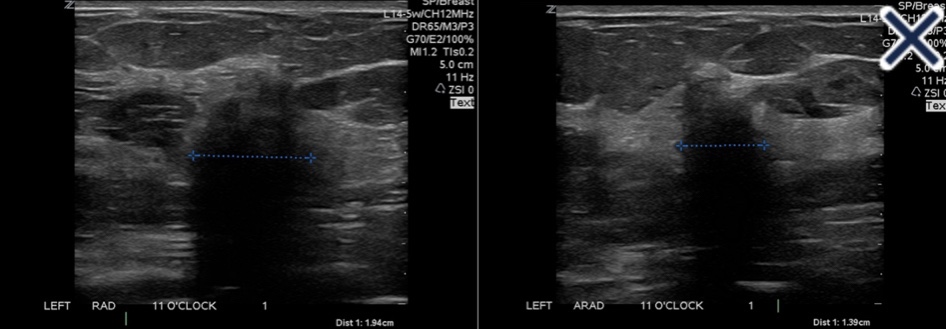
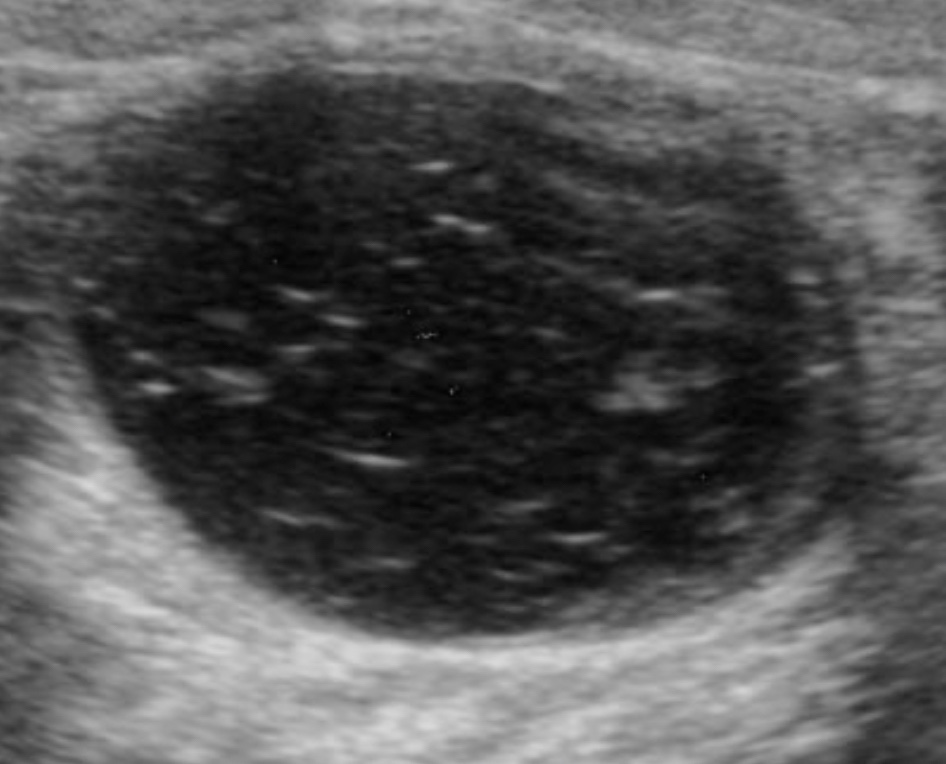
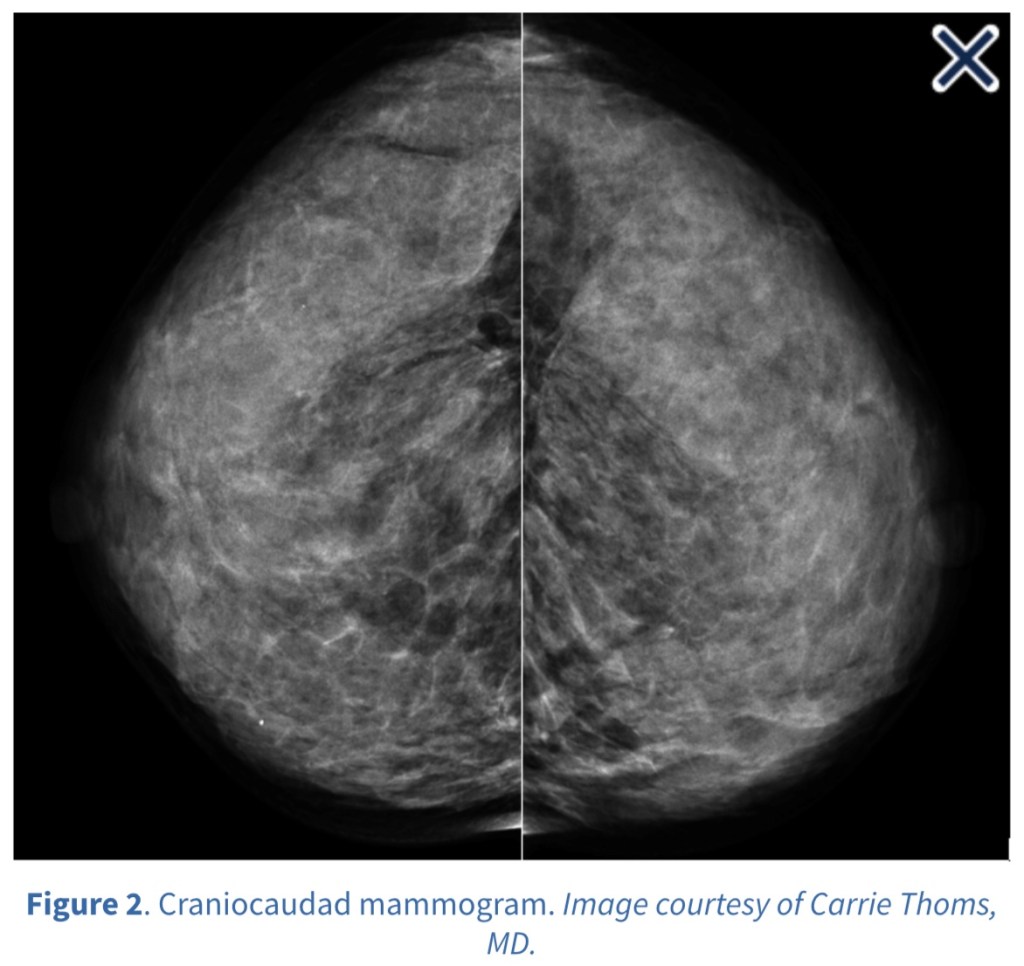
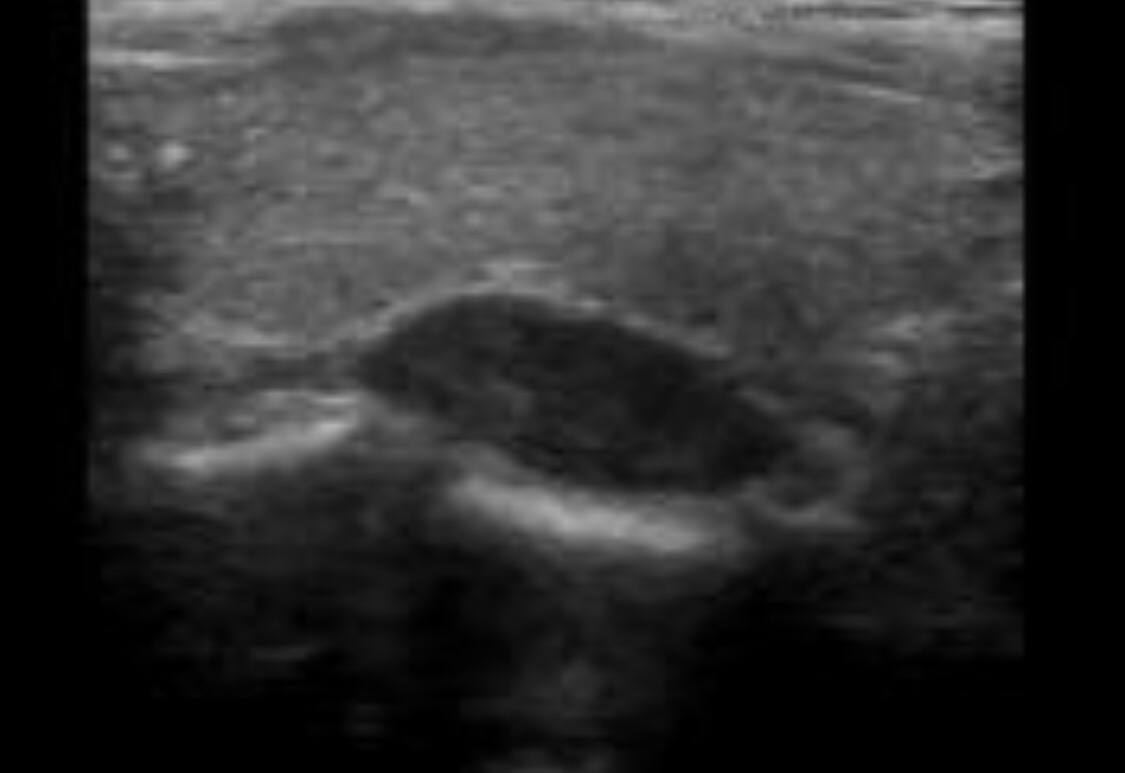
- Patients who are candidates for surgery for primary hyperparathyroidism (PHPT) but have negative or discordant imaging findings by ultrasound or Sestamibi scan:
- Should still undergo surgical evaluation according to American Association of Endocrine Surgeons guideilnes:
#Arrangoiz #ParathyroidSurgeon #ParathyroidExpert #Hyperparathyroidism #PrimaryHyperparathyroidism #CancerSurgeon #EndocrineSurgery #Teacher #Surgeon #HeadandNeckSurgeon #SurgicalOncologist #ParathyroidAdenoma #Hypercalcemia #ElevatedCalciumLevels #Miami #MountSinaiMedicalCenter #MSMC #Mexico #Hialeah
#Arrangoiz #ParathyroidSurgeon #ParathyroidExpert #Hyperparathyroidism #PrimaryHyperparathyroidism #CancerSurgeon #EndocrineSurgery #Teacher #Surgeon #HeadandNeckSurgeon #SurgicalOncologist #ParathyroidAdenoma #Hypercalcemia #ElevatedCalciumLevels #Miami #MountSinaiMedicalCenter #MSMC #Mexico #Hialeah