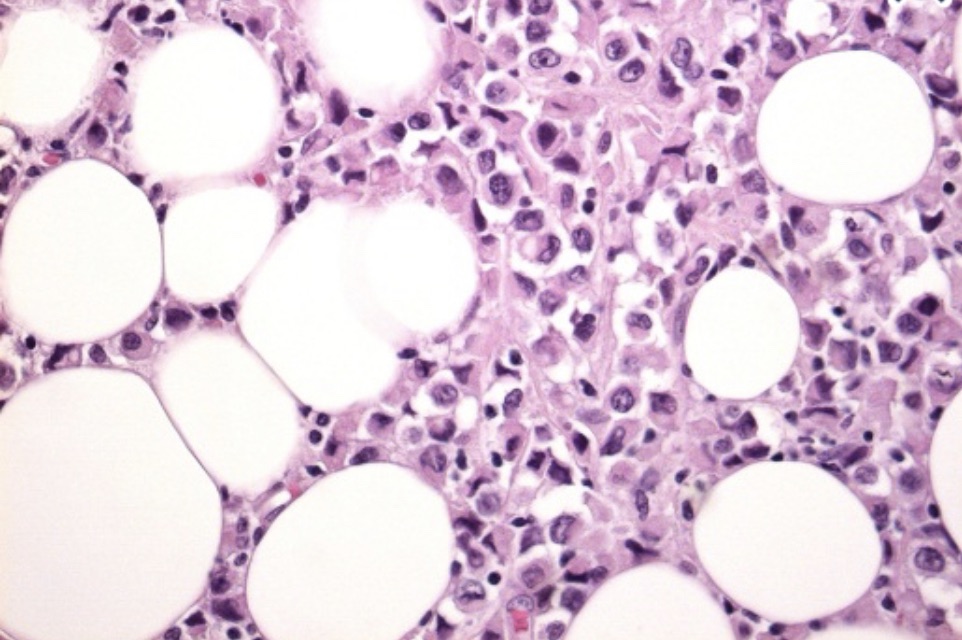
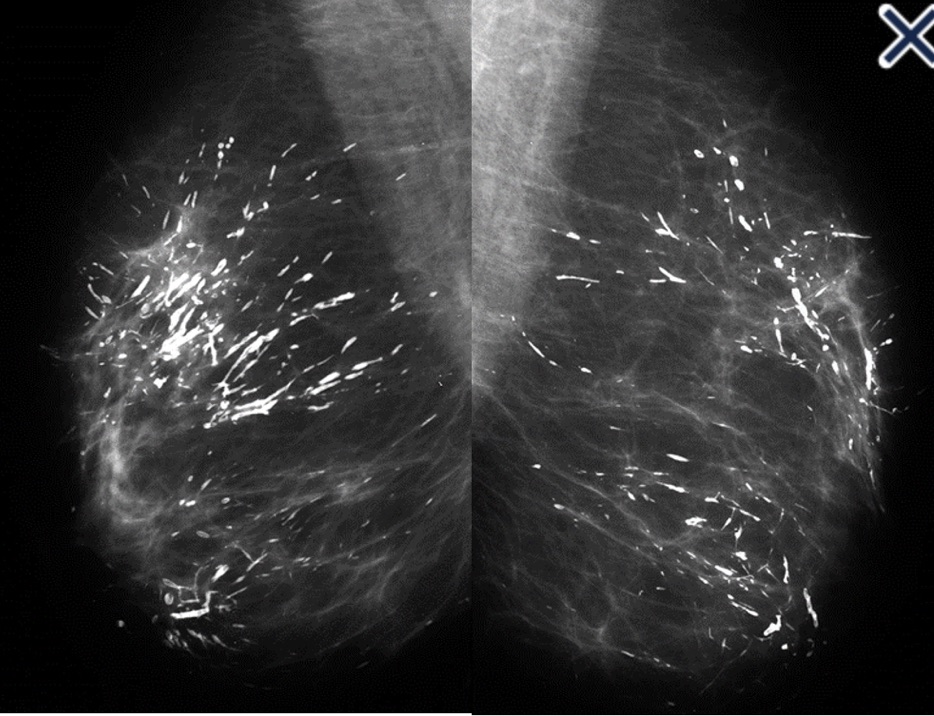
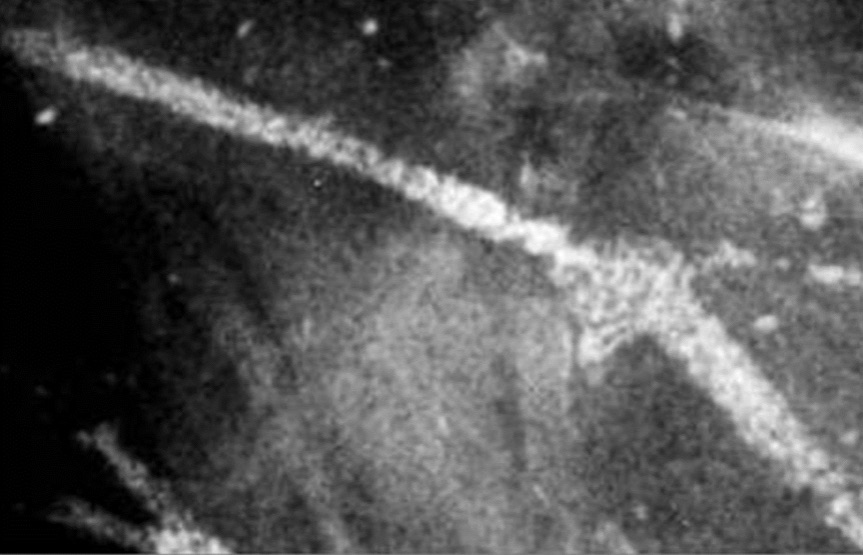
- Galactoceles:
- Are milk retention cysts:
- That result from a blocked milk duct
- Are milk retention cysts:
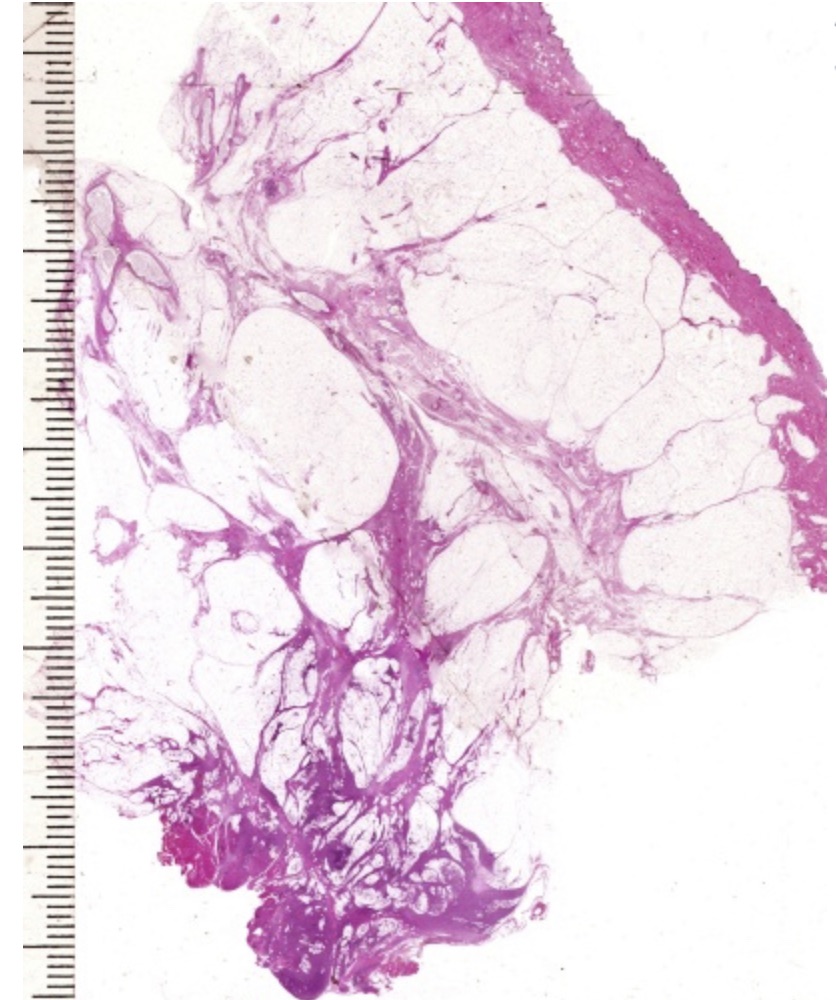
- They present as cystic, sometimes very large masses:
- During pregnancy, lactation, and after weaning
- They are often painless unless they become infected
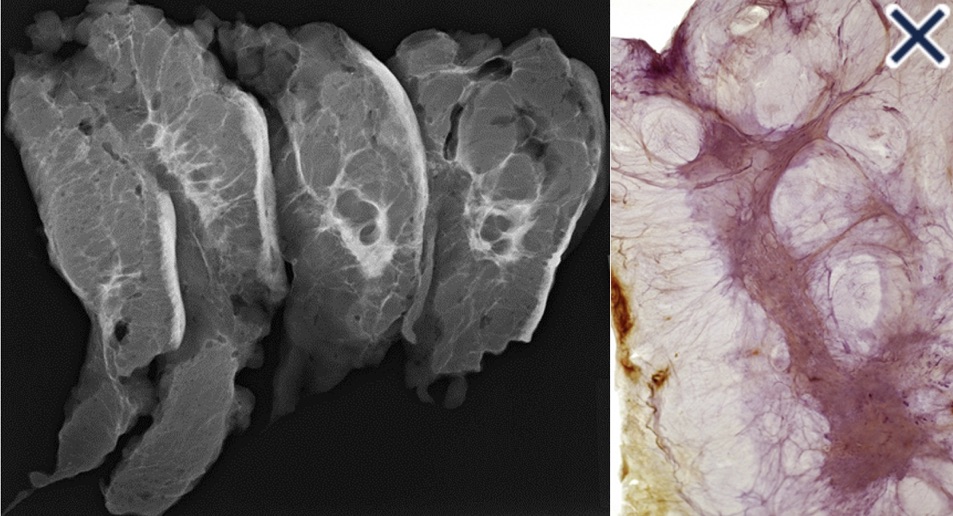
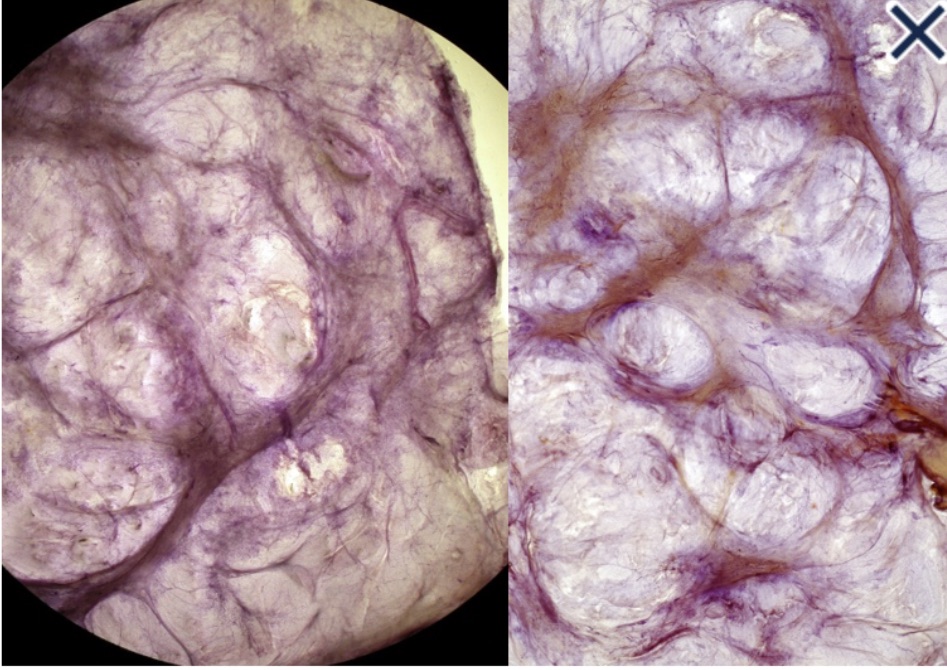
- Initially, they contain milky fluid:
- But over time, contents become thicker and more creamy or oily as the fluid is reabsorbed
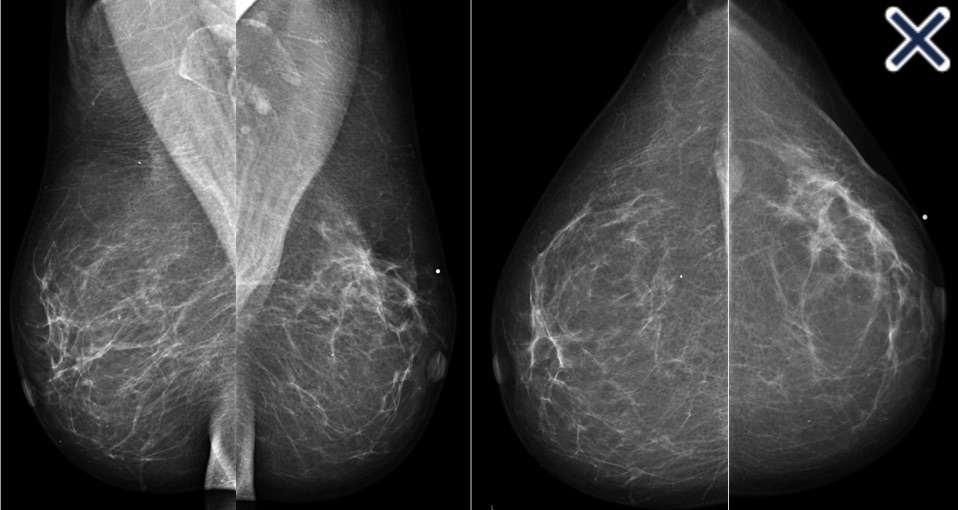
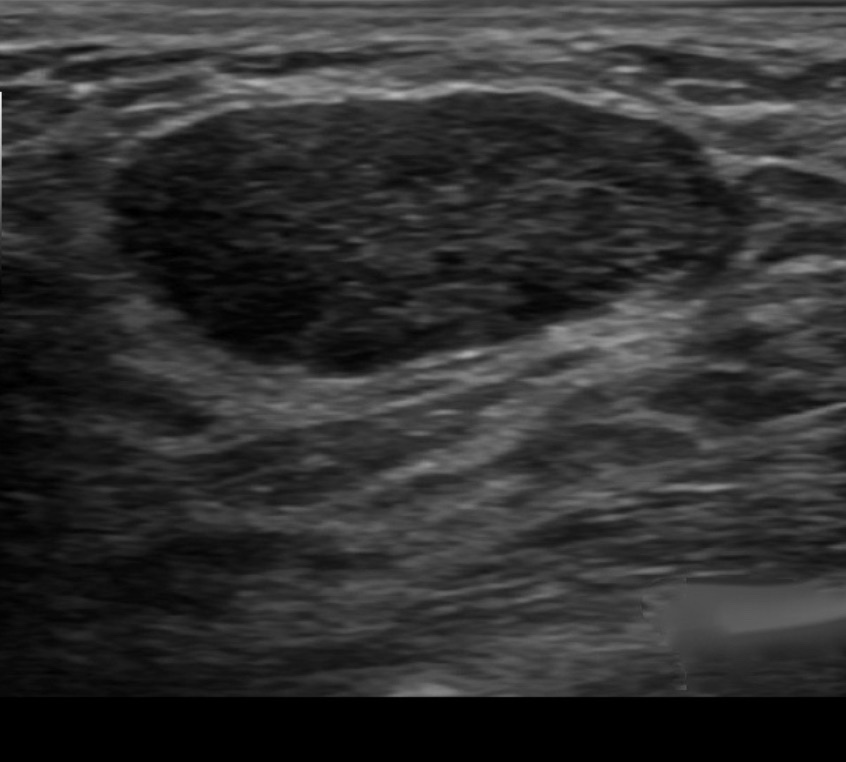
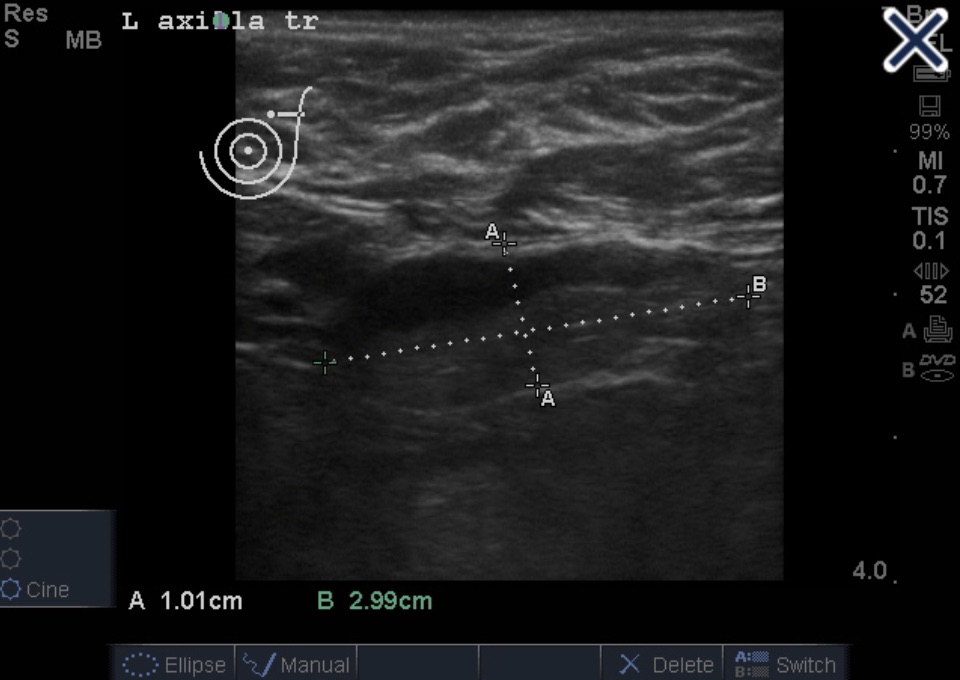
- Ultrasound is the primary diagnostic method
- Typical findings include a:
- Well-defined lesion with thin echogenic walls
- The internal appearance consists of either homogeneous contents or heterogeneous contents with fluid clefts and anechoic rims
- Typical findings include a:
- Management consists of:
- Needle aspiration demonstrating milky contents:
- Which both confirms the diagnosis and excludes malignancy
- Needle aspiration demonstrating milky contents:
- Surgical resection is reserved for:
- Cases refractory to conservative management
- References
- Sawhney S. Petkovska L, Ramadan S, Al-Muhtaseb S, Jain R, Sheikh M.Sonographic appearance of galactoceles. J Clin Ultrasound. 2002;30(1):18-22.
- Sabate JM, Clotet M, Torrubia S, Gomez A, Guerrero R, de las Heras P. Radiologic evaluation of breast disorders related to pregnancy and lactation. Radiographics.2007;27(Suppl 1):S101-S124.