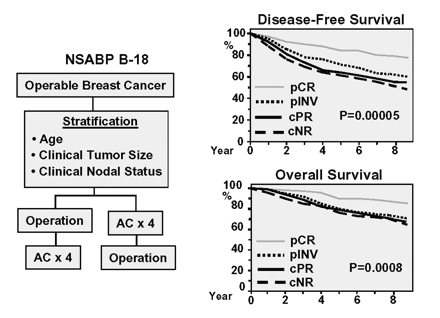
- The NSABP B-18 trial:
- Was initiated in 1988 to determine:
- Whether four cycles of doxorubicin and cyclophosphamide given preoperatively:
- Improved survival and DFS:
- When compared with the same chemotherapy given postoperatively
- Secondary aims included:
- The evaluation of preoperative chemotherapy:
- In downstaging the primary breast tumor and involved axillary lymph nodes
- The evaluation of preoperative chemotherapy:
- Improved survival and DFS:
- Whether four cycles of doxorubicin and cyclophosphamide given preoperatively:
- Women aged 50 years or older:
- Also received tamoxifen for 5 years
- The results of the trial showed that:
- With preoperative chemotherapy:
- 36% of patients:
- Achieved a clinical complete response
- 43% of patients:
- Achieved a clinical partial response
- 13% of patients:
- Achieved a pathologic complete response
- 36% of patients:
- Patients who received preoperative chemotherapy:
- Were more likely to receive breast-conserving surgery:
- 67% vs 60%:
- Than patients receiving postoperative chemotherapy
- 67% vs 60%:
- Were more likely to receive breast-conserving surgery:
- There was no difference in:
- DFS
- Distant DFS
- Overall survival:
- Between the two groups
- With preoperative chemotherapy:
- At 9 years:
- The rate of ipsilateral breast tumor recurrence:
- Was numerically higher in the preoperative chemotherapy group than the postoperative chemotherapy group (10.7% vs 7.6%):
- Although this difference was not statistically significant (P=0.12)
- Was numerically higher in the preoperative chemotherapy group than the postoperative chemotherapy group (10.7% vs 7.6%):
- The rate of ipsilateral breast tumor recurrence:
- Was initiated in 1988 to determine:
REFERENCES
- Fisher ER, Wang J, Bryant J, Fisher, Mamounas E, Wolmark N. Pathobiology of preoperative chemotherapy: findings from the National Surgical Adjuvant Breast and Bowel (NSABP) protocol B-18. Cancer. 2002;95:681-695.
- Wolmark N, Wang J, Mamounas E, Bryant J, Fisher B. Preoperative chemotherapy in patients with operable breast cancer: nine-year results from National Surgical Adjuvant Breast and Bowel Project B-18. J Natl Cancer Inst Monogr. 2001:96-102.
#Arrangoiz #BreastSurgeon #CancerSurgeon #SurgicalOncologist #BreastCancer #Surgeon #Teacher
