
Triple Negative Breast Cancer and PD-L1 Testing



| Cancer Type | Frequency of MAH |
|---|---|
| Lung cancer (especially squamous cell) | ~25–30% |
| Breast cancer | ~20–25% |
| Multiple myeloma | ~15–20% |
| Renal cell carcinoma | ~5–10% |
| Head and neck squamous cell carcinoma | ~5–10% |
| Others (ovarian, lymphoma, bladder) | <5% |
| Test | Result |
|---|---|
| Calcium | ↑ |
| PTH | Suppressed |
| PTHrP | Elevated |
| Phosphate | Low |
| 1,25-vitamin D | Low/normal |
| Test | Result |
|---|---|
| Calcium | ↑ |
| PTH | Suppressed |
| PTHrP | Normal |
| Vitamin D | Normal |
| Test | Result |
|---|---|
| Calcium | ↑ |
| PTH | Suppressed |
| 1,25-OH vitamin D | Elevated |
| Feature | Primary Hyperparathyroidism | Malignancy Hypercalcemia |
|---|---|---|
| PTH | High or inappropriately normal | Suppressed |
| Calcium level | Mild–moderate (10.5–12 mg/dL) | Often >13–14 mg/dL |
| Symptom onset | Chronic | Acute / severe |
| PTHrP | Normal | Elevated (HHM) |
| Vitamin D | Normal | May be elevated in lymphoma |




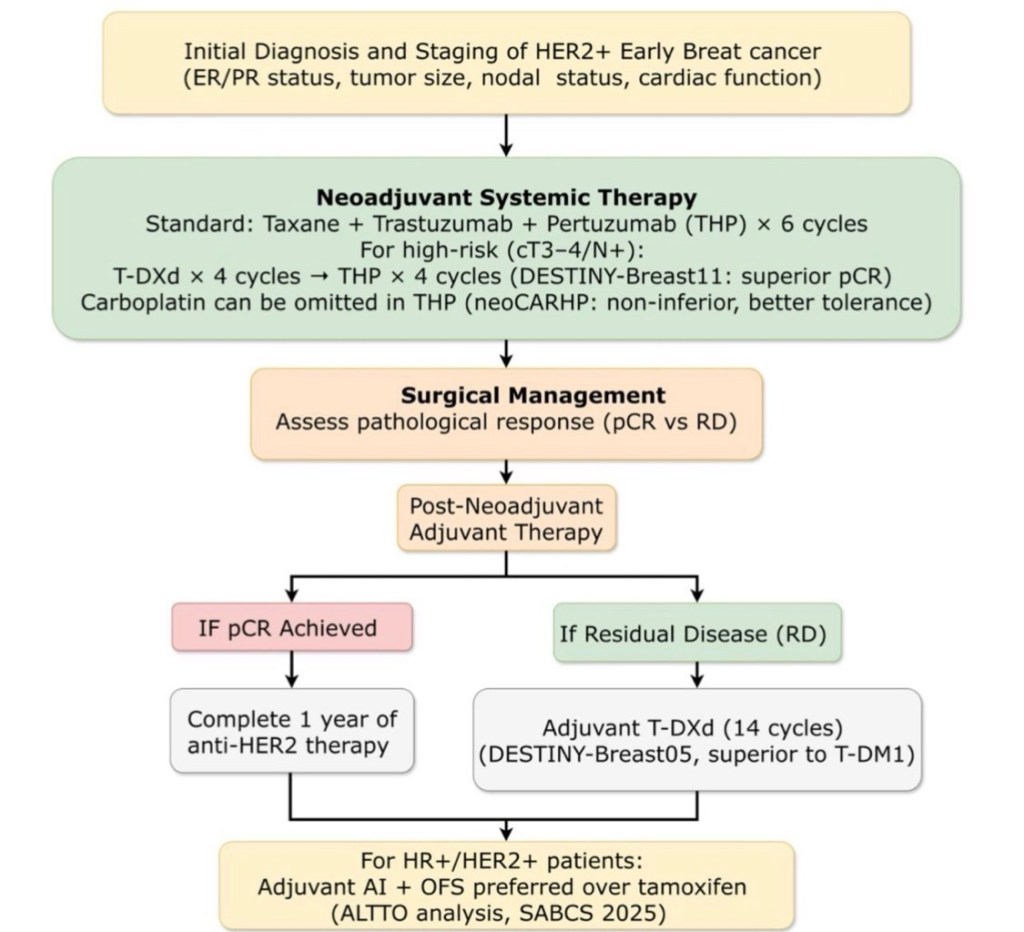
The treatment landscape for HER2-positive early breast cancer (EBC) is evolving rapidly — and trastuzumab deruxtecan (T-DXd) is emerging as a potential new standard in both the neoadjuvant and adjuvant settings.
🔹 Neoadjuvant Setting
DESTINY-Breast11
T-DXd followed by THP (docetaxel + trastuzumab + pertuzumab) demonstrated:
Significantly higher pathologic complete response (pCR) rates compared with standard anthracycline-based regimens A chemotherapy-sparing strategy with reduced anthracycline exposure Favorable tolerability profile consistent with prior T-DXd data
📊 Early reports show pCR rates approaching ~65–70%, exceeding historical benchmarks for standard neoadjuvant regimens (typically ~55–60%).
Clinical Implication:
We may be entering an era of antibody–drug conjugate (ADC)-based neoadjuvant intensification, potentially redefining the backbone of HER2-directed therapy.
Reference:
Hurvitz SA et al. DESTINY-Breast11. Presented at ESMO 2024 / SABCS 2024 (late-breaking data).
🔹 Adjuvant Setting
DESTINY-Breast05
For patients with residual invasive disease after neoadjuvant therapy, T-DXd demonstrated:
53% reduction in risk of invasive disease–free survival (iDFS) events compared with T-DM1 Superior invasive disease–free survival Manageable toxicity, with ILD rates consistent with prior experience
This builds upon the paradigm established by KATHERINE, where T-DM1 replaced trastuzumab in patients with residual disease.
Now, T-DXd appears poised to replace T-DM1 in this high-risk population.
Reference:
DESTINY-Breast05. Presented at ASCO 2025.
von Minckwitz G et al. KATHERINE trial. NEJM. 2019;380:617–628.
🔬 Why This Matters
We are witnessing:
A shift from monoclonal antibodies → ADC-based escalation Earlier deployment of highly potent HER2-directed agents Refinement of risk-adapted therapy based on response
If adopted into guidelines (NCCN, ASCO, ESMO), this could:
Redefine the management of residual disease Potentially reduce recurrence risk further in high-risk HER2+ EBC Change neoadjuvant sequencing strategies
⚠️ Considerations
ILD/pneumonitis risk requires vigilance Cost-effectiveness and long-term survival data pending Optimal sequencing with pertuzumab still being clarified
📌 Bottom Line
T-DXd is no longer just a metastatic drug.
It is rapidly reshaping the curative-intent HER2+ early breast cancer algorithm.
Choledochal Cysts – Types and Management
Choledochal cysts are congenital cystic dilatations of the biliary tree. They are associated with an abnormal pancreaticobiliary junction and carry a significant lifetime risk of malignancy (especially cholangiocarcinoma).
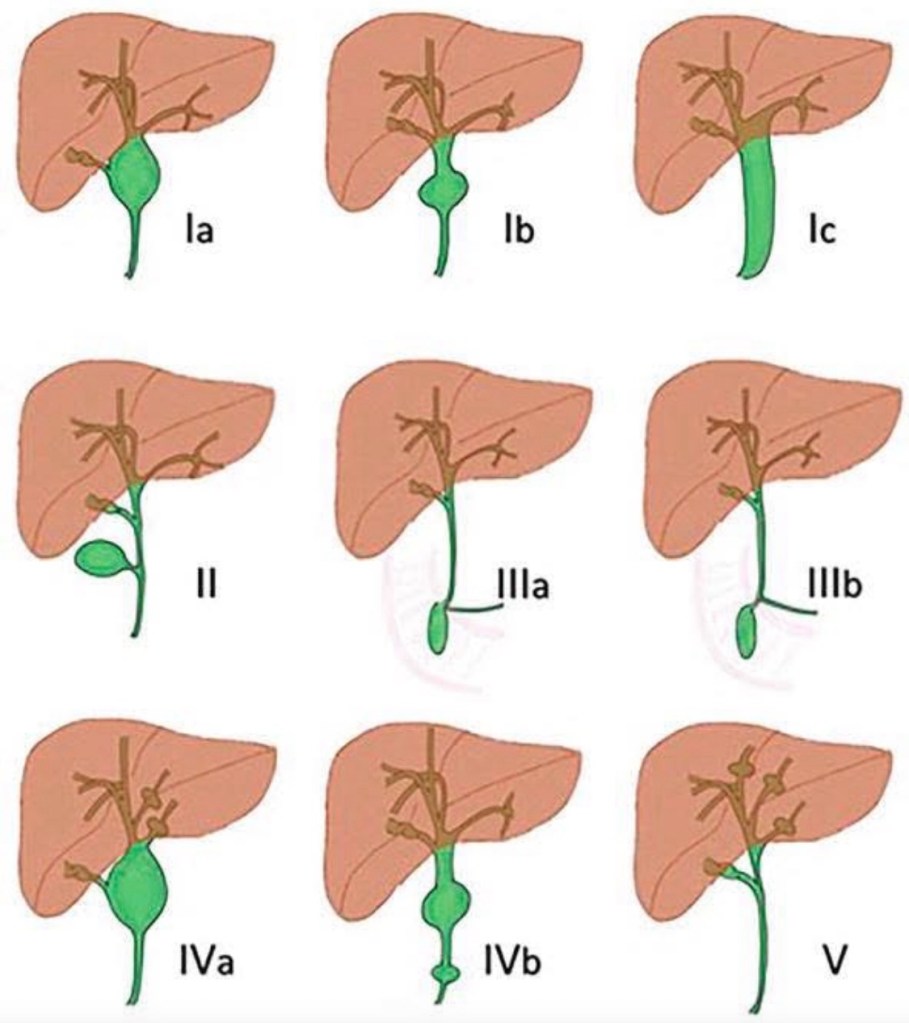
Classification (Todani Classification)
The most widely used system is the Todani classification, which divides choledochal cysts into five main types:
Type I – Extrahepatic bile duct dilatation (most common, 50–80%)
• Ia – Diffuse cystic dilatation of CBD
• Ib – Focal segmental dilatation
• Ic – Fusiform dilatation of CBD
Management:
→ Complete excision of extrahepatic bile duct + Roux-en-Y hepaticojejunostomy
Type II – True diverticulum of CBD
• Saccular outpouching from extrahepatic bile duct
Management:
→ Diverticulectomy ± primary closure of CBD
Type III – Choledochocele
• Intraduodenal dilatation of distal CBD (within ampulla)
Management:
→ Endoscopic sphincterotomy (often sufficient)
→ Surgical excision if large/symptomatic
Type IV – Multiple cysts
• IVa – Both intrahepatic and extrahepatic involvement
• IVb – Multiple extrahepatic cysts only
Management:
→ Excision of extrahepatic bile duct + Roux-en-Y hepaticojejunostomy
→ Liver resection if localized intrahepatic disease
→ Liver transplant if diffuse severe intrahepatic disease
Type V – Caroli Disease
• Multiple intrahepatic cystic dilatations only
Associated with congenital hepatic fibrosis.
Management:
→ Segmental liver resection (localized)
→ Liver transplantation (diffuse disease)
Clinical Presentation
• Children: classic triad (rarely complete)
• Abdominal pain
• Jaundice
• Palpable mass
• Adults:
• Recurrent cholangitis
• Pancreatitis
• Biliary colic
• Incidental finding
Investigations
• Ultrasound – initial test
• MRCP – investigation of choice
• CT if malignancy suspected
• LFTs
ERCP mainly therapeutic (type III).
Complications
• Cholangitis
• Pancreatitis
• Stones
• Strictures
• Rupture (rare)
• Cholangiocarcinoma (10–30% lifetime risk if untreated)
Principles of Management (Important for Practice)
Surgical Standard Operation
Cyst excision + Roux-en-Y hepaticojejunostomy
→ Gold standard for Type I and IV
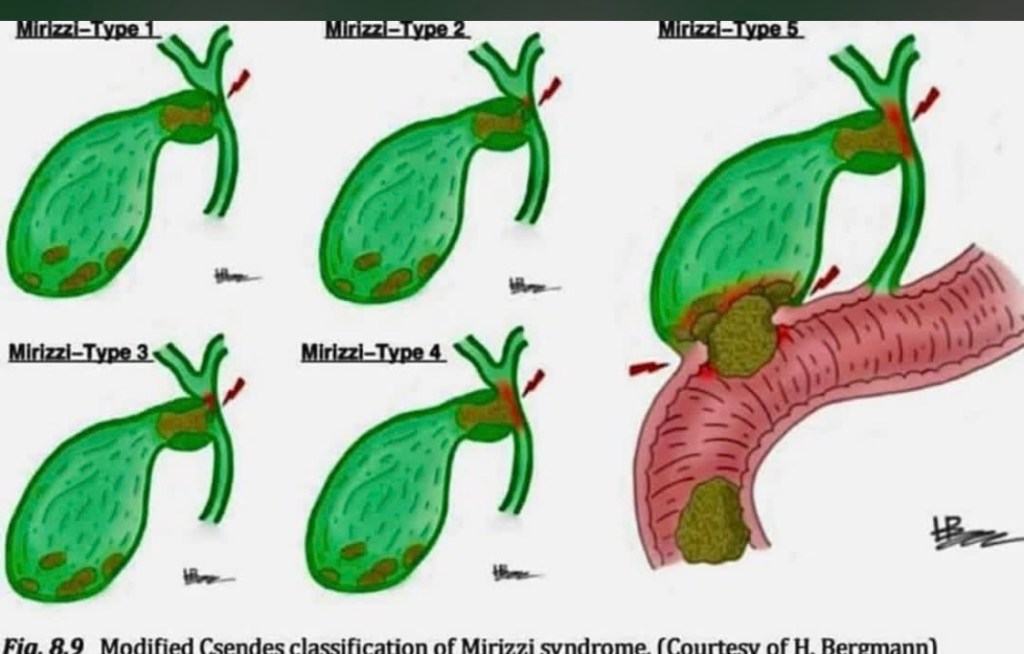
Mirizzi Syndrome: The rare but challenging complication where an impacted gallstone in the cystic duct or Hartmann’s pouch causes external compression or fistulization into the common bile duct. The modified Csendes classification grades severity from Type 1 (external compression only) through Type 5 (cholecystobiliary fistula with gallstone ileus). Type 1 shows simple compression without fistula formation. Type 2 involves erosion affecting less than one-third of the bile duct circumference. Type 3 extends to involve one-third to two-thirds of the duct. Type 4 shows complete destruction of the bile duct wall. Type 5 adds the complication of cholecystoenteric fistula with gallstone ileus. Recognition is critical during cholecystectomy as misidentification can lead to bile duct injury. Higher types require bile duct reconstruction
New 5-Year Evidence Supporting Radiofrequency Ablation (RFA) in Early-Stage Breast Cancer
I’m pleased to share results from the RAFAELO Phase 3 multicenter trial — published online in Annals of Surgical Oncology (Feb 18, 2026) — assessing radiofrequency ablation (RFA) as a minimally invasive alternative to partial mastectomy in early-stage breast cancer.
🔍 Study Overview
• Design: Multicenter, single-arm, Phase 3 clinical study.
• Population: 370 women with solitary Tis–T1 (≤1.5 cm), N0M0 breast carcinomas.
• Intervention: Percutaneous RFA followed by whole-breast radiation (45–60 Gy).
• Primary Endpoint: 5-year ipsilateral breast tumor recurrence-free survival (IBTRFS).
📈 Key Findings
✔ At 5 years, IBTRFS was 98.6% (90% CI 97.1–99.3%), exceeding the pre-specified noninferiority margin of 90%.
✔ Only 2 ipsilateral recurrences were observed at 5 years.
✔ Grade ≥3 skin ulceration was rare (1/370 patients), underscoring a favorable safety profile.
✔ These results suggest that RFA with adjuvant radiation may be comparable to partial mastectomy in appropriately selected early-stage patients.
🏷 Clinical Significance
This large prospective trial provides the most robust long-term evidence to date that RFA — a less invasive approach — may be a viable local-control strategy in small, node-negative breast cancers. These findings reinforce ongoing interest in expanding treatment options that balance oncologic safety with patient-centred care (e.g., cosmesis, procedural morbidity).
Optional Add-Ons for Engagement
🔹 Thanks to the RAFAELO Study Group and contributing centers for advancing patient-centred oncology.
🔹 Looking forward to longer follow-up, quality-of-life data, and comparative trials against standard surgery