- Inflammatory breast cancer (IBC):
- Is a clinical diagnosis characterized by:
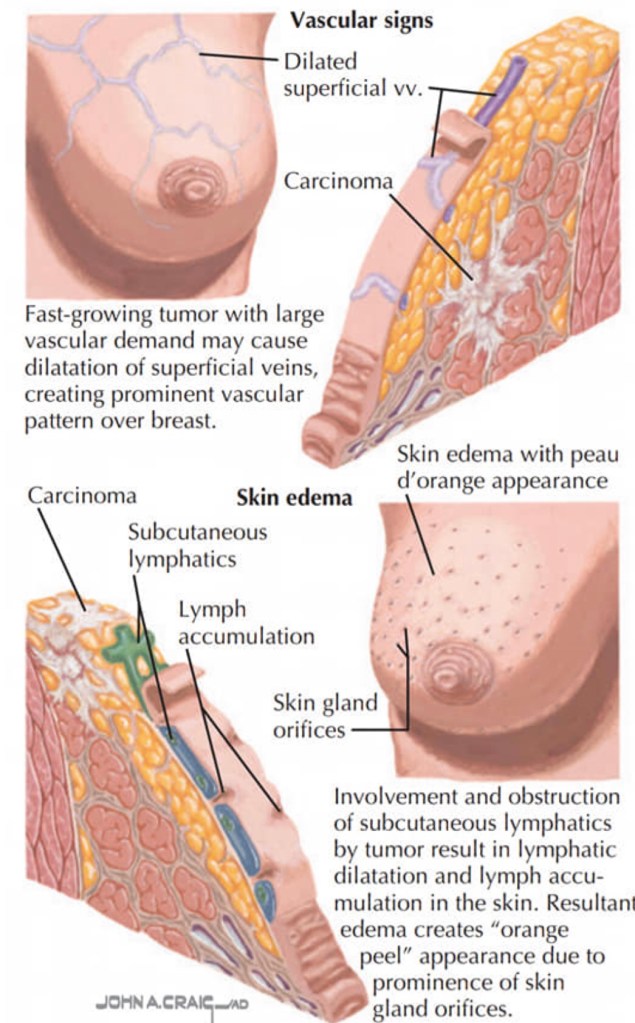
- The rapid progression of an enlarged breast with skin changes including:
- Redness
- Edema
- Peau d’orange
- The rapid progression of an enlarged breast with skin changes including:
- As mentioned previously IBC is a clinical diagnosis defined by the American Joint Committee on Cancer as a:
- “Diffuse erythema and edema involving approximately a third or more of the skin of the breast” and is staged cT4d
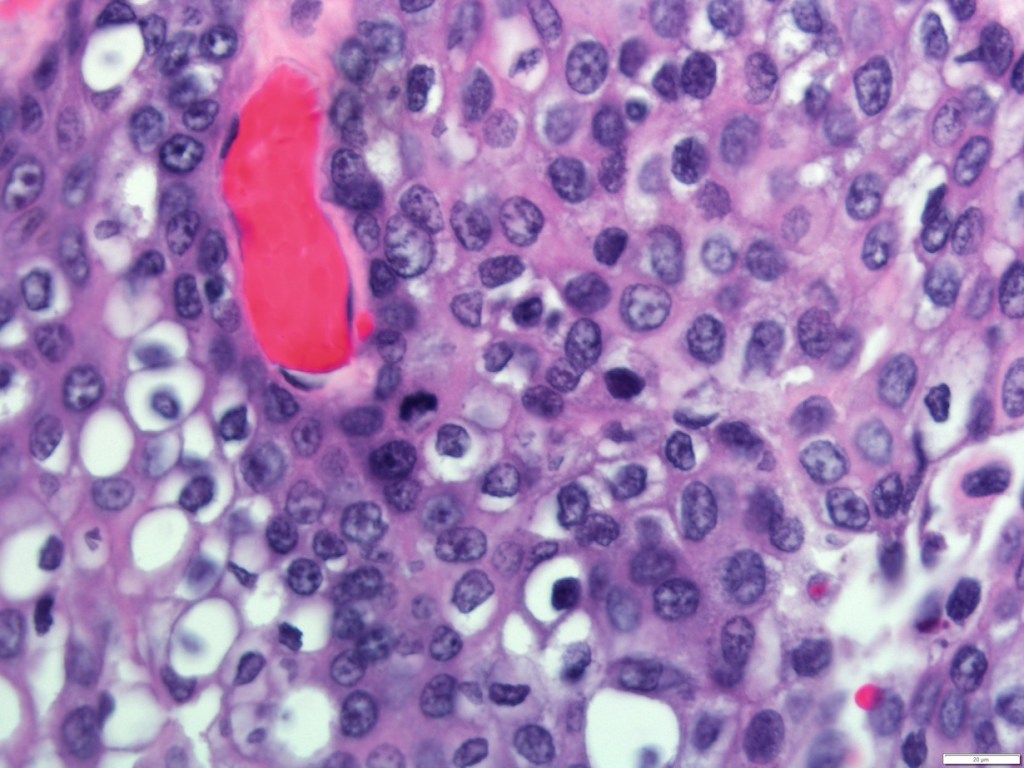
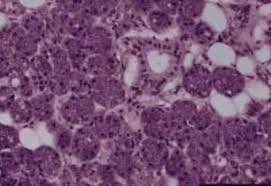
- A punch biopsy of the skin:
- Demonstrates tumor emboli within dermal lymphatics:
- Approximately 75% of the time
- A negative skin biopsy does NOT preclude the diagnosis, as it is clinical
- Demonstrates tumor emboli within dermal lymphatics:
- The appearance may lead to:
- Misdiagnosis of mastitis or breast cellulitis
- The rapid evolution of symptoms (within 3 to 6 months):
- Distinguishes IBC from a locally advanced breast cancer with associated edema
- IBC is rare:
- It presents in 2% to 4% of breast cancer patients:
- Although the reported annual incidence has been increasing.
- It presents in 2% to 4% of breast cancer patients:
- The tumor biology is disproportionately:
- ER negative and HER2 amplified, compared with non-IBC
- Patients should be evaluated in a multidisciplinary setting for trimodal therapy
- Treatment should be initiated with:
- Neoadjuvant chemotherapy, followed by aggressive local therapy
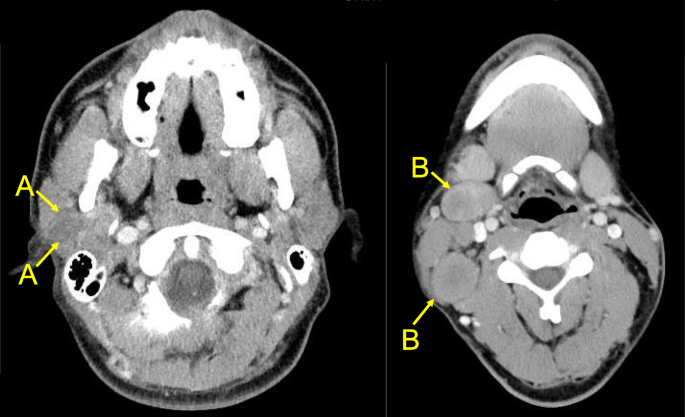
- The majority of patients with IBC present with clinical lymph node involvement:
- Sentinel lymph node biopsy is not reliable in IBC:
- Due to blockage of dermal lymphatics:
- Thus axillary dissection should be performed
- Due to blockage of dermal lymphatics:
- Sentinel lymph node biopsy is not reliable in IBC:
- Following neoadjuvant chemotherapy:
- Modified radical mastectomy is the appropriate surgery:
- Skin should not be spared so as not to leave behind residual disease
- Immediate reconstruction should be avoided
- Modified radical mastectomy is the appropriate surgery:
- Patients should receive post-mastectomy radiation:
- To the skin, chest wall, and regional lymph nodes following surgery:
- To optimize local control
- To the skin, chest wall, and regional lymph nodes following surgery:
- Survival in IBC has improved with trimodal therapy:
- A recent analysis of Surveillance, Epidemiology, and End Results data evaluated 10,197 patients with non-metastatic IBC between 1998 and 2010:
- Patients who underwent trimodal therapy had improved 5- and 10-year survival (55.4% and 37.3%) over those that did not receive all three modalities
- Survival was lowest at 10 years (16.5%) for patients who underwent surgery alone
- A recent analysis of Surveillance, Epidemiology, and End Results data evaluated 10,197 patients with non-metastatic IBC between 1998 and 2010:
- Staging scans, including a CT chest / abdomen/ pelvis, PET scan, and / or bone scan:
- Should be completed prior to initiating treatment
- Inflammatory breast cancer is a clinical stage T4d:
- And the most fatal form of breast cancer:
- Accounting for 7% of all breast cancer deaths:
- Real-world observational data have demonstrated that inflammatory breast cancer has significantly worse survival compared to other non-metastatic locally advanced and metastatic non-inflammatory breast cancers
- Despite this, 5-year survival of IBC patients has increased from:
- 40% to 50% in the 1990’s to almost 70% in 2008
- Accounting for 7% of all breast cancer deaths:
- And the most fatal form of breast cancer:
- Recent national and international guidelines for IBC recommend:
- Full staging (PET / CT preferred over CT chest / abdomen / pelvis + bone scan) and bilateral breast and axillary nodal imaging, followed by neoadjuvant systemic therapy, modified radical mastectomy (including level I and II lymph node dissection), and radiation
- Adjuvant targeted therapy and hormonal therapy should be considered in appropriate cases
- Notably, lumpectomy is contraindicated
- Breast reconstruction should be delayed
- Multi-modal therapy for IBC has resulted in the best overall survival rates
- For HER2-negative breast cancers:
- Preoperative chemotherapy regimens should include:
- Sequential doxorubicin and cyclophosphamide followed by a taxane:
- To achieve the highest pathologic complete response rate
- Sequential doxorubicin and cyclophosphamide followed by a taxane:
- Preoperative chemotherapy regimens should include:
- For HER2-positive breast cancers:
- Chemotherapy should be used with dual anti-HER2-directed therapy with pertuzumab and trastuzumab:
- To achieve the best pathologic complete response rate
- Chemotherapy should be used with dual anti-HER2-directed therapy with pertuzumab and trastuzumab:
- Is a clinical diagnosis characterized by:
- References
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Breast Cancer. Available with login at: https://subscriptions.nccn.org.
- Fouad TM, Barrera AMG, Reuben JM, Lucci A, Woodward WA, Stauder MC, et al. Inflammatory breast cancer: a proposed conceptual shift in the UICC-AJCC TNM staging system. Lancet Oncol. 2017;18(4):e228-e232.
- Ueno NT, Espinosa Fernandez JR, Cristofanilli M, Overmoyer B, Rea D, Berdichevski F, et al. International consensus on the clinical management of inflammatory breast cancer from the Morgan Welch Inflammatory Breast Cancer Research Program 10th Anniversary Conference. J Cancer. 2018;9(8):1437-1447.
- Rueth NM, Lin HY, Bedrosian I, Shaitelman SF, Ueno NT, Shen Y, et al. Underuse of trimodality treatment affects survival for patients with inflammatory breast cancer: an analysis of treatment and survival trends from the National Cancer Database. J Clin Oncol. 2014;32(19):2018-2024.
- Amin MB, Edge S, Greene F, et al., eds. AJCC Cancer Staging Manual. 8th ed. New York, NY: Springer; 2017
- Hance KW, Anderson WF, Devesa SS, Young HA, Levine PH. Trends in inflammatory breast carcinoma incidence and survival: the surveillance, epidemiology, and end results program at the National Cancer Institute. J Natl Cancer Inst 2005;97(13):966-975.
- NCCN clinical practice guidelines in oncology. National Comprehensive Cancer Network. http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed August 25, 2019.
- Menta A, Fouad TM, Lucci A, et al. Inflammatory breast cancer: what to know about this unique, aggressive breast cancer. Surg Clin North Am. 2018;98(4):787-800.
- Rueth NM, Lin HY, Bedrosian I, et al: Underuse of trimodality treatment affects survival for patients with inflammatory breast cancer: an analysis of treatment and survival trends from the National Cancer Database. J Clin Oncol. 2014;32(19):2018-2024.
#Arrangoiz #BreastSurgeon #CancerSurgeon #SurgicalOncologist #BreastCancer #InflammatoryBreastCancer #CASO #CenterforAdvancedSurgicalOncology