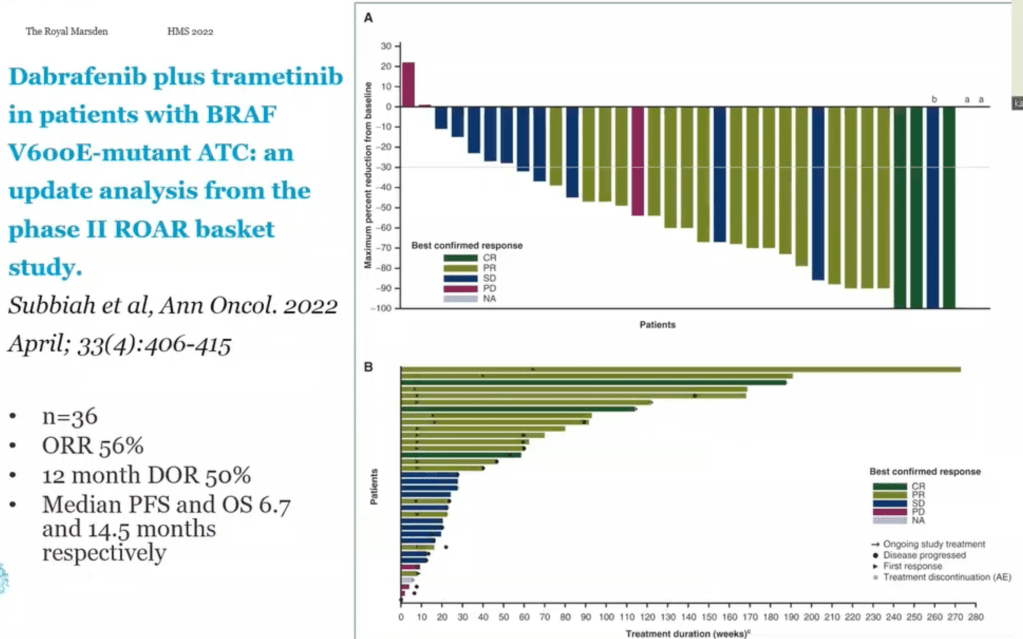
- The use of an aromatase inhibitor (letrozole) with an inhibitor of the cyclin dependent kinases 4 and 6 (ribociclib) was compared with aromatase inhibitor alone in postmenopausal women with hormone receptor positive HER2-negative metastatic breast cancer in the MONALEESA-2 study
- Results showed with the addition of ribociclib to letrozole alone:
- An improvement in:
- Progression-free survival (PFS):
- From 42.2% to 63%
- Overall response rate:
- From 37.1% to 52.7%
- Progression-free survival (PFS):
- An improvement in:
- This regimen was also investigated in premenopausal women with advanced, hormone receptor-positive breast cancer, and improved PFS compared with placebo plus endocrine therapy
References
1. Hortobagi GN, Stemmer SM, Burris HA, Yap YS, Sonke GS, Paluch-Shimon S, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med.2016;375(18)1738-1748.
2. Tripathy D, Im SA2, Colleoni M3, Franke F4, Bardia A5, Harbeck Nm et al. Ribociclib plus endocrine therapy for premenopausal women with hormone-receptor-positive, advanced breast cancer (MONALEESA-7): a randomised phase 3 trial. Lancet Oncol. 2018;19(7):904-915.