Atypia of Undetermined Significance (AUS) or Follicular Lesion of Undetermined Significance (FLUS) on Cytology
- Based on the Bethesda system:
- This diagnostic category is reserved for specimens that contain cells with architectural and / or nuclear atypia that are more prominent than expected for benign changes, but not sufficient to be placed in one of the highest-risk diagnostic categories
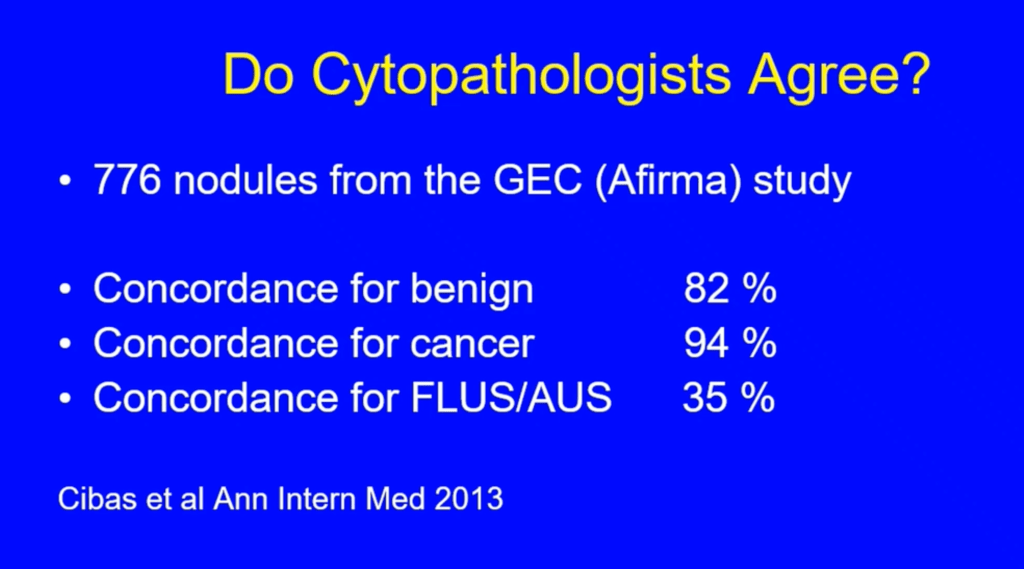
- In the studies that used the criteria established by the Bethesda system, the risk of cancer for patients with AUS / FLUS who underwent surgery was:
- 6% to 18% if NIFT (non-invasive follicular thyroid neoplasia with papillary nuclear characteristics) it is not considered cancer
- 10% to 30% if NIFT is considered a cancer
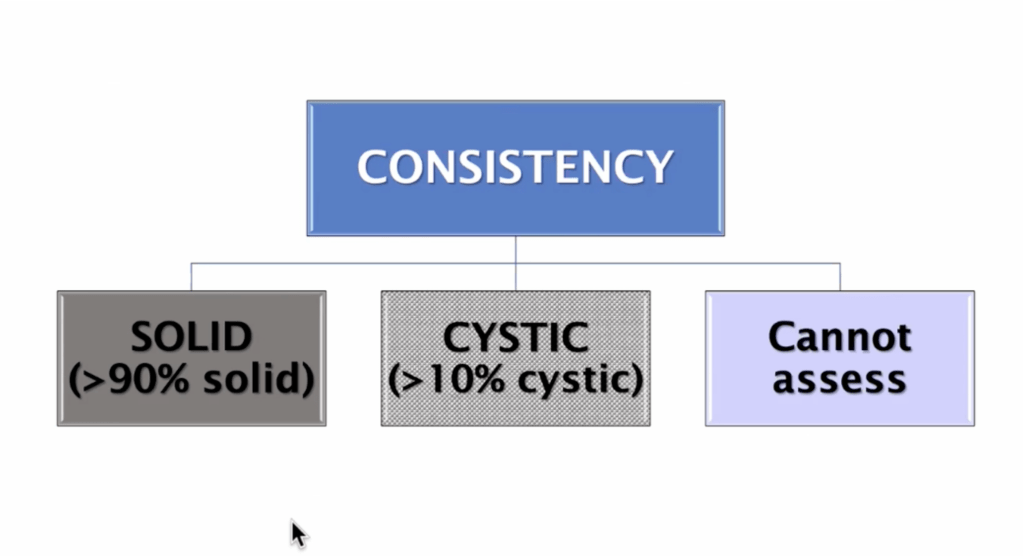
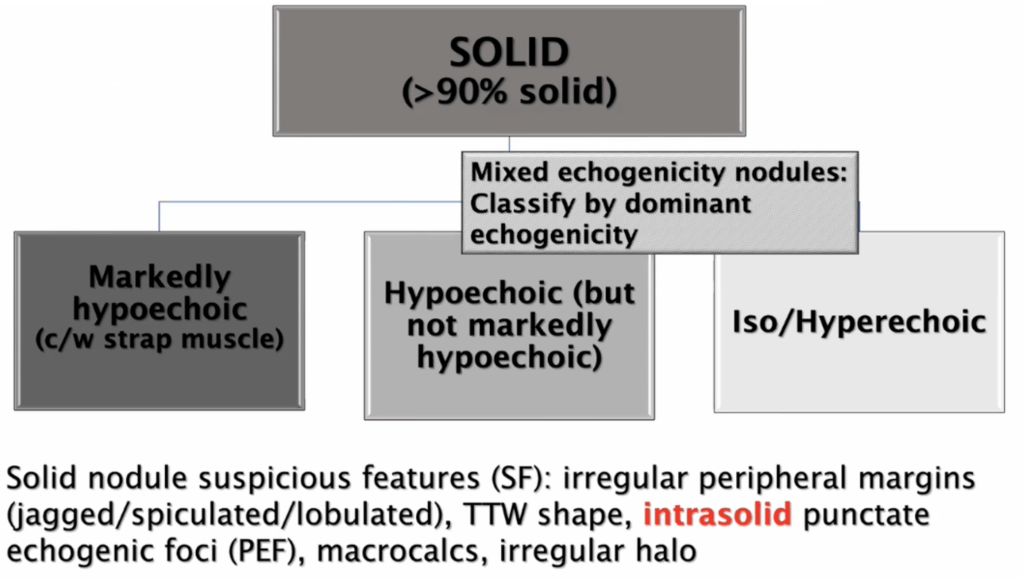
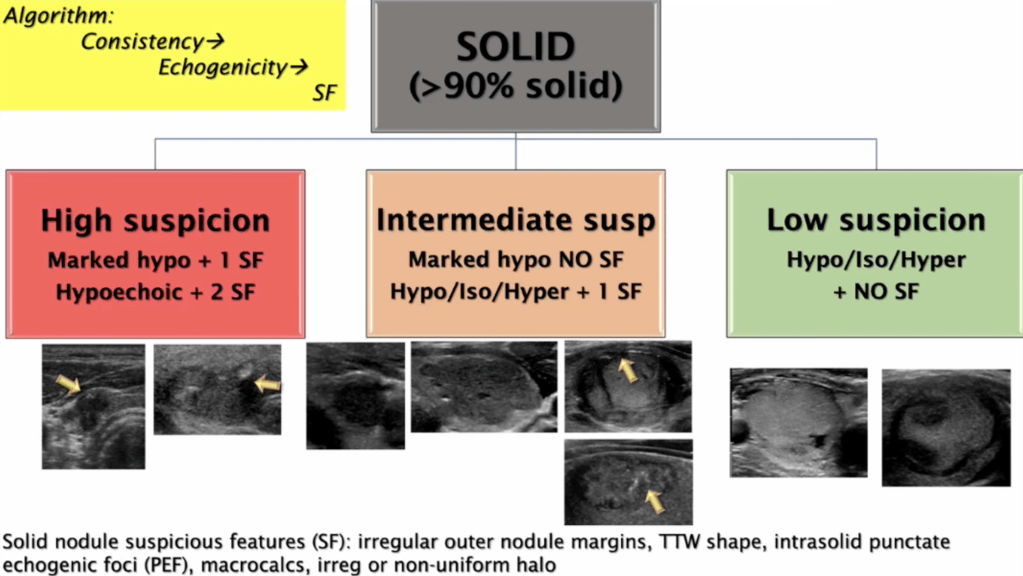
- For thyroid nodules with AUS / FLUS cytology after a FNAB, with clinical and ultrasonographic features of concern:
- The assessment can be continued by repeating the FNAB or if the technology is available, molecular tests can be used to complement the risk assessment of malignancy instead of preceding directly with a strategy of either surveillance or diagnostic surgery (lobectomy)
- Patient preference should be considered in decision-making (recommendation 15 of the ATA)
- If FNAB is not repeated, and molecular tests are not performed, or both studies were inconclusive:
- A diagnostic surgical excision may be performed for the thyroid nodules with Bethesda AUS / FLUS classification, according to the clinical risk factors, the ultrasonographic pattern, and patient preference (recommendation 15 of the ATA)