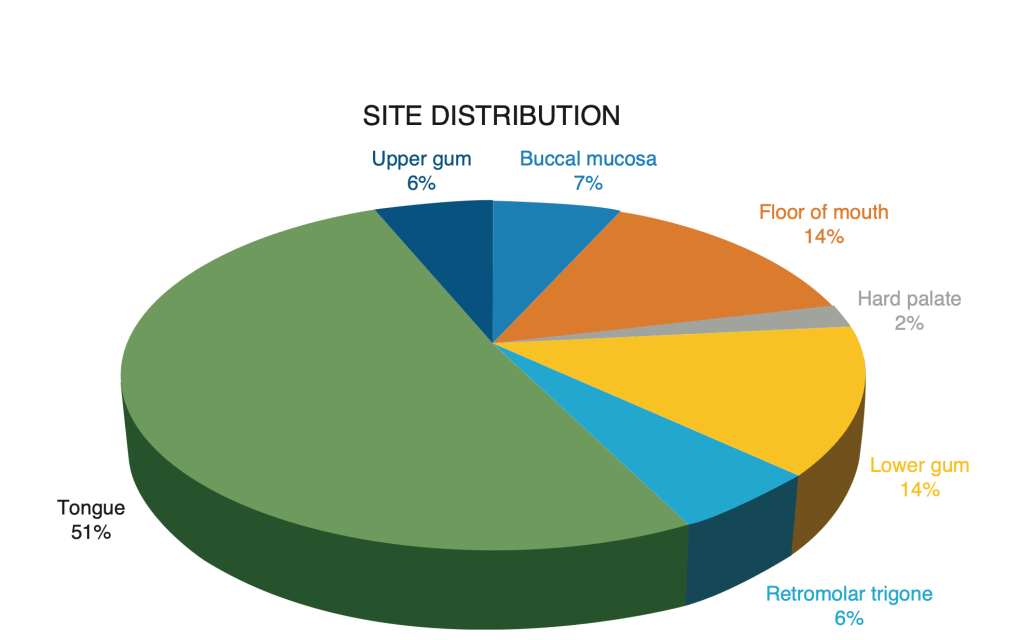
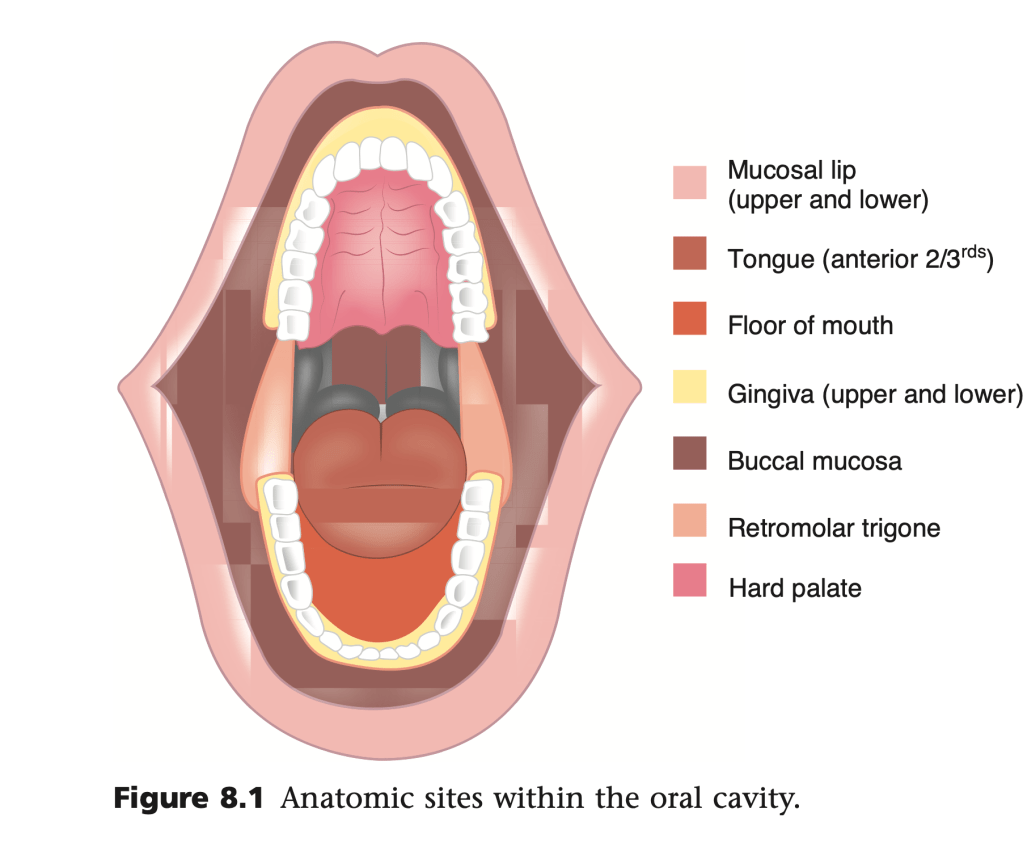
- The clinical features of primary tumors arising from the mucosal surface of the oral cavity are:
- Variable
- The tumor may be:
- Ulcerative
- Exophytic
- Endophytic
- The gross characteristics of the lesion:
- Are usually sufficient to raise the index of suspicion regarding the need for a biopsy to establish tissue diagnosis
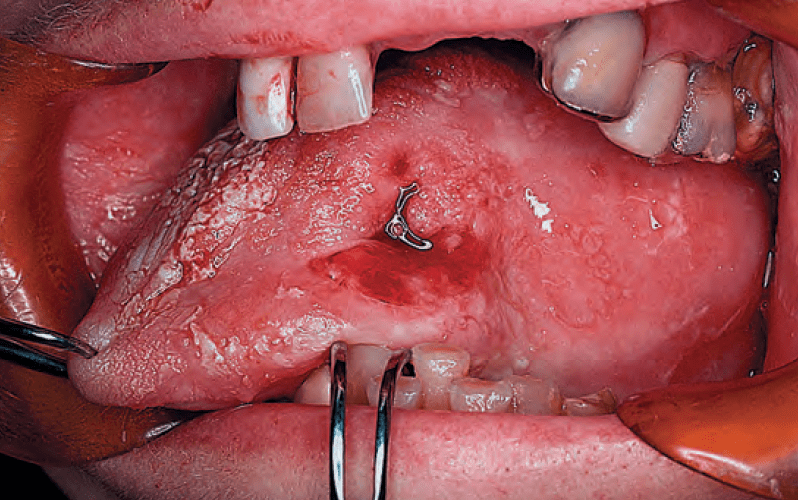
- Ulcerative lesions:
- Usually are accompanied by an irregular edge and induration of the underlying soft tissues

- Exophytic lesions may present either as a:
- Cauliflower-like irregular growth or as flat, pink to pinkish white proliferative lesions
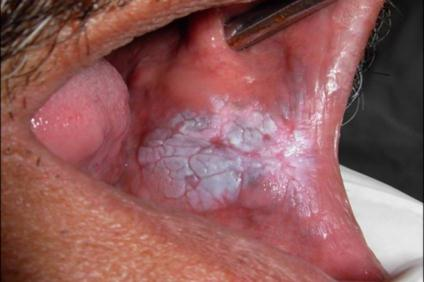
- Occasionally, a red to pink velvety flat lesion is the only manifestation of superficially invasive or in situ carcinoma

buccal mucosa

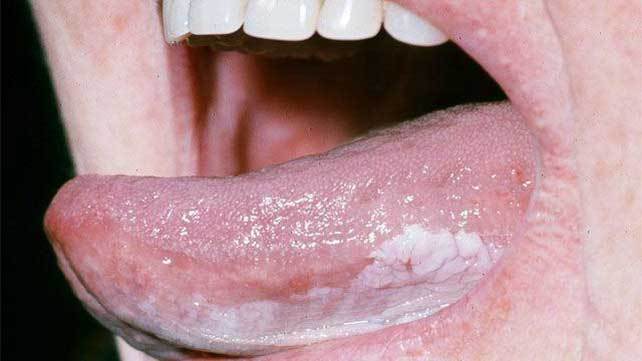
- Squamous cell carcinomas (SCC) with excessive keratin production and verrucous carcinomas:
- Present as white heaped-up keratotic lesions with varying degrees of keratin debris on the surface

hyperkeratosis


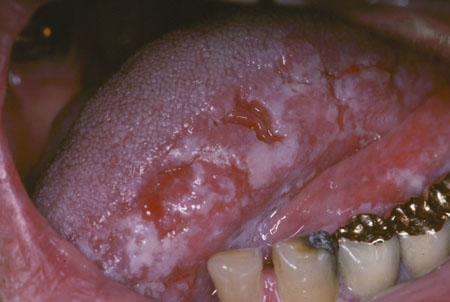
- Papillary projections:
- Often are seen in lesions that are accompanied or preceded by a squamous papilloma


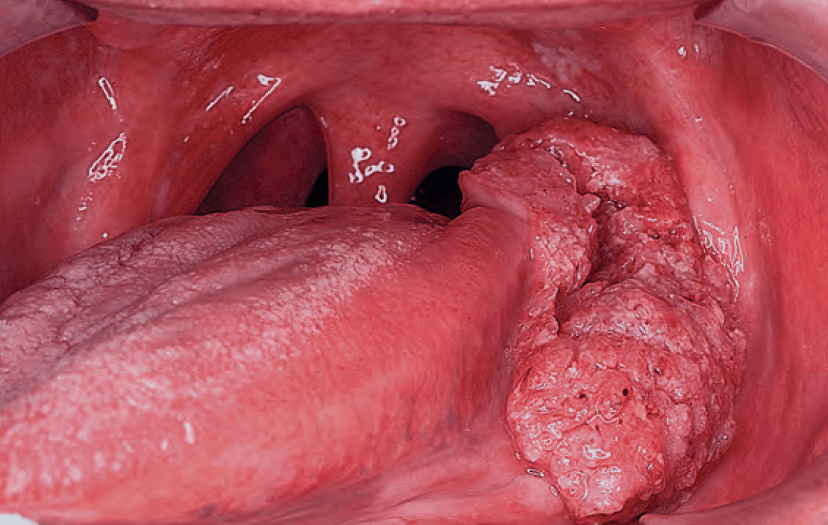
- Endophytic lesions:
- Have a very small surface component:
- But a substantial amount of soft-tissue involvement beneath the surface
- Have a very small surface component: