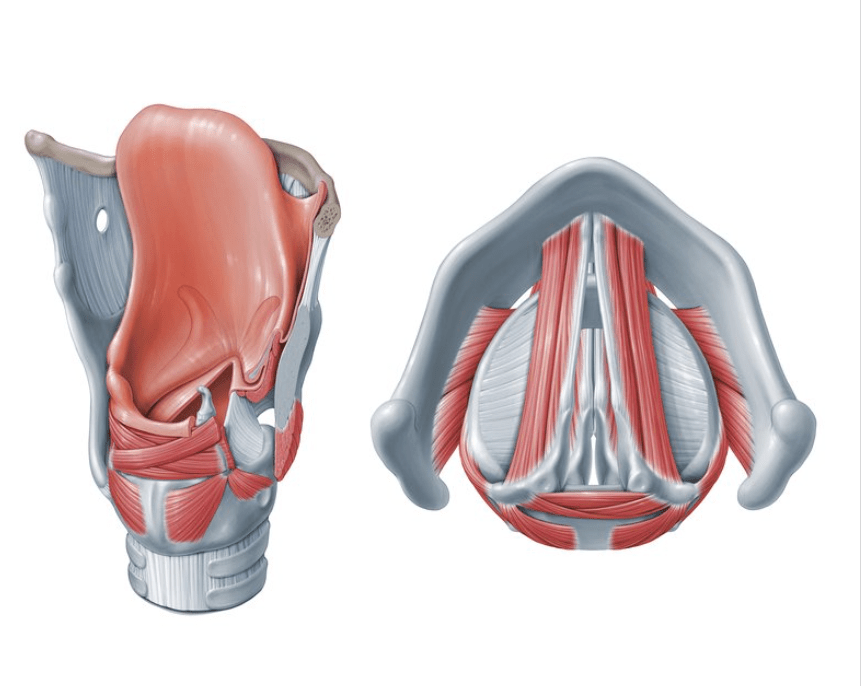
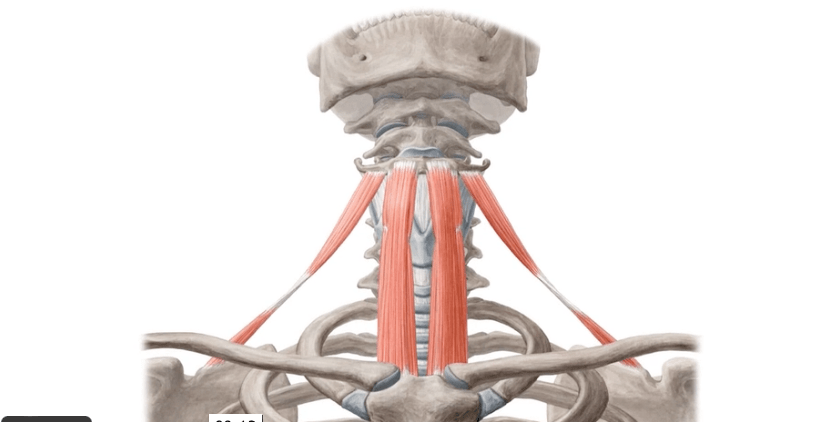
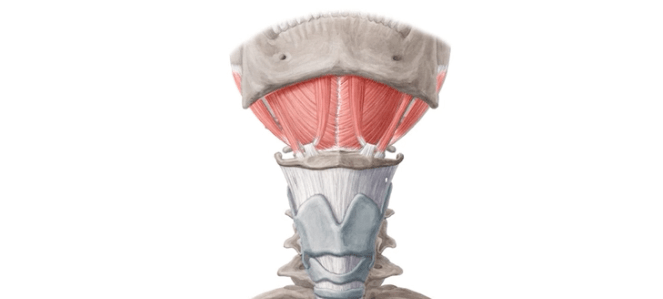
- Standard fractionation whole-breast irradiation (WBI):
- Represents a standard radiation technique used following breast-conserving surgery and is part of current evidence-based guidelines
- Hypofractionated WBI:
- Has been found to be equivalent to standard fractionation WBI in several randomized trials
- Updated American Society for Radiation Oncology (ASTRO) guidelines:
- Recommend its use at any age without any restrictions on prior chemotherapy
- Any stage can be treated as long as the regional lymph nodes do not require an addition field
- The dose homogeneity goal is to minimize the breast receiving greater than 105% of the prescription dose
- Accelerated partial breast irradiation (APBI):
- Has been found to have comparable rates of local recurrence and toxicity compared with standard / hypofractionated WBI in multiple randomized trials:
- With current evidence-based guidelines available from ASTRO and the American Brachytherapy Society for off-protocol use
- Has been found to have comparable rates of local recurrence and toxicity compared with standard / hypofractionated WBI in multiple randomized trials:
- Intraoperative radiation therapy (IORT):
- Has been found to have higher rates of local recurrence in the TARGIT and ELIOT trials compared with standard fractionation WBI, and is not included in commonly utilized evidence-based guidelines at this time
- References
- Fisher B, Anderson S, Bryant J, et al. Twenty-year followup of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med 2002;347(16):1233-1241.
- Smith BD, Bellon JR, Blitzblau R, et al. Radiation therapy for the whole breast: Executive summary of an American Society for Radiation Oncology (ASTRO) evidence-based guideline. Pract Radiat Oncol. 2018;8(3):145-152
- Correa C, Harris EE, Leonardi MC, et al. Accelerated partial breast irradiation: executive summary for the update of an ASTRO evidence-based consensus statement. Pract Radiat Oncol. 2017;7(2):73-79.
- Shah C, Vicini F, Shaitelman SF, et al. The American Brachytherapy Society consensus statement for accelerated partial-breast irradiation. Brachytherapy.2018;17(1):154-170.
- Vaidya JS, Wenz F, Bulsara M, et al. Risk-adapted targeted intraoperative radiotherapy versus whole-breast radiotherapy for breast cancer: 5-year results for local control and overall survival from the TARGIT-A randomised trial. Lancet 2014(9917);383:603-613.
- Veronesi U, Orecchia R, Maisonneuve P, et al. Intraoperative radiotherapy versus external radiotherapy for early breast cancer (ELIOT): a randomised controlled equivalence trial. Lancet Oncol. 2013;14(13):1269-1277.