- The EORTC 18071 trial:
- Was a landmark, double-blind, phase 3 study that established the efficacy of adjuvant immune checkpoint blockade in cutaneous melanoma
- In this trial, patients with completely resected high-risk stage III melanoma were randomized to receive:
- High-dose ipilimumab (10 mg/kg IV every 3 weeks for 4 doses, then every 3 months for up to 3 years) or placebo
- The primary endpoint was:
- Recurrence-free survival (RFS)
- EORTC 18071 demonstrated that:
- Adjuvant ipilimumab significantly improved compared to placebo:
- RFS:
- 5-year RFS:
- 40.8% vs 30.3%
- 5-year RFS:
- Overall survival:
- 5-year OS:
- 65.4% vs 54.4%
- 5-year OS:
- However, the benefit came at the cost of substantial toxicity:
- With grade ≥ 3 immune-related adverse events in 41.6% of patients and a 1% treatment-related mortality rate
- RFS:
- Adjuvant ipilimumab significantly improved compared to placebo:
- This trial was the first to show a survival benefit for adjuvant immunotherapy in melanoma:
- Leading to regulatory approval of ipilimumab in this setting
- The findings of EORTC 18071 provided the foundation for subsequent trials of immune checkpoint blockade:
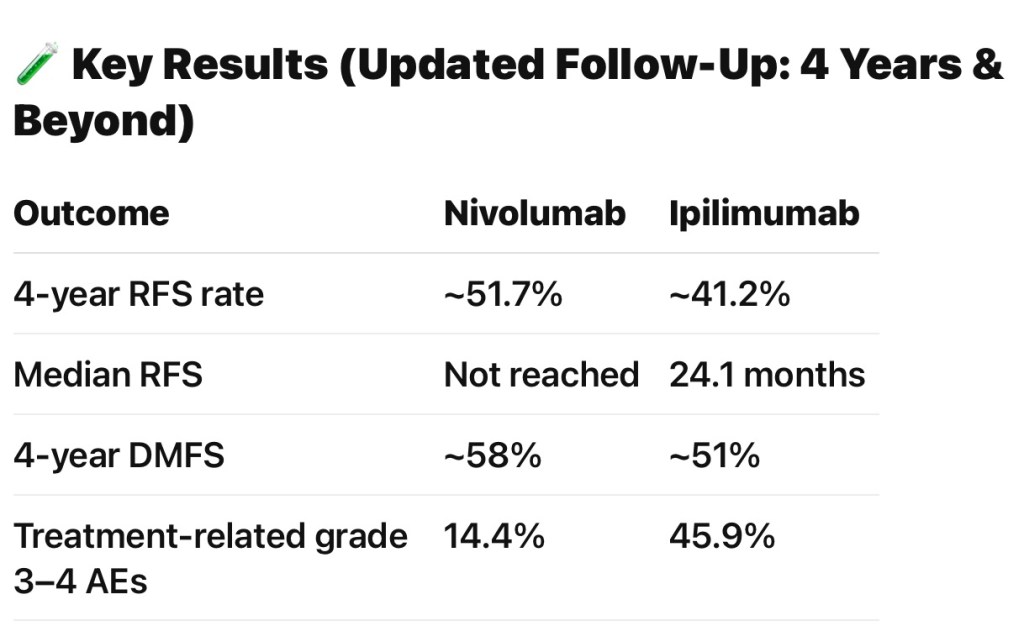
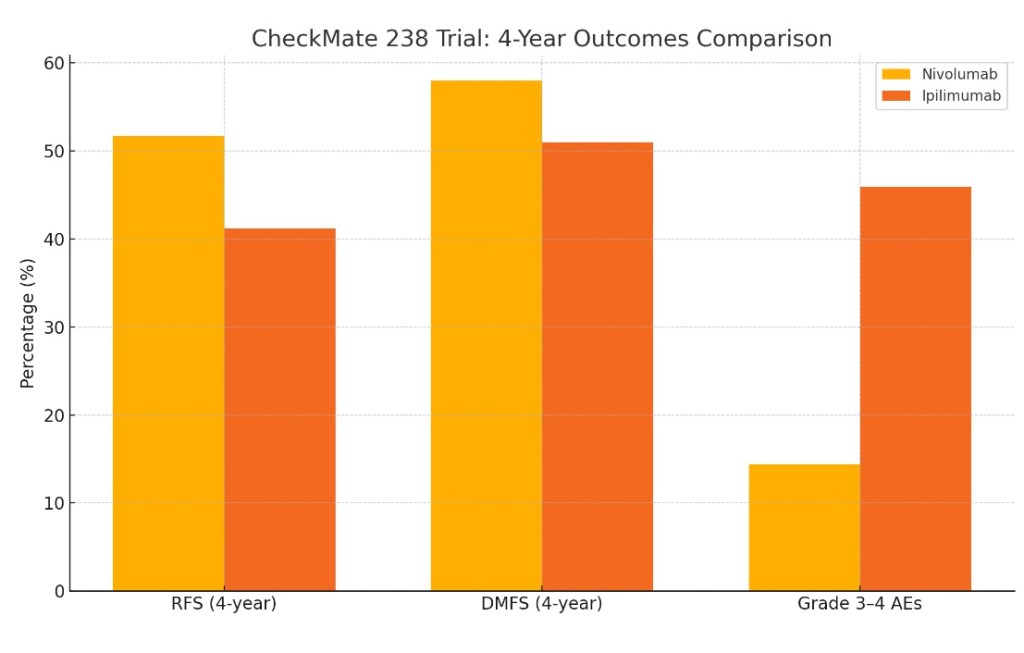
- CheckMate 238:
- Compared nivolumab (anti–PD-1) to ipilimumab:
- It showed superior RFS and a markedly improved safety profile for nivolumab:
- 5-year RFS: 50% vs 39%
- Grade 3 to 4 adverse events: 14% vs 41%
- It showed superior RFS and a markedly improved safety profile for nivolumab:
- Compared nivolumab (anti–PD-1) to ipilimumab:
- KEYNOTE-054 (EORTC 1325):
- Compared pembrolizumab (anti–PD-1) to placebo:
- It also demonstrated a significant RFS benefit:
- 3-year RFS: 63.7% vs 44.1% with lower toxicity than ipilimumab
- It also demonstrated a significant RFS benefit:
- Compared pembrolizumab (anti–PD-1) to placebo:
- CheckMate 238:
- In the context of adjuvant and recurrent therapy:
- EORTC 18071 established the principle that:
- Immune checkpoint blockade can reduce recurrence and improve survival after resection of high-risk melanoma:
- But anti–PD-1 agents (nivolumab, pembrolizumab) have since become preferred due to superior efficacy and tolerability
- Immune checkpoint blockade can reduce recurrence and improve survival after resection of high-risk melanoma:
- EORTC 18071 established the principle that:
- For patients with local recurrence after adjuvant therapy:
- Management is now guided by prior exposure and resistance patterns:
- With escalation to combination immunotherapy or targeted therapy as appropriate
- Management is now guided by prior exposure and resistance patterns:
- References:
- Adjuvant Ipilimumab Versus Placebo After Complete Resection of High-Risk Stage III Melanoma (EORTC 18071): A Randomised, Double-Blind, Phase 3 Trial. Eggermont AM, Chiarion-Sileni V, Grob JJ, et al. The Lancet. Oncology. 2015;16(5):522-30. doi:10.1016/S1470-2045(15)70122-1.
- Recent Advances in the Treatment of Melanoma. Curti BD, Faries MB. The New England Journal of Medicine. 2021;384(23):2229-2240. doi:10.1056/NEJMra2034861.
- Systemic Therapy for Melanoma: ASCO Guideline Update. Seth R, Agarwala SS, Messersmith H, et al. Journal of Clinical Oncology : Official Journal of the American Society of Clinical Oncology. 2023;41(30):4794-4820. doi:10.1200/JCO.23.01136.
- Adjuvant Therapy for Melanoma. Agha A, Tarhini AA. Current Oncology Reports. 2017;19(5):36. doi:10.1007/s11912-017-0594-5.
Adjuvant Nivolumab versus Ipilimumab in Resected Stage III or IV Melanoma. Weber J, Mandala M, Del Vecchio M, et al. The New England Journal of Medicine. 2017;377(19):1824-1835. doi:10.1056/NEJMoa1709030. - Adjuvant Nivolumab Versus Ipilimumab in Resected Stage IIIB-C and Stage IV Melanoma (CheckMate 238): 4-Year Results From a Multicentre, Double-Blind, Randomised, Controlled, Phase 3 Trial. Ascierto PA, Del Vecchio M, Mandalá M, et al. The Lancet. Oncology. 2020;21(11):1465-1477. doi:10.1016/S1470-2045(20)30494-0.
- Adjuvant Nivolumab Versus Ipilimumab in Resected Stage III/IV Melanoma: 5-Year Efficacy and Biomarker Results From CheckMate 238. Larkin J, Del Vecchio M, Mandalá M, et al. Clinical Cancer Research : An Official Journal of the American Association for Cancer Research. 2023;29(17):3352-3361. doi:10.1158/1078-0432.CCR-22-3145.
- Adjuvant Pembrolizumab versus Placebo in Resected Stage III Melanoma. Eggermont AMM, Blank CU, Mandala M, et al. The New England Journal of Medicine. 2018;378(19):1789-1801. doi:10.1056/NEJMoa1802357.
- Adjuvant Pembrolizumab Versus Placebo in Resected Stage III Melanoma (EORTC 1325-Mg/Keynote-054): Distant Metastasis-Free Survival Results From a Double-Blind, Randomised, Controlled, Phase 3 Trial. Eggermont AMM, Blank CU, Mandalà M, et al. The Lancet. Oncology. 2021;22(5):643-654. doi:10.1016/S1470-2045(21)00065-6.
- Longer Follow-Up Confirms Recurrence-Free Survival Benefit of Adjuvant Pembrolizumab in High-Risk Stage III Melanoma: Updated Results From the EORTC 1325-Mg/Keynote-054 Trial. Eggermont AMM, Blank CU, Mandala M, et al. Journal of Clinical Oncology : Official Journal of the American Society of Clinical Oncology. 2020;38(33):3925-3936. doi:10.1200/JCO.20.02110.