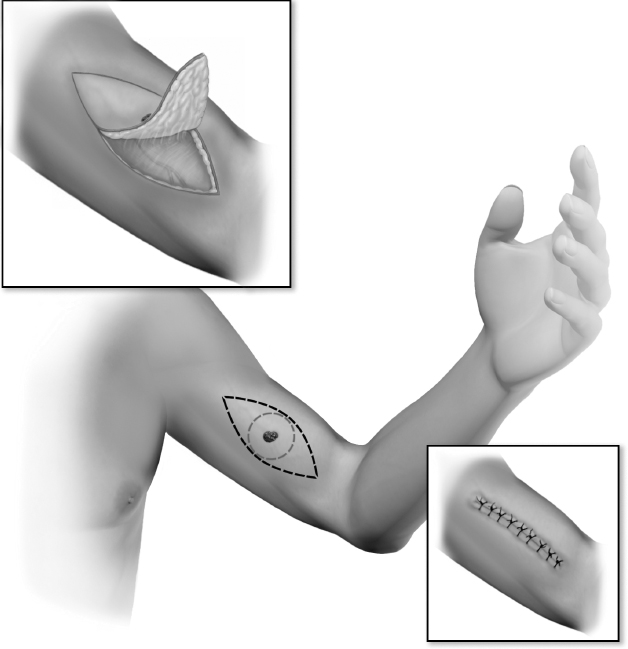
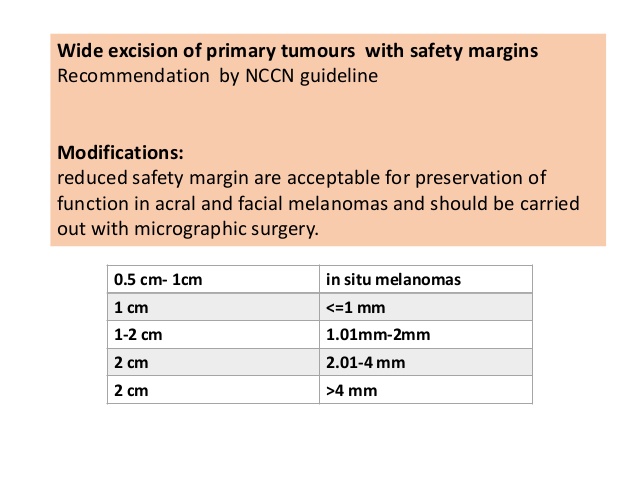
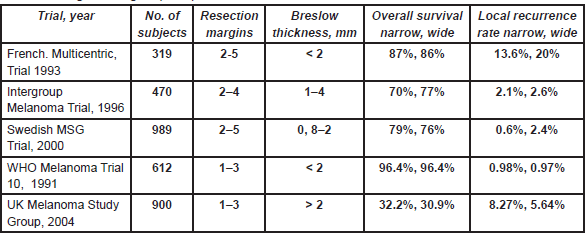
- The most recent updates on head and neck cancer presented at the American Society of Clinical Oncology (ASCO) 2025 conference:
- Continue to emphasize the evolving role of immunotherapy:
- Particularly immune checkpoint inhibitors (ICIs):
- In both recurrent / metastatic and curative-intent settings
- Particularly immune checkpoint inhibitors (ICIs):
- The American Society of Clinical Oncology has published guidelines recommending biomarker-driven use of ICIs:
- Such as pembrolizumab or nivolumab:
- For recurrent or metastatic head and neck squamous cell carcinoma (HNSCC):
- With programmed death ligand-1 (PD-L1) expression guiding first-line therapy selection
- For recurrent or metastatic head and neck squamous cell carcinoma (HNSCC):
- These guidelines also address the use of immunotherapy in platinum-refractory disease:
- They highlight the importance of biomarker testing to optimize patient selection
- Such as pembrolizumab or nivolumab:
- Continue to emphasize the evolving role of immunotherapy:
- Recent clinical trial data discussed at ASCO and summarized in the literature show that while ICIs have improved outcomes for a subset of patients with recurrent / metastatic HNSCC:
- Durable responses remain limited:
- To approximately 15% to 20% of patients
- Durable responses remain limited:
- Efforts to expand the benefit of immunotherapy into the definitive (curative) setting:
- Such as concurrent or sequential use with chemoradiation or as neoadjuvant / adjuvant therapy:
- Have yielded mixed results:
- With ongoing trials investigating optimal timing and combinations
- Have yielded mixed results:
- Such as concurrent or sequential use with chemoradiation or as neoadjuvant / adjuvant therapy:
- The American Head and Neck Society:
- Notes that neoadjuvant immunotherapy, particularly anti-PD-1 agents:
- Is under active investigation, with early-phase trials showing promising signals but no definitive change in standard of care yet
- Notes that neoadjuvant immunotherapy, particularly anti-PD-1 agents:
- For HPV-associated oropharyngeal cancer:
- De-intensification strategies and novel immunotherapeutic approaches, including therapeutic vaccines and adoptive cell therapies, are being explored, but these remain investigational and are not yet standard practice
- In summary, the ASCO 2025 updates reinforce the central role of immunotherapy in recurrent /metastatic HNSCC, the need for biomarker-driven treatment selection, and the ongoing investigation of immunotherapy in earlier disease settings, as recommended by the American Society of Clinical Oncology and highlighted by the American Head and Neck Society
- References:
- Immunotherapy and Biomarker Testing in Recurrent and Metastatic Head and Neck Cancers: ASCO Guideline. Yilmaz E, Ismaila N, Bauman JE, et al. Journal of Clinical Oncology : Official Journal of the American Society of Clinical Oncology. 2023;41(5):1132-1146. doi:10.1200/JCO.22.02328.
- Current Progress and Future Directions of Immunotherapy in Head and Neck Squamous Cell Carcinoma: A Narrative Review. Sim ES, Nguyen HCB, Hanna GJ, Uppaluri R. JAMA Otolaryngology– Head & Neck Surgery. 2025;151(5):521-528. doi:10.1001/jamaoto.2024.5254.
- Neoadjuvant Therapy for Mucosal Head and Neck Squamous Cell Carcinoma: A Review From the American Head and Neck Society. Contrera KJ, Kansara S, Goyal N, et al. JAMA Otolaryngology– Head & Neck Surgery. 2025;:2833345. doi:10.1001/jamaoto.2025.0410.
- Update: Immunotherapeutic Strategies in HPV-Associated Head and Neck Squamous Cell Carcinoma. Sun F, Colevas AD. Viruses. 2025;17(5):712. doi:10.3390/v17050712.