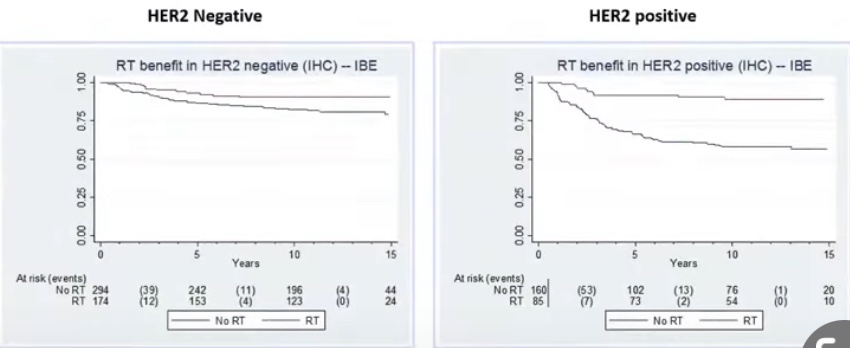
- To investigate the risk of ipsilateral breast events (IBEs) in patients with DCIS treated with local excision without radiation:
- The ECOG and North Central Cancer Treatment Group (NCCTG):
- Conducted a prospective trial between 1997 and 2002 known as the E5194 study
- Patients were stratified into two groups based on grade:
- Low- and intermediate-grade DCIS measuring 2.5 cm or smaller
- High-grade DCIS measuring 1 cm or smaller
- Margin widths of 3 mm or wider were required along with no residual calcifications on postoperative mammograms
- The low- and intermediate-grade DCIS group:
- Had a 12-year IBE rate of 14.4%
- The high-grade DCIS group:
- Had a 12-year IBE rate of 24.6% (p=0.003)
- Larger tumor size:
- Was also found to be statistically significantly associated with developing an IBE (P = .01)
- The risks of developing an IBE for either DCIS or invasive cancer:
- Increased over time through 12 years of follow-up:
- Without plateau
- Increased over time through 12 years of follow-up:
- The ECOG and North Central Cancer Treatment Group (NCCTG):
- Current literature reports a 50% decrease in local recurrence with radiotherapy after surgical excision of DCIS:
- However, we continue to look for low risk subsets of patients with DCIS:
- In whom the absolute benefit of radiation therapy of lumpectomy may be small
- However, we continue to look for low risk subsets of patients with DCIS:
- The DCIS score (12-gene signature):
- Was developed from tissue samples from the E5194 study:
- With the goal of identifying low and high-risk subsets of patients with DCIS following lumpectomy alone
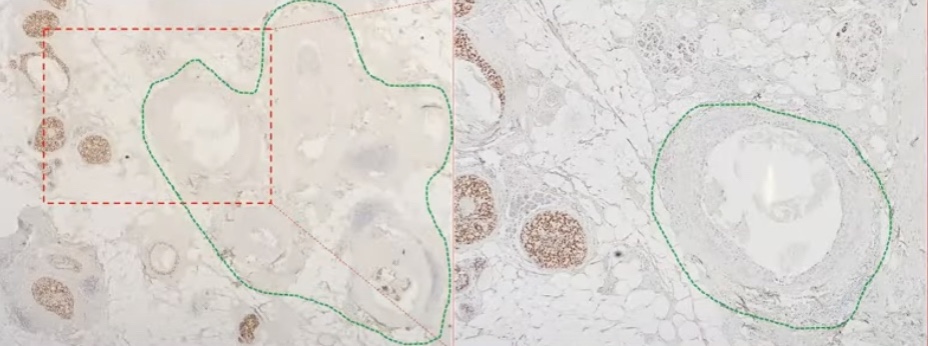
- This assay utilized quantitative reverse transcriptase-polymerase chain reaction (RT-PCR):
- From tumor specimens from 327 patients with DCIS treated with surgical excision without radiation from the E5194 study
- The DCIS score of 0 to 100 (low less than 39, intermediate 39 to 54, high ≥ 55):
- Was then designed to predict the recurrence of IBE overall, as well as DCIS or invasive cancer recurrence
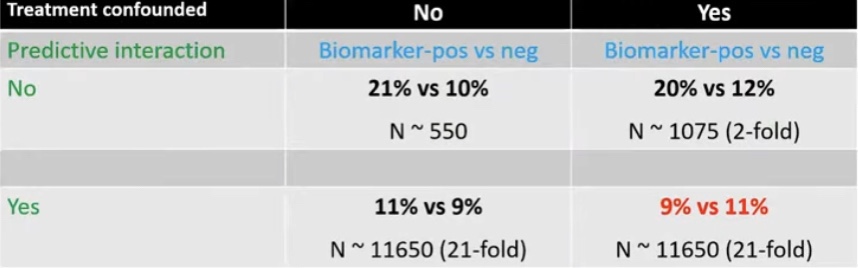
- The DCIS score correlated with 10-year IBE risk of:
- 10.6% in the low-risk group
- 26.7% in the intermediate-risk group
- 25.9% in the high-risk group
- Young age and larger tumor size:
- Also were found to be independent predictors of recurrence
- So, this test may be most beneficial in post-menopausal women with small tumors
- Was developed from tissue samples from the E5194 study:
- References:
- Solin LJ, Gray R, Hughes LL, Wood WC, Lowen MA, Badve SS, et al. Surgical excision without radiation for ductal carcinoma in situ of the breast: 12-year results from the ECOG-ACRIN E5194 Study. J Clin Oncol. 2015;33(33):3938-3944.
- Solin LJ, Gray R, Baehner FL, Butler SM, Hughes LL, Yoshizawa C, et al. A multigene expression assay to predict local recurrence risk for ductal carcinoma in situ of the breast. J Natl Cancer Inst. 2013;105(10):701-710.

#Arrangoiz #BreastSurgeon #CancerSurgeon #SurgicalOncologist #BreastCancer #DCIS #ECOG5194 #Teacher #Surgeon #Miami #MountSinaiMedicalCenter #Mexico