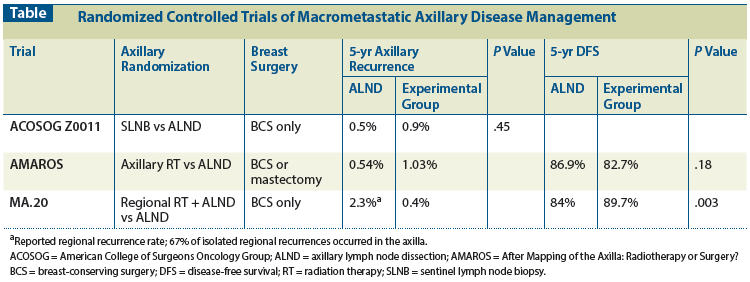
- The European Organisation for Research and Treatment of Cancer (EORTC) 10981 After Mapping of the Axilla, Radiation or Surgery? (AMAROS) trial:
- Randomized 4806:
- Clinically node-negative
- T1 to T2 tumors:
- To completion axillary lymph node dissection (ALND) or axillary radiation (AR):
- If they had positive nodes
- To completion axillary lymph node dissection (ALND) or axillary radiation (AR):
- Of the 1425 patients with positive sentinel nodes:
- 744 had been randomly assigned to ALND
- 681 to AR
- Initially:
- Patients with tumors 3 cm or smaller were eligible:
- But the protocol was later modified to include:
- Tumors up to 5 cm, multifocal tumors, or both
- But the protocol was later modified to include:
- Patients with tumors 3 cm or smaller were eligible:
- In the AR arm 21% of patients had T2 lesions
- There were no age limits for eligibility:
- Patients ranged from age 48 to 64 years
- In the AR arm:
- 42% of women were premenopausal
- Patients who had either partial or total mastectomy were enrolled in the trial:
- 18% of women had mastectomy
- There was no limit on the number of positive nodes for the radiation arm:
- But the majority of patients had 1 or 2 positive nodes
- Crossover was allowed:
- For patients with extensive axillary disease:
- From the radiation arm to the dissection arm
- Patients in the dissection arm who had 4 or more positive nodes:
- Where allowed to have axillary radiation
- Four percent of patients who stayed in the radiation arm had 3 positive nodes, and 1% had 4 or more positive nodes.
- For patients with extensive axillary disease:
- Importantly:
- In the axillary dissection arm:
- 25% of patients had an additional 1 to 3 positive nodes (in addition to the positive sentinel nodes) at dissection
- 8% had 4 or more additional positive nodes
- Since it was a randomized trial:
- We can assume the same numbers were present in the radiation arm:
- So these patients did not necessarily have low-volume axillary disease.
- We can assume the same numbers were present in the radiation arm:
- In the axillary dissection arm:
- Axillary radiotherapy (RT) included:
- The contents of all 3 levels of the axilla and the medial part of the supraclavicular fossa
- The prescribed dose was 25 fractions of 2 Gy each
- For patients in the AR arm who had mastectomy:
- Radiation to the chest wall in addition to the axilla was optional but not mandatory
- There were no significant differences:
- In 5-year overall survival or disease-free survival between the two arms:
- At 6.1 years of follow-up:
- There was no significant difference:
- In the rate of axillary failure:
- 0.43% ALND vs 1.19% RT
- In the rate of axillary failure:
- There was no significant difference:
- At 5-year follow-up there was a significant difference:
- In clinical signs of lymphedema between the groups:
- 23% in ALND vs. 11% in AR:
- There was greater than a 10% difference in arm size compared to the contralateral arm:
- In 13% of the ALND arm and 5% of the AR arm
- There was greater than a 10% difference in arm size compared to the contralateral arm:
- 23% in ALND vs. 11% in AR:
- In clinical signs of lymphedema between the groups:
- At 6.1 years of follow-up:
- In 5-year overall survival or disease-free survival between the two arms:
- Randomized 4806:
- The AMAROS study findings would suggest that axillary RT is an appropriate alternative to ALND in patients with a positive sentinel node:
- However, the clinical characteristics of the AMAROS cohort are remarkably similar to the American College of Surgeons Oncology Group (ACOSOG) Z0011 cohort:
- With 80% of AMAROS patients having:
- A tumor less than 2 cm
- 90% patients:
- Receiving any systemic therapy
- 95% of patients:
- Having only 1 to 2 positive sentinel nodes
- With 80% of AMAROS patients having:
- Patients in ACOSOG Z0011 treated with sentinel lymph node biopsy only demonstrated:
- Similar 5-year rates of regional recurrence as the AMAROS patients receiving axillary RT:
- 0.9% [ACOSOG Z0011] vs 1.2% [EORTC 10981 AMAROS axillary RT]):
- Thus, while AMAROS indicates that sentinel node biopsy and nodal RT is an alternative to ALND:
- It does not demonstrate that RT is necessary in all patients with a positive sentinel node:
- Particularly in those treated with breast-conserving surgery
- It does not demonstrate that RT is necessary in all patients with a positive sentinel node:
- Thus, while AMAROS indicates that sentinel node biopsy and nodal RT is an alternative to ALND:
- 0.9% [ACOSOG Z0011] vs 1.2% [EORTC 10981 AMAROS axillary RT]):
- Similar 5-year rates of regional recurrence as the AMAROS patients receiving axillary RT:
- The decision to include axillary RT in patients with 1 to 2 positive sentinel nodes:
- Should be tailored to the individual:
- Taking into account other clinical factors which may place the patient at higher risk for locoregional recurrence
- Should be tailored to the individual:
- However, the clinical characteristics of the AMAROS cohort are remarkably similar to the American College of Surgeons Oncology Group (ACOSOG) Z0011 cohort: