- Pendrin, a transporter protein:
- Is crucial for iodide efflux in thyroid follicular cells:
- Playing a key role in thyroid hormone synthesis
- It’s specifically located on the apical membrane of the thyroid follicular cells:
- Where it mediates the movement of iodide:
- From inside the cell to the colloid space within the follicle
- Where it mediates the movement of iodide:
- Is crucial for iodide efflux in thyroid follicular cells:
- Function:
- Pendrin acts as a transporter:
- Facilitating the movement of iodide and chloride ions across the cell membrane
- In the thyroid, it specifically transports iodide from the cytoplasm of thyroid follicular cells into the follicular lumen (colloid)
- Pendrin acts as a transporter:
- Location:
- Pendrin is located on the apical membrane of thyroid follicular cells:
- Which is the membrane facing the colloid within the thyroid follicle.
- Pendrin is located on the apical membrane of thyroid follicular cells:
- Significance:
- This transport is essential for the synthesis of thyroid hormones (T4 and T3)
- Iodide is first transported into the cell by the:
- Sodium-iodide symporter (NIS:
- Which is located on the basolateral membrane of the thyroid follicular cell
- Then, pendrin facilitates the efflux of iodide into the colloid:
- Where it can be used in the synthesis of thyroglobulin (TG)
- The precursor to thyroid hormones
- Where it can be used in the synthesis of thyroglobulin (TG)
- Sodium-iodide symporter (NIS:
- Pendred Syndrome:
- Mutations in the PDS gene, which codes for pendrin:
- Can lead to Pendred syndrome:
- A condition characterized by goiter (enlarged thyroid), and hearing loss:
- Due to impaired iodide transport in the thyroid and inner ear
- A condition characterized by goiter (enlarged thyroid), and hearing loss:
- Can lead to Pendred syndrome:
- Mutations in the PDS gene, which codes for pendrin:
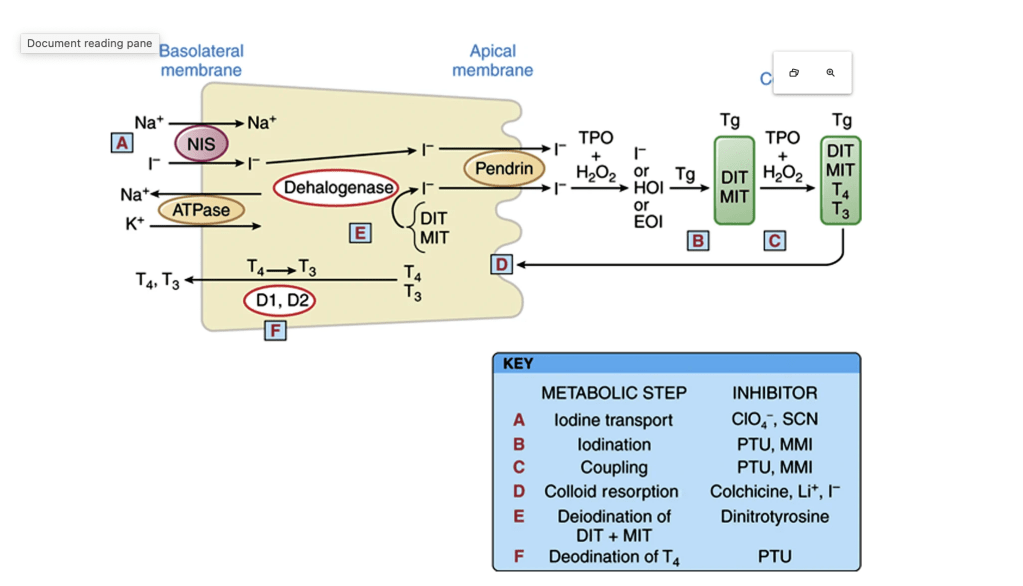
Thyroid hormone synthesis and secretion are activated when Thyroid stimulating hormone (TSH) binds to the TSH receptor on the basolateral membrane. Iodide is transported into the cell via the Na+/I− (NIS) symporter and flows down an electrical gradient, maintained by the sodium-potassium ATPase. Iodide becomes covalently attached to the tyrosyl residues of the precursor thyroid hormone glycoprotein, thyroglobulin, by thyroperoxidase (TPO) to form monoiodotyrosine (MIT) and diiodotyrosine (DIT). These are subsequently coupled by the action of TPO to form the iodothyronine hormones, tetraiodothyronine (T4) and triiodothyronine (T3). In the process of thyroid hormone secretion, Tg enters the cell by pinocytosis, forming colloid droplets. These fuse with lysosomes, forming phagolysosomes in which Tg is broken down by proteolysis, and then T4 and T3 are released and diffuse into circulation. MIT and DIT are formed by the iodination of tyrosyl amino acids on the thyroglobulin molecule. In a subsequent step, two DITs are coupled to form T4, or one DIT and one MIT are coupled to form T3. (From Brent GA, Koenig RJ. Thyroid and antithyroid drugs. In: Brunton L [ed]. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 13th ed. New York: McGraw-Hill; 2017.)