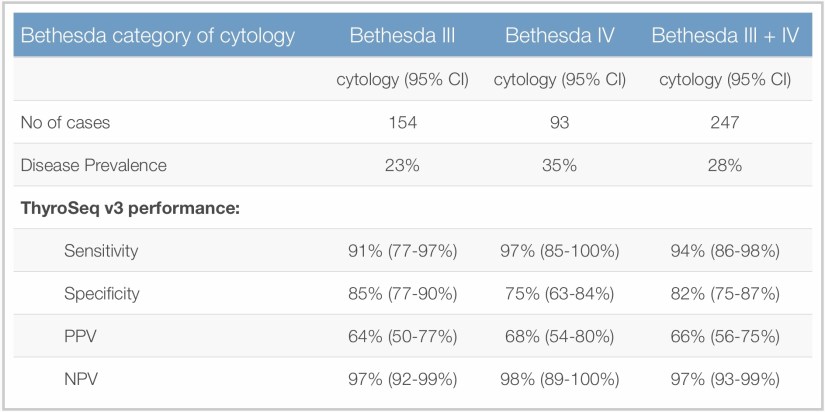
👉In this multicenter study, clinical performance of ThyroSeq v3 was validated in all main types of thyroid cancer, including Hurthle cell (oncocytic) cancer.
👉In fact, the study included 10 Hurthle cell carcinomas, 34 Hurthle cell adenomas, and 5 hyperplastic nodules with Hurthle cell predominance.
👉The performance of ThyroSeq v3 allowed to detect all Hurthle cell carcinomas (sensitivity, 100%; 95%CI:69.2-100%), with all 5 hyperplastic nodules with Hurthle cell predominance classified as Negative, and overall test specificity of 66.7% (95%CI: 49.8-80.9%).
👉In the study, the majority of nodules with false-positive test results were clonal neoplasms and not hyperplastic nodules.
👉There were 5 missed cancers (3%) that were all intrathyroidal and low stage.
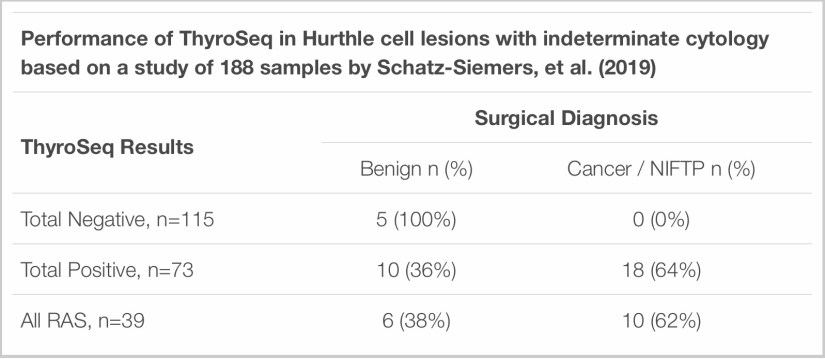
👉Additionally, ThyroSeq performance in Hurthle cell lesions was assessed in an independent study from NYUthat included 188 indeterminate Hurthle cell predominant thyroid FNAs that had ThyroSeq v2/v3 results.
👉Surgical follow up was available for 33 cases:
- The study found that the majority of cases (61%) were negative by ThyroSeq, meaning ThyroSeq had a 61% negative call rate in Hurthle cell lesions in this study.
- Five of the ThyroSeq-negative cases went for surgery and all were benign on histology, which means ThyroSeq had 100% NPV for Hurthle cell lesions that went to surgery in this study.
- 28 samples were ThyroSeq-positive and went for surgery with 18 diagnosed as cancer or NIFTP on final histology, meaning ThyroSeq had a 64% PPV for cancer/NIFTP in Hurthle cell nodules in this study.
-
Rodrigo Arrangoiz MS, MD, FACS a head and neck surgeon / endocrine surgeon / surgical oncologist and is a member of Sociedad Quirúrgica S.C at the America British Cowdray Medical Center in Mexico City:

- Rodrigo Arrangoiz MS, MD, FACS:
- Is a member of the American Head and Neck Society

-
- He is a member of the American Thyroid Association:





