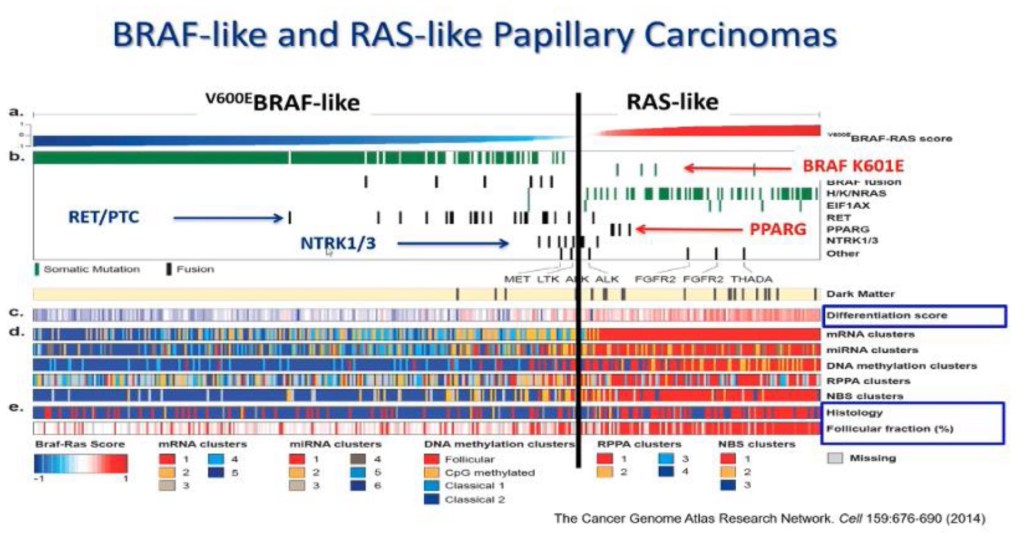
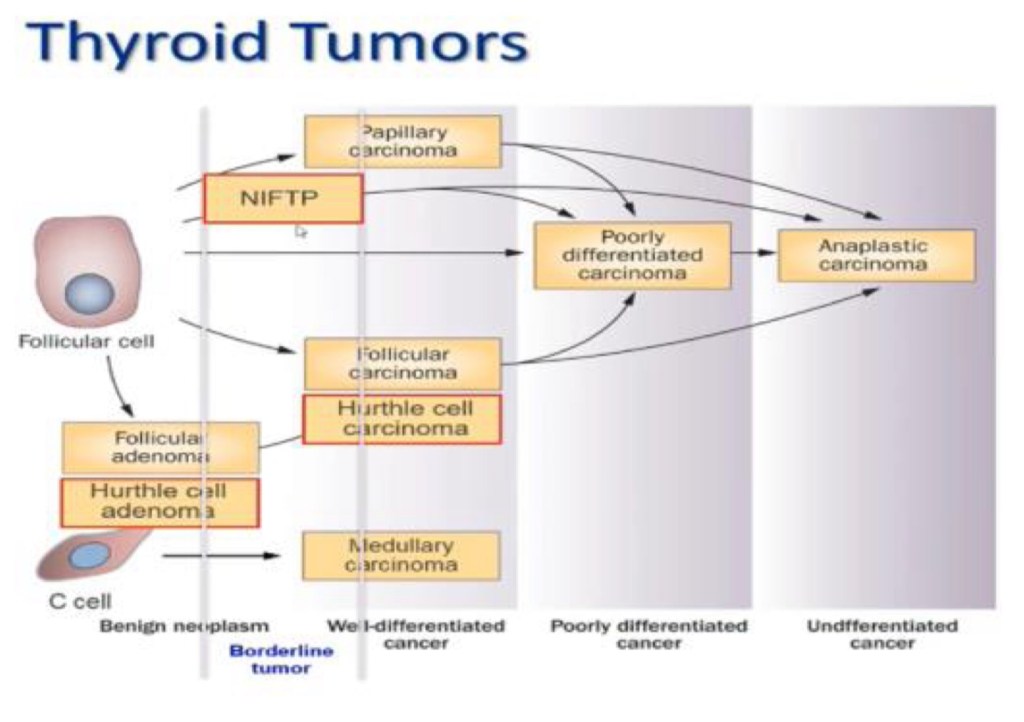
Medullary Thyroid Cancer (MTC) accounts for 1% to 2% of thyroid cancers in the United States.
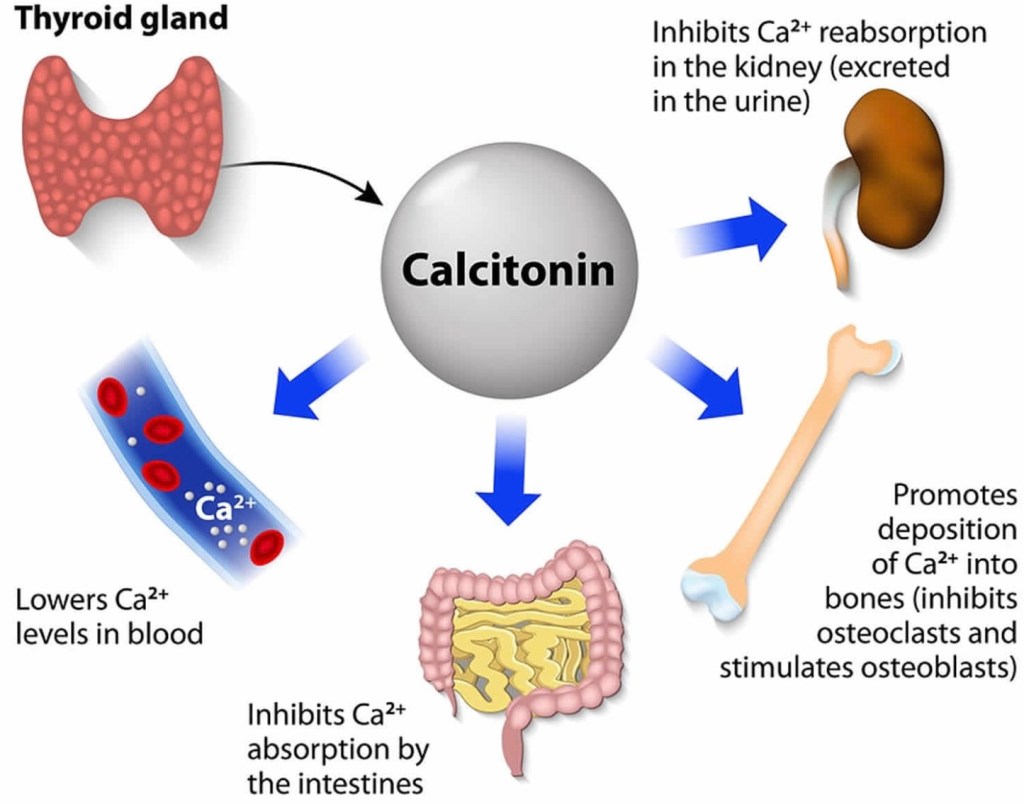
- MTC is different from other types of thyroid cancers (which are derived from thyroid follicular cells – the cells that make thyroid hormone), because it originates from the parafollicular C cells (also called “C cells”) of the thyroid gland. These cells do not make thyroid hormone and instead make a different hormone called calcitonin.
MTC can, and frequently does, spread to lymph nodes and can also spread to other organs.
MTC is likely to run in families (inherited forms) in up to 25% of diagnoses, and inherited forms can be associated with other endocrine tumors, in syndromes called Multiple Endocrine Neoplasia (MEN) 2A and MEN 2B.
- In addition to MTC, patients with MEN2A may have tumors of the adrenal glands called pheochromocytomas or in the parathyroid glands (parathyroid adenomas). Patients with MEN2B, have MTC, pheochromocytomas and neuromas (typically a benign growth or tumor of nerve tissue) in the lining of the mouth and/ or gastrointestinal track.
- Patients with an inherited form of MTC usually have a mutation in a gene called the RET proto-oncogene. This mutation is present in all of the cells in their body (a germline mutation) and these mutations cause the development of MTC. This is important because in family members of a person with an inherited form of MTC, a blood test for a mutation in the RET protooncogene can lead to an early diagnosis of MTC and, to curative surgery to remove it. However, in the majority of patients (~ 75%) a germline mutation is not found – indicating that MTC is not an inherited or inheritable condition. In these cases, MTC is called sporadic.
Whether MTC is sporadic or familial can be determined by a blood test for the RET protooncogene. Anyone diagnosed with MTC should have this test run to determine whether the MTC is familial (meaning other family members may also have MTC that has not yet been diagnosed) or sporadic.
What are the Symptoms of Medullary Thyroid Cancer?
Medullary thyroid cancer usually presents as a lump or nodule in the thyroid. It may be noted by the patient or discovered during routine neck examination by the doctor. Sometimes, the nodule is discovered incidentally by imaging studies done for other unrelated reasons (CT of the neck, PET scan, or carotid ultrasound). The nodule may cause no symptoms, but in some cases the tumor may have spread to lymph nodes in the neck, which may be enlarged on physical examination.
Patients with advanced MTC may complain of pain in the neck, jaw, or ear. If a nodule is large enough to compress the windpipe or the esophagus, it may cause difficulty with breathing or swallowing. Hoarseness can be present if the cancer invades the nerve that controls the vocal cords.
MTC is usually more aggressive than the other more common types of thyroid cancer, and it is usually easier to treat and control if it is found before it spreads to lymph nodes in the neck or other parts of the body.
Thyroid function tests such as TSH are usually normal, even when MTC is present.
If you have a family history of MTC and have tested positive for the RET mutation, then you should see an endocrinologist to help determine how best to follow you or treat you.
How is Medullary Thyroid Cancer Diagnosed?
A diagnosis of thyroid cancer is usually made by a fine needle aspiration (FNA) biopsy of a thyroid nodule, or after the nodule is surgically removed. Patients in whom the results of an FNA biopsy (or histopathology) are suggestive or indicative of MTC should be further evaluated with measurement of the proteins calcitonin and carcinoembryonic antigen (CEA) in the blood, which are typically elevated in patients with MTC. These tests are useful to confirm the diagnosis of MTC which can help ensure the surgeon plans the correct surgery, and also serve as tumor markers during long-term follow-up to detect any remaining disease or recurrence of the cancer.
What is a RET Mutation?
The RET proto-oncogene is located on chromosome 10. A genetic mutation in the RET oncogene is seen in all cells in the body in patients with the hereditary forms of MTC. Mutations in RET can also be seen only in the tumor cells in patients with sporadic MTC. Since the discovery of the RET oncogene, more than 100 different mutations have been identified in the gene in patients with MTC.
Genetic counseling and testing for RET gene mutations should be offered to patients diagnosed with MTC and first-degree relatives (parents, siblings and children of someone diagnosed with MTC) of all patients with proven germline mutations (hereditary MTC). If close relatives, especially children, are found to have the RET mutation on a blood test, the thyroid gland can be removed before MTC has a chance to develop or at least in its very early stages.
How is Medullary Thyroid Cancer Treated?
The primary treatment for MTC is surgery, and the currently accepted approach is to remove the entire thyroid gland (total thyroidectomy) (See thyroid surgery brochure). Often patients with MTC will have thyroid cancer present in the lymph nodes of the neck or upper chest. These lymph nodes are usually removed at the time of thyroid surgery or sometimes, at a later surgery if found subsequently. After surgery, patients need to take thyroid hormone replacement medication for life.
Unlike papillary and follicular thyroid cancer, medullary thyroid cancer does not take up iodine, and consequently radioactive iodine treatment is not a treatment option for patients with MTC.
Patients with MTC with very high levels of calcitonin should have imaging prior to surgery to determine whether the tumor has spread to sites outside the thyroid and/or outside the neck. If there is evidence of cancer outside the neck, surgery may be more palliative, aimed at reducing local complications caused by the tumor, rather than completely eliminating all tumor. Other treatment options (external beam radiation, or chemotherapy) may need to be used together with surgery after careful discussion with the patient.
New chemotherapeutic agents that have shown promise treating other advanced cancers are increasingly available for treatment of thyroid cancers. Two such agents, Vandetanib and Cabozantinib have been FDA approved for use by patients with MTC. These drugs do not cure advanced cancers that have spread widely throughout the body, but they can often slow down or partially reverse the growth of the cancer. These treatments are usually given by an oncologist (cancer specialist) and require care at specialized medical centers.
What is the Follow-Up of Patients with Medullary Thyroid Carcinoma?
Periodic follow-up examinations are essential for all patients with MTC because the thyroid cancer can return, sometimes many years after successful initial treatment. These follow-up visits include a careful history and physical examination, with particular attention to the neck area. Neck ultrasound is also a very important tool to visualize the neck and look for nodules, lumps or enlarged lymph nodes that might indicate that the cancer has recurred.
Blood tests are also important in the follow-up of MTC patients. All patients who have had their thyroid glands removed require thyroid hormone replacement with levothyroxine. Thyroid stimulating hormone (TSH) should be checked periodically, and the dose of levothyroxine adjusted to keep TSH in the normal range. There is no need to keep TSH suppressed in patients with MTC.
Measurement of calcitonin and CEA are a necessary routine part of the follow-up of patients with MTC. Following thyroidectomy, it is hoped that calcitonin levels will be essentially undetectable for life. A detectable or rising calcitonin level should raise suspicion for possible cancer recurrence. Detectable calcitonin levels may require additional tests.
Rodrigo Arrangoiz MS, MD, FACS
-
He is an expert in the management medullary thyroid carcinoma.
-
Articles published on MTC:
-
https://www.gavinpublishers.com/articles/Review-Article/Journal-of-Clinical-Endocrinology-and-Diabetes/Medullary-Thyroid-Carcinoma-Literature-Review-and-Current-Management
-
http://www.remedypublications.com/american-journal-of-otolaryngology-and-head-and-neck-surgery/articles/pdfs_folder/ajohns-v1-id1026.pdf
-