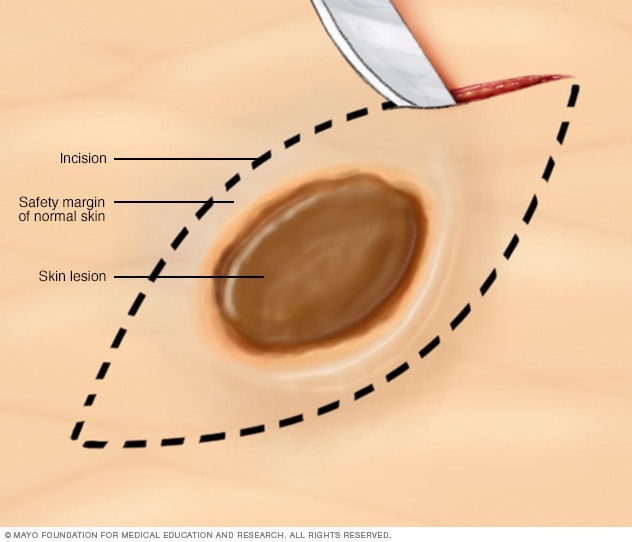
Boost radiation therapy is an additional dose of radiation delivered to the tumor bed after whole-breast irradiation (WBI) in patients who undergo breast-conserving surgery (BCS). The goal is to reduce local recurrence rates. The decision to give a boost is typically based on patient-specific risk factors.
Indications for Boost Radiation after BCS
1. Age < 50 years (especially < 40 years)
Data: The EORTC 22881–10882 trial (Bartelink et al., 2001; 2007 update) Population: 5,318 women with stage I/II breast cancer who received BCS + WBI (50 Gy) ± boost (16 Gy). Results: 10-year local recurrence (LR): Without boost: 10.2% With boost: 6.2% (Absolute reduction: 4%, p < 0.0001) Age stratification: < 40 years: LR reduced from 24% to 14% with boost (10% absolute benefit) 41–50 years: LR reduced from 11.7% to 6.4% (5.3% absolute benefit) 50 years: Less pronounced benefit
2. Positive or Close Surgical Margins
Rationale: Tumor cells may remain near the excision cavity, increasing recurrence risk. Guideline Support: NCCN recommends a boost for positive margins (even if re-excised) or if final margins are close, particularly in young patients.
3. High-Grade Tumors (Grade 3)
Data: High-grade tumors are more biologically aggressive and associated with higher LR rates. Boost helps reduce recurrence in these patients, especially when combined with other risk factors (young age, close margins).
4. Lymphovascular Invasion (LVI)
LVI is a marker for higher local and regional recurrence risk. While not an absolute indication, it adds weight to the decision in a patient with other risk factors.
5. Extensive Intraductal Component (EIC)
Especially in younger patients, an EIC increases the risk of residual disease and LR.
6. Triple-negative or HER2-positive subtypes (in younger patients)
While molecular subtype alone isn’t a formal indication for boost, aggressive biology may influence the decision when combined with other features (e.g., young age, LVI, close margins).
Guidelines
ASTRO 2016: Endorses boost in patients <50 years or with high-risk features. NCCN 2024: Recommends consideration of boost based on age, margin status, histology, grade, and LVI.
Clinical Takeaway
Boost radiation significantly reduces local recurrence in high-risk patients following breast-conserving surgery. The greatest benefit is seen in younger women and those with adverse pathological features.
Let me know if you’d like a one-slide summary for a presentation.