Thyroid Awareness Month
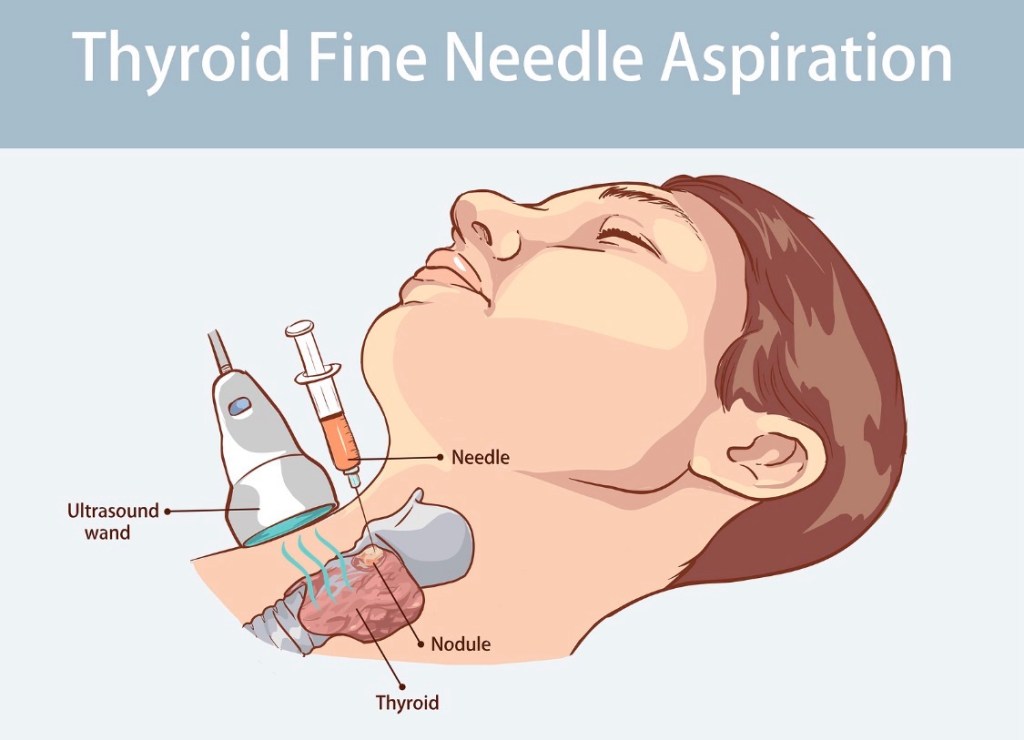
What Is a Thyroid Fine-Needle Aspiration (FNA) Biopsy?
A thyroid FNA biopsy is the standard, evidence-based test used to determine whether a thyroid nodule is benign or malignant—and it is far simpler than most patients expect.
🧪 How is the biopsy performed? (Step-by-step)
Ultrasound guidance precisely targets the nodule A very thin needle is used to collect cells Multiple passes may be taken to ensure accuracy The procedure takes 5–10 minutes You go home the same day and resume normal activities
✔️ No general anesthesia
✔️ Minimal discomfort
✔️ Very low risk of complications
🔬 What happens to the sample?
The cells are analyzed by an experienced cytopathologist and reported using the Bethesda System, which helps guide:
Observation Repeat biopsy Molecular testing (when appropriate) Surgery (only when necessary)
📊 Key facts for patients
Most biopsies come back benign FNA prevents unnecessary thyroid surgery Results allow for personalized, evidence-based care
🦋 Why this matters
A properly performed, ultrasound-guided FNA is:
Accurate Safe Essential for making the right treatment decision
👨⚕️ Dr. Rodrigo Arrangoiz, MD
Surgical Oncologist – Thyroid, Head & Neck, Breast
📌 Take-home message:
A thyroid biopsy is not something to fear —
it is a tool that helps ensure you get the right care and avoid unnecessary treatment.
📚 References
Cibas ES, Ali SZ. The Bethesda System for Reporting Thyroid Cytopathology. Thyroid Haugen BR et al. ATA Guidelines for Thyroid Nodules. Thyroid Gharib H et al. Fine-Needle Aspiration of Thyroid Nodules. Endocrine Practice