- Clin Thyroidol 2021;33:121–123.
- Background
- Low-risk papillary thyroid microcarcinomas (PTMCs) have been shown to have an:
- Indolent course
- This observation has led to alternative management approaches such as:
- Active surveillance (AS) rather than standard treatment with surgery
- Studies examining AS have shown promising results, with a low incidence of disease progression such as:
- Tumor enlargement (5.3%)
- Lymph node involvement (1.6%)
- No evidence of distant metastasis
- However, delayed surgeries during AS do occur:
- Likely driven by patient and physician:
- Anxiety
- Likely driven by patient and physician:
- Therefore ultrasound-guided thermal ablation, particularly radiofrequency ablation (RFA):
- Has been proposed as an alternative therapy in this setting
- A recent meta-analysis of 11 cohorts:
- Has shown RFA to be safe and effective in the management of PTMC over a relatively short follow-up period of up to two years
- As clinicians consider the management of PTMC:
- Longer-term data on the durability of RFA outcomes are needed
- This study was a single-institution report of the authors’ experience with RFA in a cohort of patients with PTMC
- Low-risk papillary thyroid microcarcinomas (PTMCs) have been shown to have an:
- Methods
- This was a retrospective study of 84 PTMCs in 74 patients at a single center in South Korea
- The study population was derived from a previously reported cohort of 133 patients who underwent RFA after September 2008 and had a follow-up of longer than 5 years
- Participants had PTMC, with exclusion of aggressive subtypes on cytology, while imaging was used to exclude gross extrathyroidal extension, cervical adenopathy and distant metastases
- All participants had either a contraindication to surgery or refused it
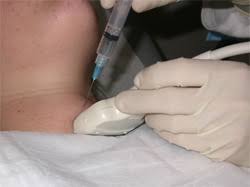
- The RFA procedure was performed under local anesthesia by a single radiologist, and the approach was trans-isthmic, using the moving-shot technique
- After RFA, patients were followed using ultrasound, interpreted by the same radiologist who performed the procedure, as well as clinical evaluations at 1, 6, and 12 months and annually thereafter for a minimum of 5 years
- The outcomes of interest included PTMC volume changes, newly developed PTMCs, lymph node involvement, distant metastasis, delayed surgery during the follow-up period, and RFA safety, defined by immediate and long-term complications
- Results
- The study cohort consisted of 74 adults with a mean age of 46 years
- There were 84 PTMCs treated with RFA, with 62% of them measuring < 5 mm (range, 3 to 10 mm maximum diameter)
- Only 13 lesions required a second RFA session because of incomplete ablation during the first RFA session (average, 1.2 RFA sessions / patient)
- Over a mean (±SD) follow-up period of 72±18 months:
- There were no cases of tumor progression, lymph node or distant metastases, or delayed surgery
- Complete tumor disappearance was noted by 60 months in all PTMCs:
- With 98.8% disappearance by 36 months
- Four new PTMCs developed in three participants’ remaining thyroid glands:
- These were also treated effectively with RFA
- Minor complications occurred in 4.1% of participants and included:
- Hematomas and first-degree burns
- Major complications occurred in 1.4% and included:
- Voice changes, with recovery by 2 months
- Conclusions
- In adults with low-risk PTMCs, RFA has been shown to be effective and safe in over 5 years of follow-up
- This approach has led to complete disappearance of all treated lesions and prevention of local tumor progression, metastasis, or delayed surgery, all with minimal risk of complication
- This study is the first to report on long-term follow-up of a sizable cohort of PTMC patients treated with RFA and found excellent efficacy and safety
- Previous reports were limited by a short duration of follow-up—a significant limitation considering the indolent course of PTMC
- However, there are some notable considerations:
- First, these data reflect experience at a single center with a single and highly experienced ultrasound and RFA operator, thus limiting external validity to centers with similar expertise:
- Experience with RFA is paramount given the reported doubling in ablated lesion volume early-on post ablation
- Due to ablation of parenchyma surrounding PTMCs:
- Which needs to be separated from residual tumor presence with need for repeat RFA
- Thus, without adequate expertise regarding interpretation of post-RFA imaging, unnecessary procedures might be performed
- Due to ablation of parenchyma surrounding PTMCs:
- Experience with RFA is paramount given the reported doubling in ablated lesion volume early-on post ablation
- Second, the favorable outcomes with regard to tumor recurrence and LN metastasis are in contrast with some previous reports:
- This is likely related to selection of less aggressive tumors:
- As evidenced by cytologic criteria and small tumor size (greater than 62% of participants had a tumor size less than 0.5 cm)
- Since thyroid cancer screening is no longer pursued, it is likely that the median size of RFA-treated PTMCs will increase, hence the need for revisiting the efficacy of the procedure periodically
- This is likely related to selection of less aggressive tumors:
- First, these data reflect experience at a single center with a single and highly experienced ultrasound and RFA operator, thus limiting external validity to centers with similar expertise:
- Overall, these results are certainly promising, but their application to all PTMCs remains limited
- In our practice, we do not routinely biopsy thyroid nodules less than 10 mm, in line with current American Thyroid Association (ATA) guidelines
- For greater adoption of RFA, this approach would need to be reconsidered
- At the same time, as RFA is being considered as an alternative to AS and surgery, patient-centered outcomes such as anxiety, pain, quality of life, and cost will be essential in future studies
- Currently, without direct comparison between RFA, AS, and surgery:
- RFA should be individualized to patient-specific situations:
- Small low-risk PTMCs
- Poor candidates for surgery or refusing surgery
- Anxiety about AS
- It should be considered only after careful discussion of standard treatment options
- For patients interest in RFA, special emphasis should be placed on the required expertise for this procedure, in order to replicate the excellent results described here
- RFA should be individualized to patient-specific situations:
#Arrangoiz #CancerSurgeon #HeadandNeckSurgeon #SurgicalOncologist #ThyroidSurgeon #ThyroidExpert #CASO #ThyroidNodules #CenterforAdvancedSurgicalOncology