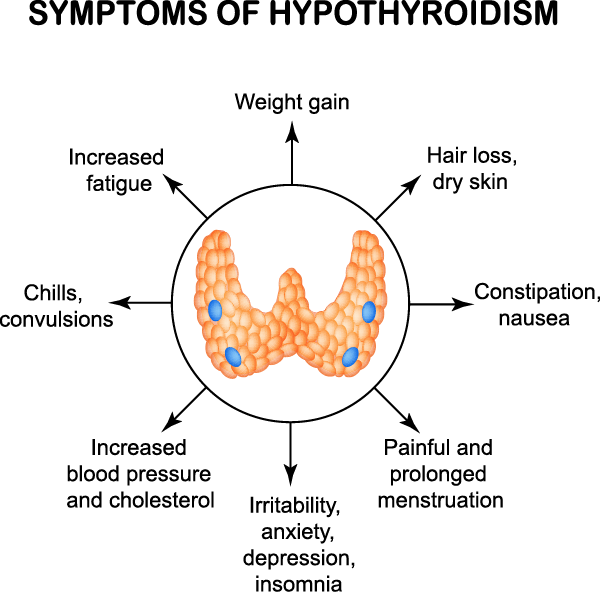
- Subacute thyroiditis (like painless sporadic thyroiditis and postpartum thyroiditis):
- Is a spontaneous remitting inflammatory disorder of the thyroid:
- That may last for weeks to months (has a more sudden onset)
- This disorder has a number of eponyms, including:
- De Quervain’s thyroiditis
- Giant cell thyroiditis
- Pseudo-granulomatous thyroiditis
- Subacute painful thyroiditis
- Subacute granulomatous thyroiditis
- Acute simple thyroiditis
- Noninfectious thyroiditis
- Acute diffuse thyroiditis
- Migratory “creeping” thyroiditis
- Pseudotuberculous thyroiditis
- Viral thyroiditis
- The first description of subacute thyroiditis was in:
- 1895 by Mygind:
- Who reported 18 cases of “thyroiditis akuta simplex
- Who reported 18 cases of “thyroiditis akuta simplex
- 1895 by Mygind:
- The pathology of subacute thyroiditis was first described:
- In 1904 by Fritz De Quervain:
- Whose name is associated with the disorder:
- He showed giant cells and granulomatous type changes in the thyroids of affected patients
- Whose name is associated with the disorder:
- In 1904 by Fritz De Quervain:
- Subacute thyroiditis:
- Is the most common cause of:
- The painful thyroid:
- May account for up to 5% of clinical thyroid abnormalities
- The painful thyroid:
- Is the most common cause of:
- As with other thyroid disorders:
- Women are more frequently affected than men:
- 5 to 1 (Hashimoto’s Thyroiditis is 8 to 9 / 1)
- Women are more frequently affected than men:
- The peak incidence is in the:
- Fourth and fifth decades of life (20 to 60 years of age):
- This disorder is rarely observed in children and the elderly
- Fourth and fifth decades of life (20 to 60 years of age):
- Although the term subacute thyroiditis connotes a temporal quality that could apply to any thyroidal inflammatory process of intermediate duration and severity:
- It is actually referring specifically to the granulomatous appearance of the thyroid found on pathologic exam
- Pathogenesis:
- Infectious Association:
- Although there is no clear evidence for a specific etiology:
- Indirect evidence suggests that subacute thyroiditis:
- May be caused by a viral infection of the thyroid
- Indirect evidence suggests that subacute thyroiditis:
- The condition is often preceded by a:
- Prodromal phase of:
- Myalgia General
- Malaise
- Low-grade fevers
- Fatigue
- Frequently by an upper respiratory tract infection
- Prodromal phase of:
- Although there is no clear evidence for a specific etiology:
- Infectious Association:
- It has been reported most frequently in:
- The temperate zone:
- Only rarely from other parts of the world
- The temperate zone:
- It has been found to occur seasonally:
- The highest incidence is in the summer months:
- July through September:
- Which coincide with the peak of enterovirus:
- Echovirus infection
- Coxsackie virus A and B infection
- Which coincide with the peak of enterovirus:
- July through September:
- The highest incidence is in the summer months:
- The incidence rate has been shown to vary directly with:
- Viral epidemics:
- Specifically mumps:
- The incidence of subacute thyroiditis has been found to be higher during these viral epidemics
- Interestingly:
- Antibodies to the mumps virus have even been detected in individuals with subacute thyroiditis who do not have clinical evidence of mumps
- Subacute thyroiditis has also been associated with:
- Measles
- Influenza
- The common cold
- Adenovirus
- Infectious mononucleosis
- Coxsackie virus
- Myocarditis
- Cat scratch fever
- St. Louis encephalitis
- Hepatitis A
- The parvovirus B19 infection
- Antibodies to Coxsackie virus, adenovirus, influenza, and mumps have been detected in the:
- Convalescent phase of this disease
- Coxsackie virus is most commonly:
- Associated with subacute thyroiditis
- Coxsackie virus antibody titers:
- Have been shown to directly follow the course of the thyroid disease
- Specifically mumps:
- Certain non-viral infections, including:
- Q fever and malaria:
- Have been associated with a clinical syndrome similar to subacute thyroiditis
- Have been associated with a clinical syndrome similar to subacute thyroiditis
- A case of subacute thyroiditis occurring simultaneously with:
- Giant cell arteritis has been reported
- Giant cell arteritis has been reported
- Another case of subacute thyroiditis developed during:
- Alfa-interferon treatment for hepatitis C
- Alfa-interferon treatment for hepatitis C
- Q fever and malaria:
- Viral epidemics:
- Autoimmune Association:
- Unlike painless or postpartum thyroiditis:
- There is no clear association between subacute thyroiditis and autoimmune thyroid disease:
- Serum thyroid peroxidase and thyroglobulin antibodies levels:
- Are usually normal
- Are usually normal
- When decreased the levels of thyroid peroxidase and thyroglobulin antibodies:
- Correlated with the phase of transient hypothyroidism
- Correlated with the phase of transient hypothyroidism
- Antibodies to an un-purified thyroid preparation can be detected:
- For up to 4 years after a bout of subacute thyroiditis
- For up to 4 years after a bout of subacute thyroiditis
- Antibodies to the thyrotropin (TSH) receptor:
- Have been rarely detected during the course of subacute thyroiditis
- In most studies:
- There was no correlation between the presence of thyrotropin receptor binding inhibitory immunoglobulin (TBII) or of thyrotropin receptor stimulating immunoglobulin and the thyrotoxic phase of the thyroiditis
- On the other hand, there has been some correlation between thyroid-blocking antibodies and the development of hypothyroidism
- It is thought that the appearance of the TSH-receptor antibodies results from an immune response:
- That occurs after there is damage to the thyrocytes, specifically membrane desquamation
- That occurs after there is damage to the thyrocytes, specifically membrane desquamation
- Serum thyroid peroxidase and thyroglobulin antibodies levels:
- There is no clear association between subacute thyroiditis and autoimmune thyroid disease:
- Unlike painless or postpartum thyroiditis:
- Following recovery from the inflammatory process of subacute thyroiditis:
- All immunologic phenomena disappear:
- The transitory immunologic markers that are observed during the course of subacute thyroiditis:
- Appear to occur in response to the release of antigenic material from the thyroid
- Appear to occur in response to the release of antigenic material from the thyroid
- The transitory immunologic markers that are observed during the course of subacute thyroiditis:
- All immunologic phenomena disappear:
- Genetic Association:
- There is an apparent genetic predisposition for subacute thyroiditis:
- With HLA-Bw 35 reported in all ethnic groups:
- The relative risk of HLA-Bw 35 in subacute thyroiditis:
- Is high:
- Ranging from 8 to 56
- Is high:
- Additional evidence for genetic susceptibility is the:
- Simultaneous development of subacute thyroiditis in identical twins heterozygous for the HLA-Bw 35 haplotypes
- A weak association of subacute thyroiditis with:
- HLA-DRw8 has been reported in Japanese patients
#Arrangoiz #CancerSurgeon #ThyroidSurgeon #ParathyroidSurgeon #HeadandNeckSurgeon #ThyroidExpert #SurgicalOncologist #EndocrineSurgery #MountSinaiMedicalCenter #Miami #ThyroidNodule #deQuervain’sthyroiditis #Subacutethyroiditis
- HLA-DRw8 has been reported in Japanese patients
- Simultaneous development of subacute thyroiditis in identical twins heterozygous for the HLA-Bw 35 haplotypes
- There is an apparent genetic predisposition for subacute thyroiditis:
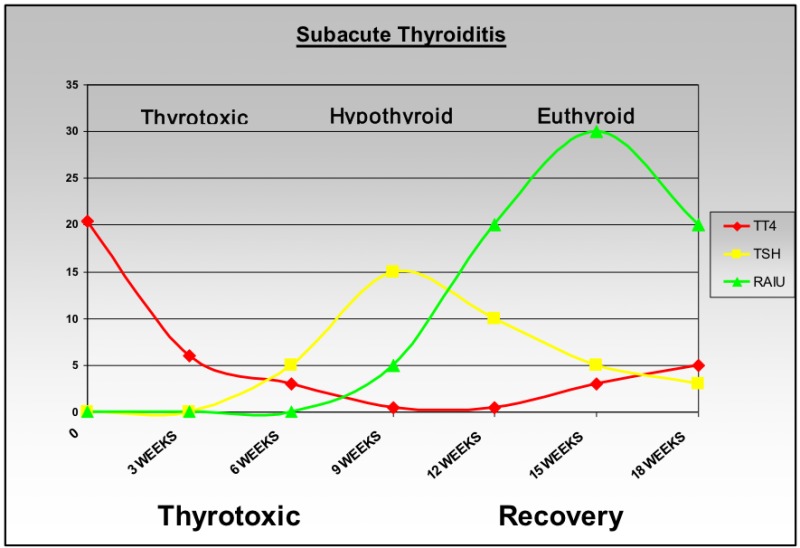









 Management
Management