Thyroid cancer (TC) represents the most common endocrine malignancy, accounting for 3.4% of all cancers diagnosed annually.
The transformation of the thyroid follicular cells may result in differentiated or undifferentiated TC, through a multistep process that is the most accepted theory of follicular cell carcinogenesis.
In this model, distinct molecular alterations have been associated with specific stages, driving progression from well-differentiated to undifferentiated follicular-derived thyroid carcinomas.
More recently, the cancer stem-like cells theory has been proposed, according to which phenotypically different cancer cells could be generated by a small subpopulation of stem cells after genetic and epigenetic transformations.
Differentiated TC, accounting for more than 90% of thyroid malignancies, comprises papillary thyroid carcinoma (PTC) and follicular thyroid carcinoma (FTC).
Poorly differentiated thyroid carcinoma (PDTC) and anaplastic thyroid carcinoma (ATC) are rare tumors (5 and 1%, respectively) associated with aggressive behavior and short median time of survival (5 years and 6 months, respectively).
Differently, medullary thyroid carcinoma (MTC), representing 5% of TC, arises from parafollicular C cells.
In the last 30 years, the availability of the genome sequence has produced much progress in elucidating the molecular mechanisms underlying TC.
TC is a relatively simple genetic disease with a relatively low somatic mutation burden in each tumor.
Driver mutations, i.e., mutations that provide a selective growth advantage thus promoting cancer development, are identified in more than 90% of TC.
The molecular pathogenesis of the majority of TC involves dysregulation of the mitogen-activated protein kinase (MAPK) and phosphatidylinositol-3 kinase (PI3K)/AKT signaling pathways.
MAPK activation is considered to be crucial for PTC initiation, through point mutations of the BRAF and RAS genes or gene fusions of RET/PTC and TRK.
On the other hand, PI3K/AKT activation is thought to be critical in FTC initiation and can be triggered by activating mutations in RAS, PIK3CA, and AKT1 as well as by inactivation of PTEN, which negatively regulates this pathway.
TC progression and dedifferentiation to PDTC and ATC involves a number of additional mutations affecting other cell signaling pathways, such as p53 and Wnt/β-catenin.
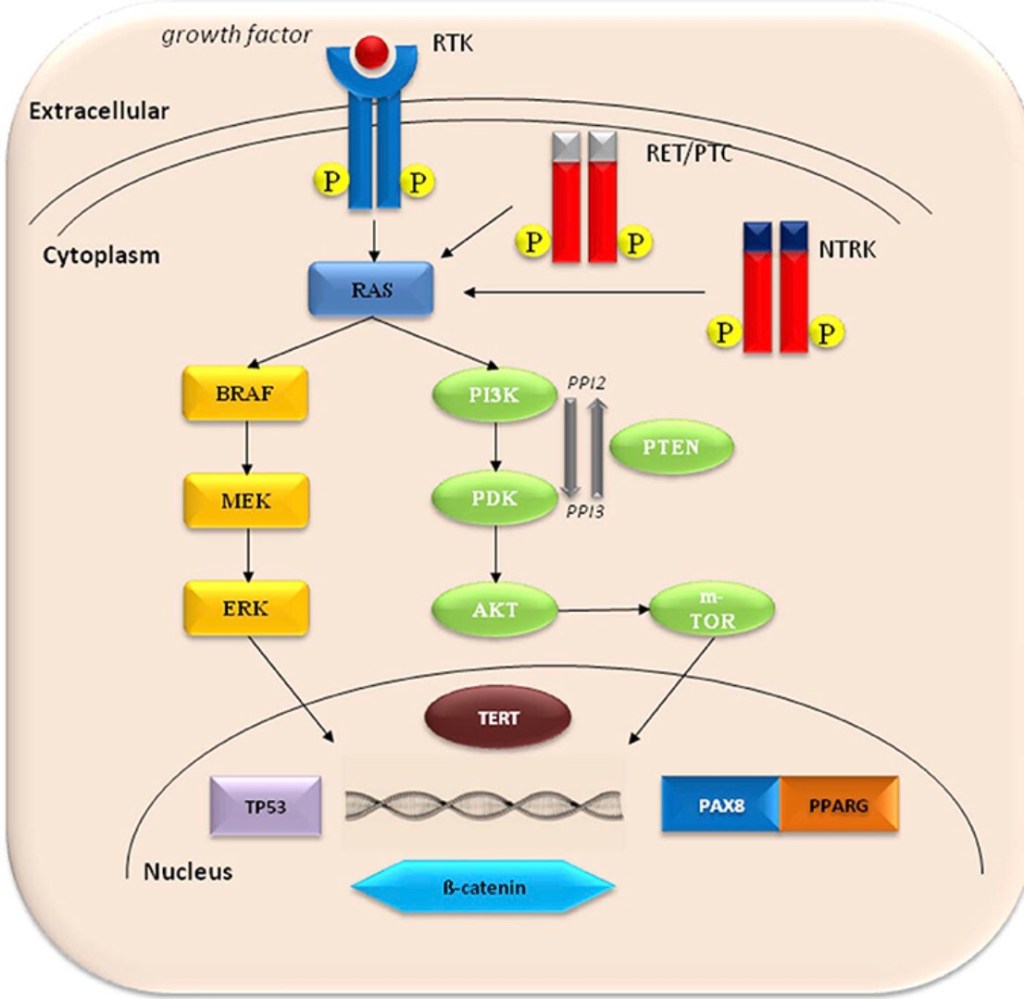
Common activating mutations in the MAPK pathway include RET-PTC and NTRK rearrangements, and RAS and BRAF mutations.
Common genetic alterations in the PI3K pathway include RAS mutations, PTEN mutations or deletions, PIK3K mutations or amplifications, and AKT1mutations.
PAX8-PPARG fusions are common in FTC. Activation of Wnt/b-catenin pathway, inactivating mutations in TP53, and activating mutations in TERT promoter are frequent in undifferentiated thyroid cancer.
More recently, TERT promoter mutations have been described in all the histological TC type, with a significantly higher prevalence in aggressive and undifferentiated tumors, indicating their role in TC progression.
Mutations in the RET (Rearranged during transfection) proto-oncogene account for most MTC cases and can occur sporadically or as inherited germline events in the multiple endocrine neoplasia type 2A (MEN2A) and 2B (MEN2B) syndromes.
A minority of sporadic MTC are caused by H-, K-, and N-RAS mutations.
#Arrangoiz #ThyroidSurgeon #ThyroidExpert #ThyroidCancer #CancerSurgeon #SurgicalOncologist #HeadandNexkSurgeon
