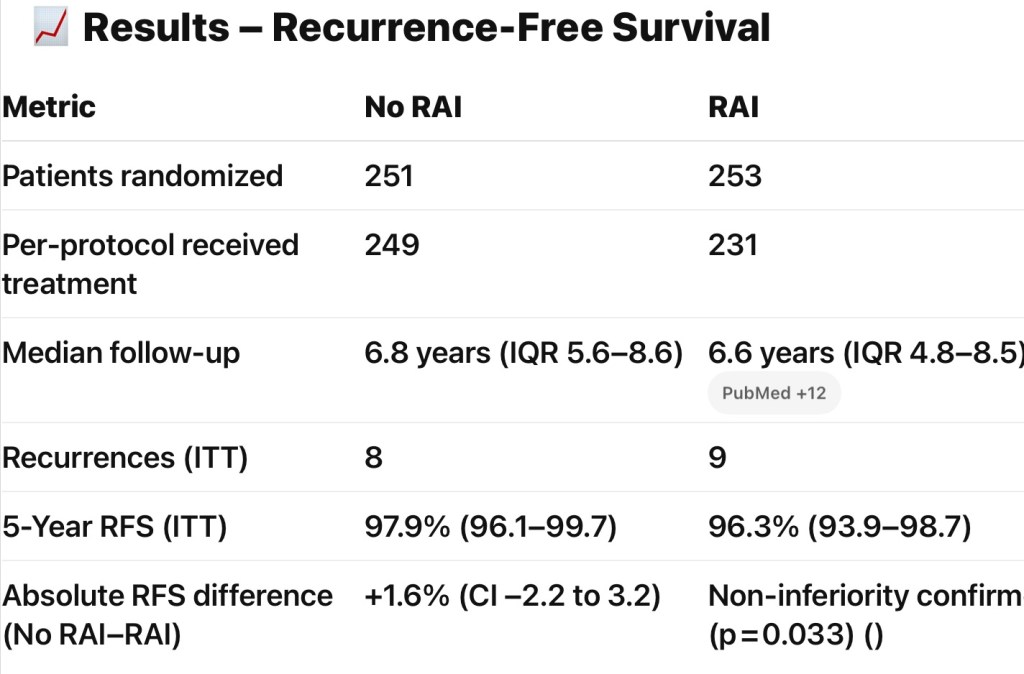
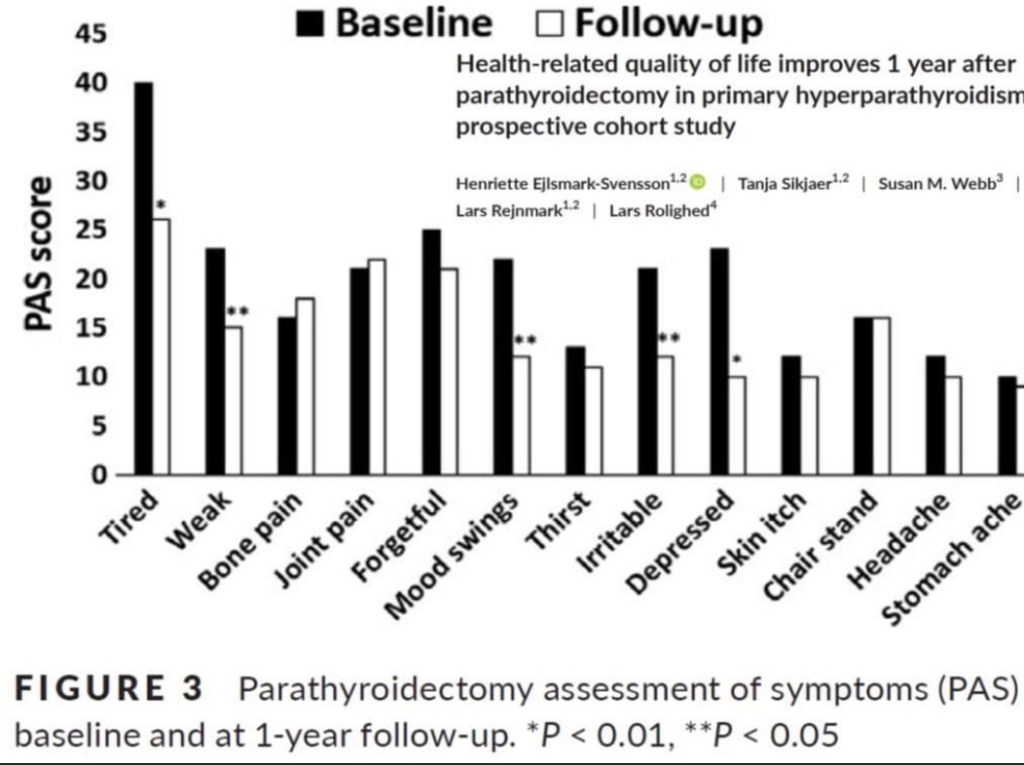
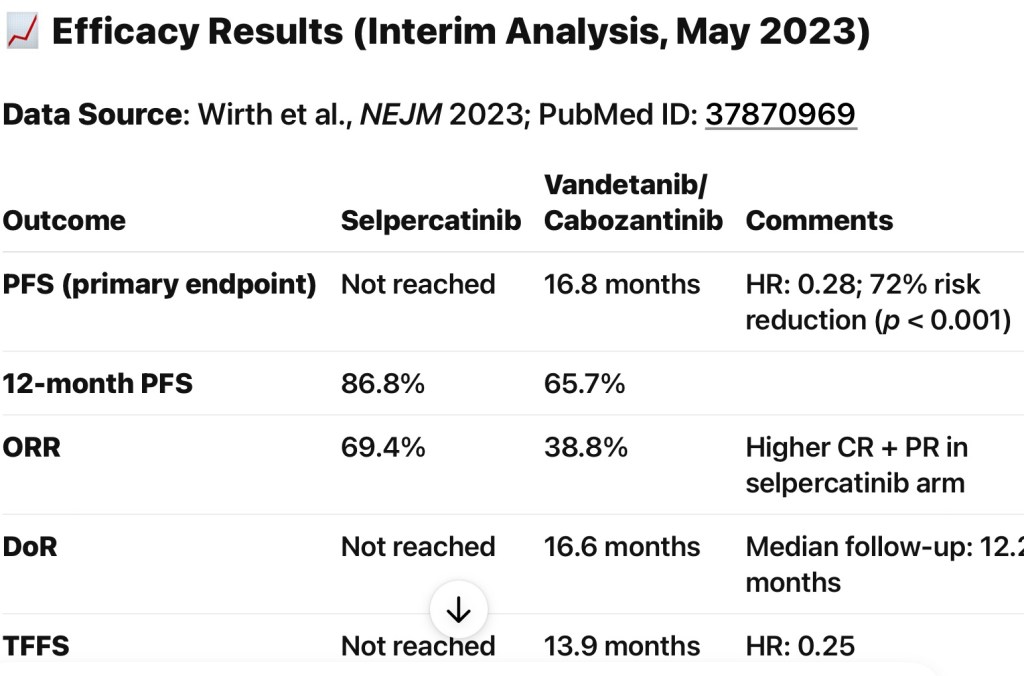
- Postoperative radioactive iodine (RAI) use in ATA intermediate-risk differentiated thyroid cancer (DTC):
- With stratification into low‑intermediate and high‑intermediate subsets
- Evidence Base Overview:
- No randomized trials specifically focus on RAI impact in ATA intermediate-risk patients
- Evidence is primarily retrospective:
- Using multivariate, propensity-adjusted analyses, registry data (SEER), cohort series, and a few prospective studies
- ATA Intermediate‑Risk Definition:
- The 2015 ATA guidelines (1) classified patients as intermediate-risk if they exhibit:
- Microscopic extrathyroidal extension (ETE)
- Vascular invasion
- Clinical N1 or > 5 pathologic N1 with nodes < 3 cm
- Aggressive histotypes (e.g., Hobnail, tall cell, insular)
- RAI-avid foci outside thyroid bed
- Multifocal microcarcinoma with ETE and BRAF mutation
- The 2015 ATA guidelines (1) classified patients as intermediate-risk if they exhibit:
- Within this, it’s helpful to subdivide into (most likely will appear in the new 2025 ATA guidelines):
- Low-intermediate:
- A single low-volume risk feature:
- Microscopic ETE
- Small-volume N1a
- A single low-volume risk feature:
- High-intermediate:
- Multiple or higher-risk features, such as:
- ≥ 5 nodes
- Vascular invasion
- Multiple or higher-risk features, such as:
- Low-intermediate:
- Observational and Registry Data:
- SEER Registry Studies (2,3):
- Showed improved overall survival following RAI in:
- N1 disease
- pT3 (or > 4 cm)
- Aggressive histology
- However:
- Absolute benefit is small in patients < 45 years (~ 1%) vs larger in older patients (~ 4%) (3)
- Showed improved overall survival following RAI in:
- Single‑Center Cohorts:
- Mayo Clinic (4) (20‑year follow-up):
- Found no impact on outcomes for node-positive with MACIS < 6
- One Hong Kong study (5):
- Showed better lymph node recurrence-free survival post-RAI in:
- N1b or nodes > 1 cm
- Showed better lymph node recurrence-free survival post-RAI in:
- Mayo Clinic (4) (20‑year follow-up):
- SEER Registry Studies (2,3):
- Retrospective Low–Intermediate Series (6) (Italy, 2024):
- RAI reduced recurrence by 42%:
- 9.6% vs 15.9% using inverse‑probability regression in patients with ≥ 2 intermediate risk factors
- RAI reduced recurrence by 42%:
- Polish Prospective RAI Dose Trial (Arch Med Sci, 2022): (7)
- Intermediate-risk group receiving 60 vs 100 mCi:
- Had similar excellent response (~ 85%), low structural recurrence (~ 6.5%), and excellent long-term outcomes
- Intermediate-risk group receiving 60 vs 100 mCi:
- Prospective / Clinical Trials:
- No RCTs isolating intermediate-risk exist:
- However, the rhTSH + RAI Prep trial included many intermediate-risk patients (n ≈ 307) and found non-inferiority of rhTSH vs withdrawal for RAI effectiveness (8)
- No RCTs isolating intermediate-risk exist:
- Guideline Position Statements:
- The 2015 ATA guidelines recommend selective use of RAI in intermediate-risk DTC based on individual risk features, with a weak recommendation and low-quality evidence (1)
- The 2022 European Thyroid Association / EANM consensus also supports personalized RAI use in this group, prioritizing shared decision-making and individual risk-benefit assessment (9)
- Clinical Take‑Home Points:
- Selective RAI yields a survival / recurrence benefit:
- Strongest in older patients or those with > 1 intermediate risk feature
- Low-intermediate risk (e.g., single small node, microscopic ETE):
- RAI benefit is less definitive:
- Consider active surveillance vs low-dose RAI
- High‑intermediate risk (multi-node, vascular invasion, aggressive histology):
- More likely to benefit:
- RAI recommended:
- Dose considerations:
- 60 mCi sufficient for intermediate risk (Polish RCT)
- rhTSH prep is validated
- Shared decision-making essential, given evidence limitations and QoL trade-offs
- More likely to benefit:
- RAI benefit is less definitive:
- Selective RAI yields a survival / recurrence benefit:
- Recommendation Framework:
- Low‑intermediate (one minor feature, younger age):
- Discuss omission vs low-dose RAI (e.g., 60 mCi + rhTSH) with surveillance plan
- High‑intermediate (multi-factor, older age):
- Offer RAI routinely; evidence supports 60 to 100 mCi
- Shared consent vital, addressing patient goals, comorbidities, and center’s surveillance capabilities
- Low‑intermediate (one minor feature, younger age):
- References:
- 1. Haugen BR, et al. 2015 ATA Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26(1):1–133.
- 2. Nixon IJ, et al. The impact of RAI on survival in intermediate-risk patients: SEER analysis. Ann Surg Oncol. 2012;19(6):2025–33.
- 3, Haymart MR, et al. Radioactive iodine in thyroid cancer: SEER-based outcomes. JAMA. 2011;306(7):721–8.
- 4. Mazzaferri EL, et al. Management of differentiated thyroid cancer: Mayo Clinic experience. J Clin Endocrinol Metab. 2001;86(4):1447–63.
- 5. Lam AK, et al. RAI impact on lymph node recurrence: Hong Kong cohort study. Cancer. 2005;103(5):920–9.
- 6. Prete A, et al. Benefit of RAI in intermediate-risk DTC patients with multiple features. Endocrine. 2024;84(1):123–131.
- 7. Piciu D, et al. Prospective evaluation of 60 vs 100 mCi in intermediate-risk thyroid cancer. Arch Med Sci. 2022;18(4):1002–1012.
- 8. Mallick U, et al. Preparation for RAI: rhTSH vs withdrawal in low- and intermediate-risk patients. Lancet. 2012;379(9825):823–830.
- 9. Luster M, et al. EANM/ETA consensus on RAI therapy in thyroid cancer. Eur J Nucl Med Mol Imaging. 2022;49(1):13–25.
- 1. Haugen BR, et al. 2015 ATA Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26(1):1–133.