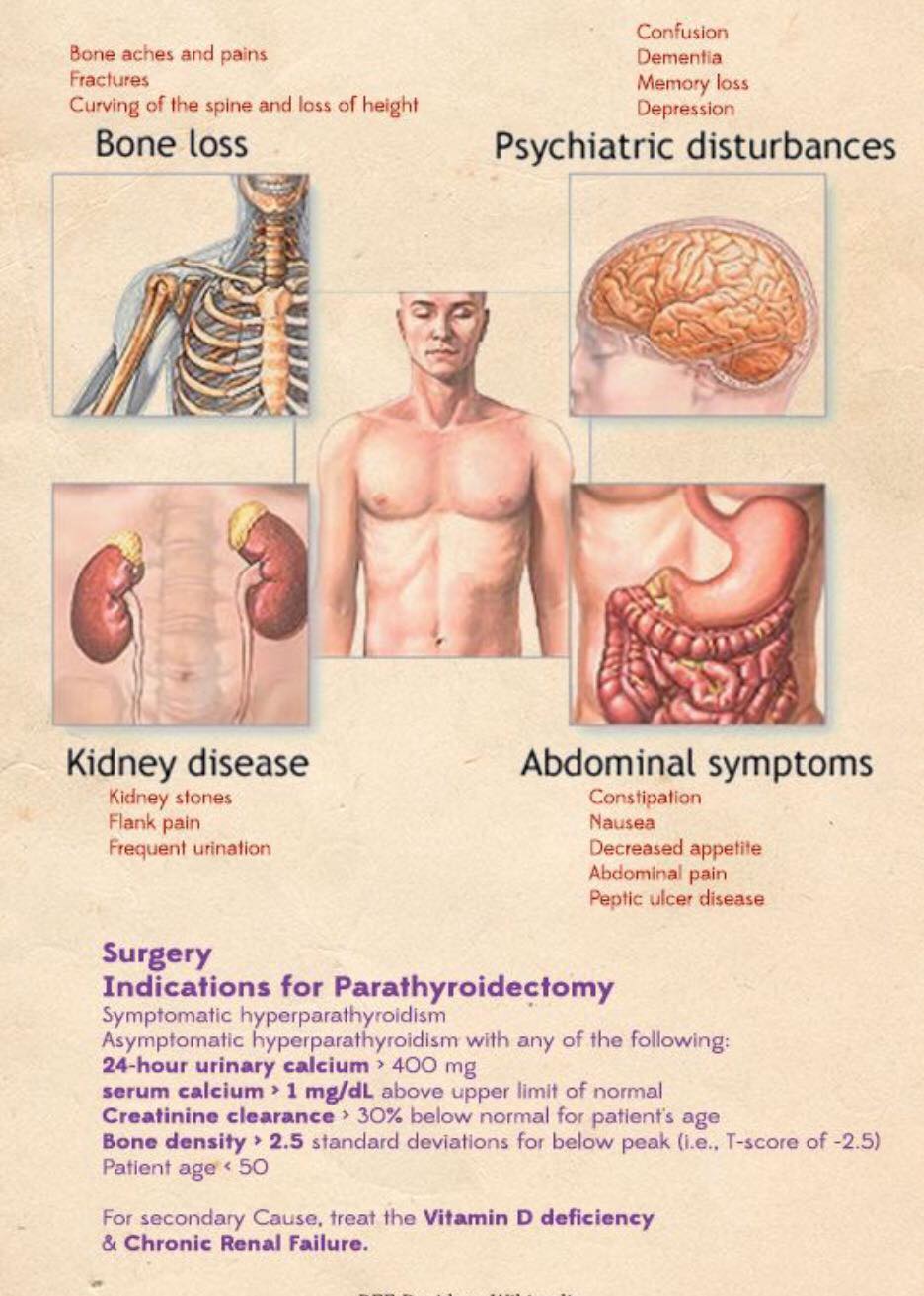
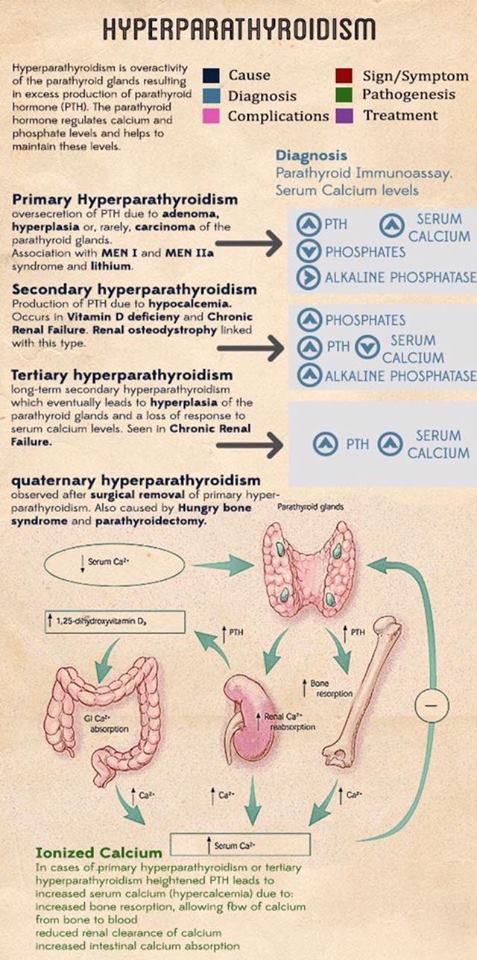
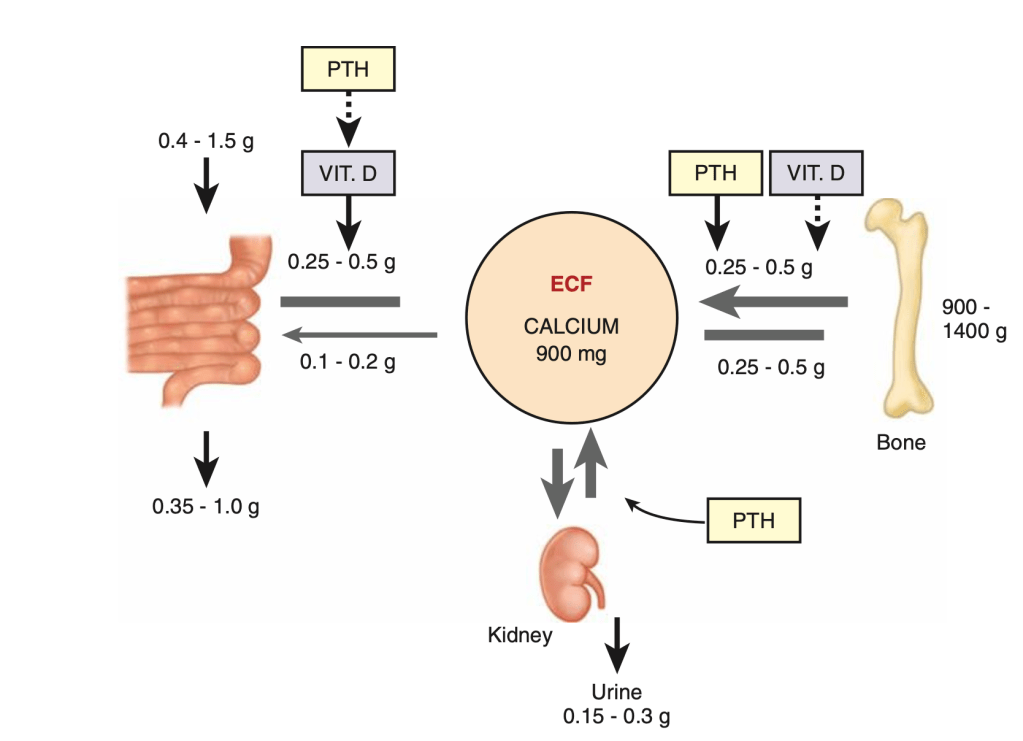
- Exposure of the Prevertebral Fascia:
- The first step in identifying the superior gland:
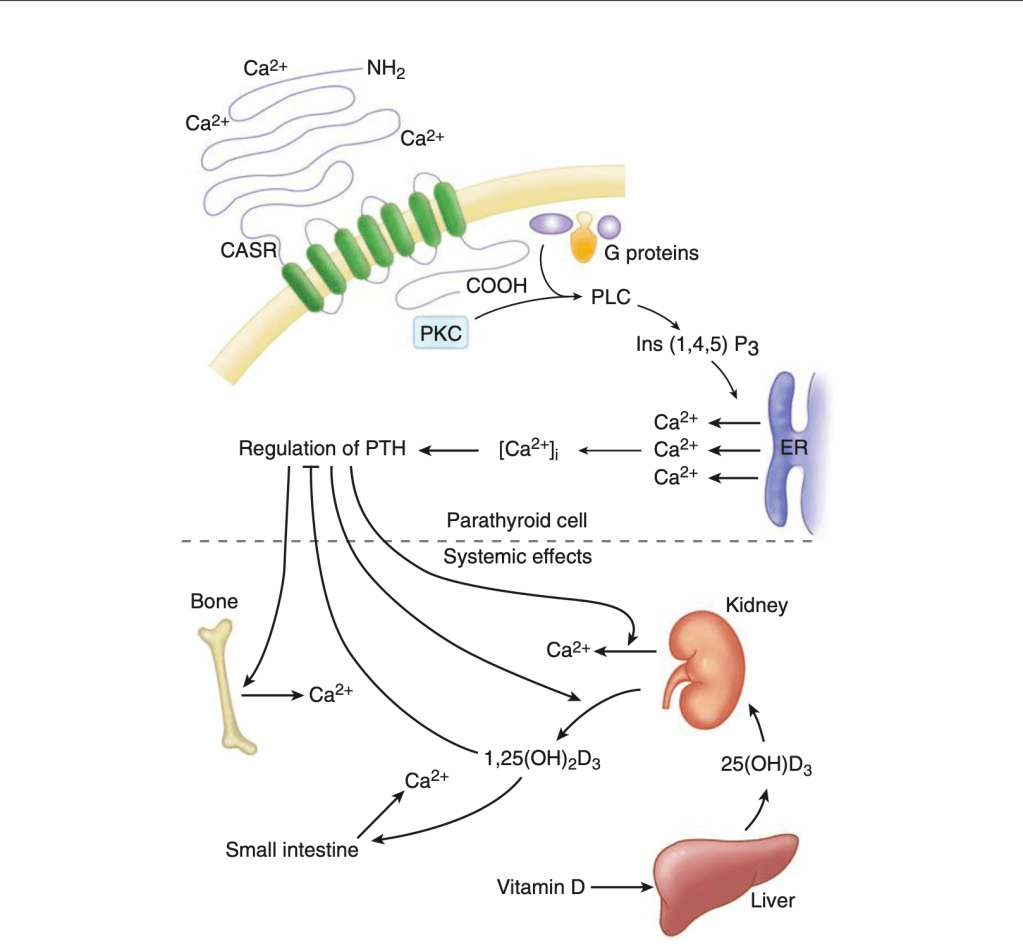
- Is to locate the inferior thyroid artery (ITA)
- The ITA is a crucial reference point for the location of the superior parathyroid gland:
- The superior parathyroid gland is usually found in an area 1 cm cranial to the ITA
- Once the ITA has been identified, blunt dissection cranial to the artery and directly posterior is performed down to the shiny prevertebral fascia
- The prevertebral fascia defines the posterior extent of the dissection, and the most posterior location possible for a superior parathyroid gland
- The first step in identifying the superior gland:
- Visual:
- Although it is tempting to dive straight in and attempt to dissect the first piece of tissue that resembles parathyroid tissue, considerable time and effort can be saved by slowly and deliberately confirming the key landmarks and looking for some of the morphological features described in section “Pearls for Identification of Parathyroid Glands” (published in a previous Blog)
- The identification of a parathyroid gland begins withcareful visual inspection:
- Start by looking for a gland or fat pad in a 1 to 2-cm area cranial to the ITA on the posterior surface of the thyroid lobe (Figure 1)
- Digital
- The superior glands can be found in a number of positions in association with structures from the fourth branchial arch, including:
- Retropharyngeal
- Retroesophageal
- Para-esophageal
- Adjacent the hyoid bone
- Further, when a superior gland enlarges:
- It tends to do so in a posterior and caudal direction and can pass behind the ITA to lie below the inferior gland
- After careful visual inspection in the area 1 cm cranial tothe ITA, these potential positions are digitally palpatedfor using five maneuvers (Fig. 1.4):
- The index finger is introduced into the space previously created above the ITA and directly down to the prevertebral fascia / retroesophageal space and then swept along the esophagus to feel in the retroesophageal / retropharyngeal positions
- The finger is then swung caudally until the finger lies vertically with the tip below the ITA
- The tissue over the tip of the finger is gently balloted, feeling for an enlarged superior gland to contact the tip of the posterior index finger
- The finger is then swung back to a horizontal position
- The finger is withdrawn slowly while the tip remain in contact with the esophagus and trachea, deliberately feeling for the trachea-esophageal groove
- The superior glands can be found in a number of positions in association with structures from the fourth branchial arch, including:



#Arrangoiz #ParathyroidSurgeon #ParathyroidExpert #Hyperparathyroidism #Hypercalcemia #HeadandNeckSurgeon #MSMC #MountSinaiMedicalCenter #Miami #Mexico #Surgeon #Teacher



 • Cirugia oncológica / tumores de cabeza y cuello / cirugia endocrina:
• Cirugia oncológica / tumores de cabeza y cuello / cirugia endocrina: • Maestria en ciencias (Clinical research for healthprofessionals):
• Maestria en ciencias (Clinical research for healthprofessionals): • Cirugia de tumores de cabeza y cuello / cirugiaendocrina
• Cirugia de tumores de cabeza y cuello / cirugiaendocrina