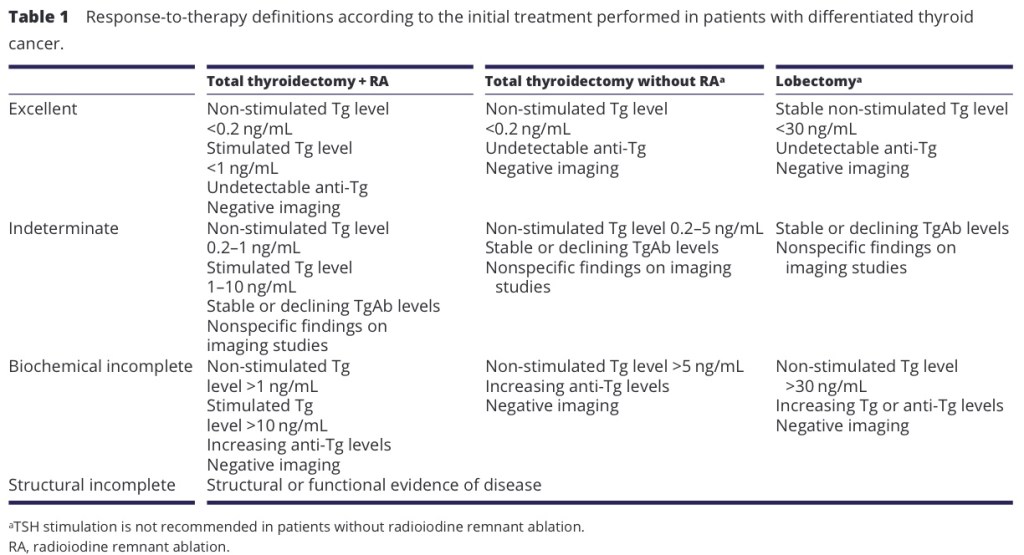
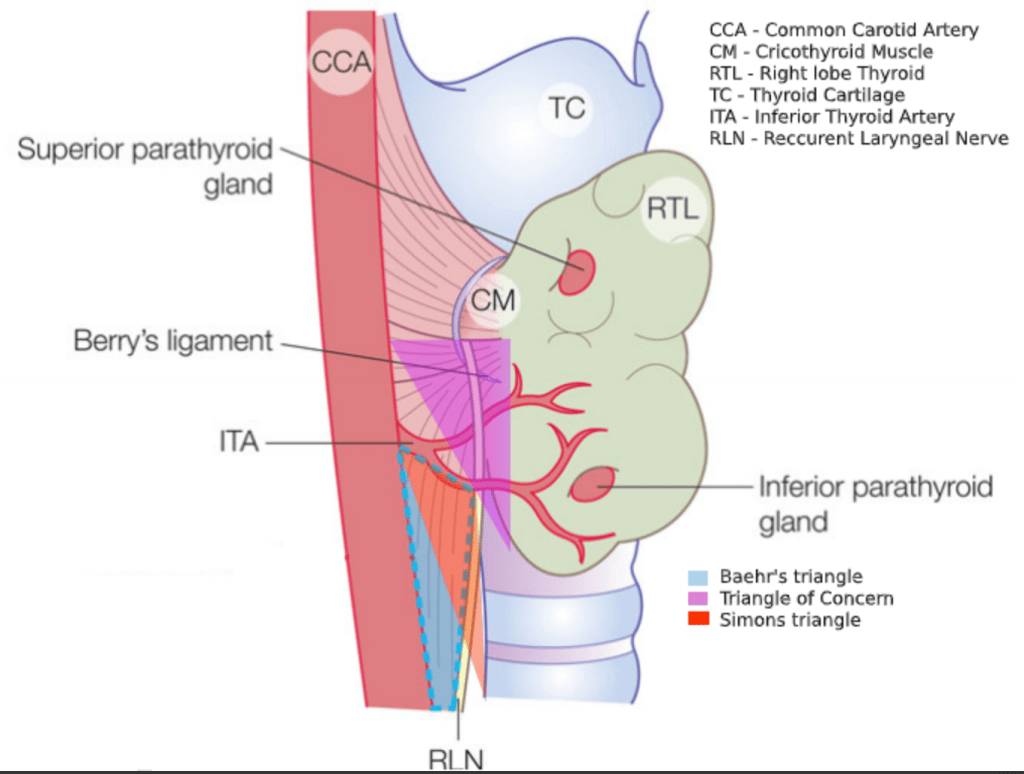
- Papillary thyroid cancer (PTC):
- Has a high predilection for spread to locoregional lymph nodes (LNs):
- Occurring in up to 40% to 90% of cases:
- When prophylactic nodal dissection is performed:
- Though such high rates of metastatic disease may prove enticing to recommend routine prophylactic node dissection:
- Recurrence-free survival is not effected by the removal of sonographically normal, microscopically diseased nodes
- Instead, prophylactic central neck dissection may be individually considered for those patients with:
- T3 or T4 tumors, or in the presence of lateral neck metastases
- Clinically suspicious or biopsy-proven nodal disease warrants a “therapeutic” dissection of the involved compartments
- “Berry picking,” or selective removal of suspicious LN metastases, is not recommended:
- As it is associated with significantly higher recurrence rates and does not lower the rate of postoperative complications compared with systematic compartmental dissections
- “Berry picking,” or selective removal of suspicious LN metastases, is not recommended:
- Occurring in up to 40% to 90% of cases:
- Has a high predilection for spread to locoregional lymph nodes (LNs):
- The risk of surgical complications with nodal dissection should be weighed against the benefit of LN removal:
- Central neck dissections may result in temporary or permanent injury to the RLN and hypoparathyroidism
- Surgeon case volume predicts patient outcomes:
- Those performing less than 10 cases compared with those performing more than 100 cases per year had complications in 24% and 14.5% of cases, respectively
- Although dissection of the lateral neck is less often associated with adverse events:
- Injury to the spinal accessory nerve may occur with dissection of level II or V
- Similarly, chyle leaks may be seen after removal of nodes in level IV:
- Particularly on the left side