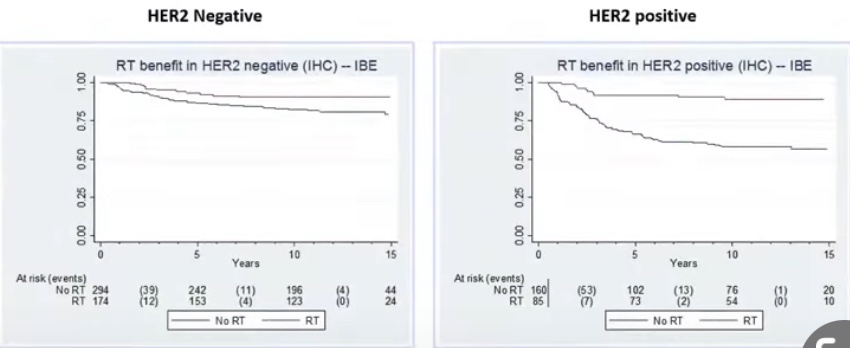
- The National Surgical Adjuvant Breast and Bowel Project (NSABP) B-17 phase III randomized clinical trial and three other prospective randomized clinical trials and their follow-up studies:
- Have demonstrated and continue to show the benefit of radiation therapy (RT) following breast-conserving surgery for patients with DCIS:
- With a significant 50% reduction in IBTR
- Have demonstrated and continue to show the benefit of radiation therapy (RT) following breast-conserving surgery for patients with DCIS:
- In a report by Wapnir et al. on long-term outcomes of the NSABP B-17 and B-24 trials:
- Patients who only received lumpectomy:
- Had a local recurrence rate of:
- 19.4% compared with 8.9% in the lumpectomy plus RT group at 15 years of follow-up:
- Demonstrating a 52% reduction in the risk of IBTR
- 19.4% compared with 8.9% in the lumpectomy plus RT group at 15 years of follow-up:
- Had a local recurrence rate of:
- Patients who only received lumpectomy:
- Similarly, European Organization for Research and Treatment of Cancer (EORTC) 10853:
- Showed a risk reduction of 48% with the addition of radiotherapy to locally excised DCIS
- The Radiation Therapy Oncology Group (RTOG) 9804 trial:
- Evaluated the impact of RT after breast-conserving therapy (BCT) in patients with mammographically detected low-risk DCIS as defined by:
- Size less than 2.5 cm
- Unicentric
- Low- and intermediate-grade
- Margins > 3 mm
- The primary endpoint of ipsilateral local failure was:
- 0.9% in the RT arm versus 6.7% in the observation arm
- Evaluated the impact of RT after breast-conserving therapy (BCT) in patients with mammographically detected low-risk DCIS as defined by:
- The E5194 trial:
- Included a similar patient population of 665 patients placed into two cohorts:
- Low- and intermediate-grade DCIS with a tumor size less than 2.5 cm or high-grade DCIS with a tumor size less than 1 cm
- All patients were treated with BCT yielding greater than 3 mm margins without RT with a median follow-up of 12.3 years
- These results demonstrated tumor size and cohort to be significant predictors of developing an ipsilateral breast event:
- 14.4% for the low-risk cohort versus 24.6% for the high-risk cohort
- This risk continued to increase without plateau through the follow-up period, necessitating further follow-up to determine long-term outcomes
- Included a similar patient population of 665 patients placed into two cohorts:
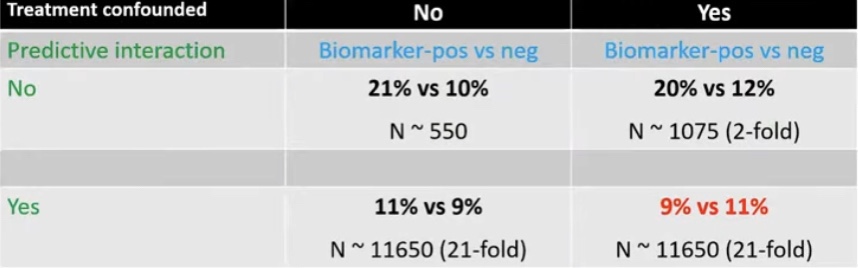
- Differences in the IBTR rates between these studies:
- May be due to the effect of tamoxifen use:
- Which was not controlled in either trial
- Which is known to reduce the risk of IBTR in combination with RT after BCT as shown by the NSABP B-24 trial
- May be due to the effect of tamoxifen use:
- Together, the results of both RTOG 9804 and E5194:
- Support the individualized treatment of DCIS:
- Based on identification of patients at low risk for IBTR determined by:
- Pathologic and clinical features that may omit RT
- Based on identification of patients at low risk for IBTR determined by:
- Support the individualized treatment of DCIS:
- A 12-year follow-up study of RTOG 9804 demonstrated:
- That the 12-year cumulative incidence of local recurrence (LR) was:
- 2.8% with RT and 11.4% with observation alone
- The 12-year cumulative incidence of invasive LR was:
- 1.5% with RT and 5.8% with observation alone
- That the 12-year cumulative incidence of local recurrence (LR) was:
- References:
- Wapnir IL, Dignam JJ, Fisher B, Mamounas EP, Anderson SJ, Julian TB, et al. Long-term outcomes of invasive ipsilateral breast tumor recurrences after lumpectomy in NSABP B-17 and B-24 randomized clinical trials for DCIS. J Natl Cancer Inst. 2011;103(6):478-488.
- EORTC Breast Cancer Cooperative Group; and EORTC Radiotherapy Group; Bijker N, Meijnen P, Peterse JL, et al. Breast-conserving treatment with or without radiotherapy in ductal carcinoma-in-situ: ten-year results of European Organisation for Research and Treatment of Cancer randomized phase III trial 10853—a study by the EORTC Breast Cancer Cooperative Group and EORTC Radiotherapy Group. J Clin Oncol. 2006;24(21):3381-3387.
- McCormick B, Winter K, Hudis C, Kuerer HM, Rakovitch E, Smith BL, et al. RTOG 9804: a prospective randomized trial for good-risk ductal carcinoma in situ comparing radiotherapy with observation. J Clin Oncol. 2015;33(7):709-715.
- Solin LJ, Gray R, Hughes L, Wood WC, Lowen MA, Badve SS, et al. Surgical excision without radiation for ductal carcinoma in situ of the breast: 12-year results from the ECOG-ACRIN E5194 Study. J Clin Oncol. 2015;33(33):3938-3944.
- 5. McCormick B. Randomized trial evaluating radiation following surgical excision for “good risk” DCIS: 12-year report from NRG/RTOG 9804. Int J Radiat Oncol Biol Phys. 2018;102(5):1603.

#Arrangoiz #BreastSurgeon #CancerSurgeon #SurgicalOncologist #BreastCancer #DCIS #RadiationTherapy #Miami #Mexico #Teacher