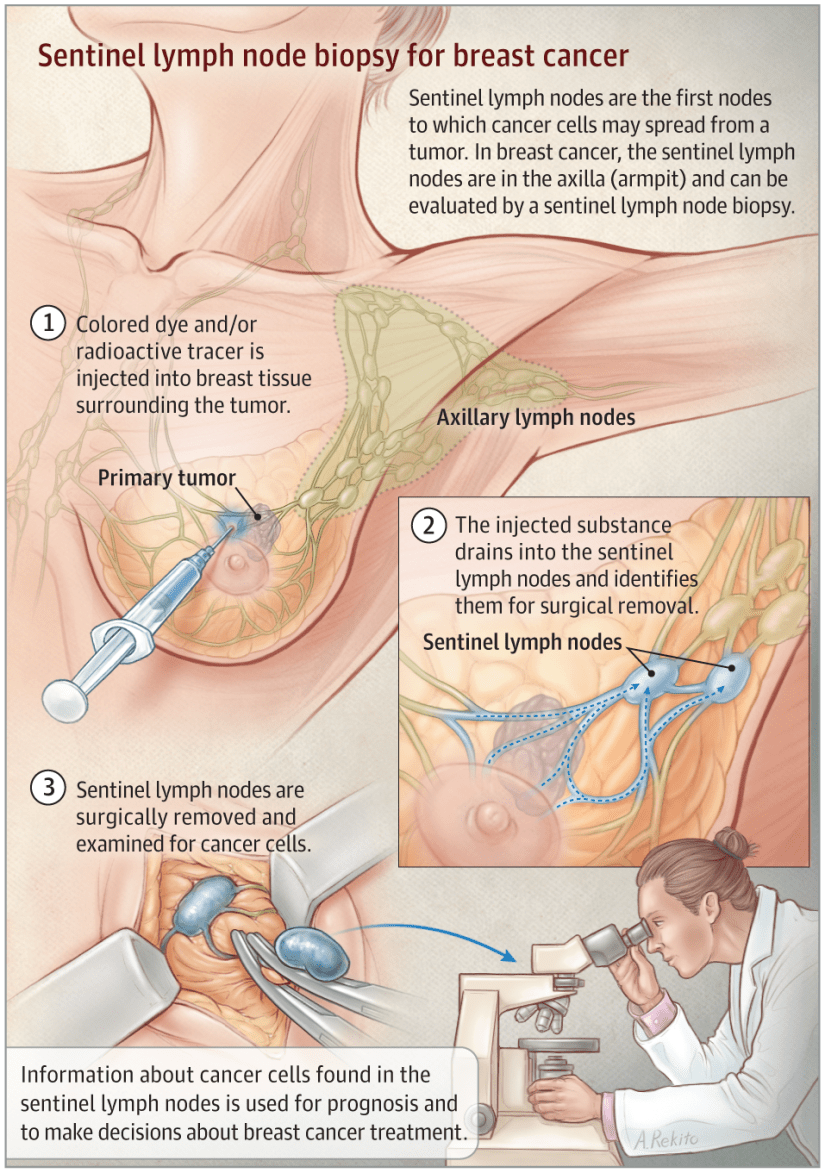
Following neoadjuvant chemotherapy for breast cancer:
- Pathologic complete response (pCR) in the axillary nodes:
- Is found in 40% of patients presenting with biopsy proven node-positive disease.
- This observation led to several clinical trials including:
- American College of Surgeons Oncology Group (ACOSOG Z1071)
- SENTINA (Arm C)
- SN FNAC
- This trials evaluated the feasibility and accuracy of sentinel lymph node mapping (SLNM) and sentinel lymph node biopsy (SLNB) dissection as axillary staging after neoadjuvant chemotherapy in clinical N1 disease.
- Although the false-negative rate of SLNB in this setting was above the predetermined threshold, several important factors associated with and impacting the false negative rate were described.
- This included:
- Use of dual agent mapping technique
- Evaluation of at least 3 SLNs
- Immunohistochemistry (IHC) evaluation in addition to H&E staining
- Excision of the clipped (biopsy proven) node.
- This included:
- This observation led to several clinical trials including:
- Is found in 40% of patients presenting with biopsy proven node-positive disease.
- The ACOSOG Z1071 trial evaluated the false-negative rate (FNR) in patients with clinical N1 disease in whom at least 2 SLNs were identified:
- This FNR was 12.6%.
- The FNR was improved to 10.8%:
- In patients in whom both radioisotope and blue dye were used.
- In addition, the FNR in the Z1071 trial varied based on the number of SLNs identified:
- For patients with a single SLN identified,:
- The FNR was 31.5%
- For those with 2 SLNs identified:
- The FNR was 21.1%
- For those with 3 or more SLNs identified:
- The FNR was 9.1%
- For patients with a single SLN identified,:
- Data from the SENTINA trial also showed an improved FNR in patients in whom 3 or more SLNs were identified:
- The trial was a 4-arm multicenter study.
- Arm C was similar to the ACOSOG Z1071 trial in that it included patients who were clinically node-positive and converted to clinically node-negative (non-palpable) after chemotherapy:
- The FNRs were 24.3% when a single SLN was identified
- 18.5% when 2 SLNs were removed
- Less than 10% when 3 or more SLNs were removed
- Similar to the Z1071 trial:
- The FNR was lower (8.6%) for patients who underwent mapping with radioisotope and blue dye.
- The more recently reported SN FNAC study required the use of immunohistochemistry and considered SLN metastases of any size, including isolated tumor cells, to be positive:
- Using this definition,:
- The FNR was 8.4%.
- If isolated tumor cells identified by immunohistochemistry had been considered negative:
- The FNR would have increased to 13.3%.
- Using this definition,:
- The results of these findings led to refined technique termed targeted axillary dissection (TAD):
- This procedure involves SLNB dissection and selective targeted excision of the clipped (biopsy proven) axillary node.
- Response is documented during chemotherapy with ultrasound and clinical exam.
- In selected patients planned for TAD, the clipped node is preoperatively localized with an I-125 radioactive seed or wire.
- Intraoperatively, dual lymphatic mapping for SLN identification is employed and targeted excision of the clipped node is performed.
- Excision is confirmed with specimen radiograph.
- In approximately 23% of cases the clipped node will not be a sentinel node.
- Compared with ALND:
- The false-negative rate of this approach was:
- 10.1% with sentinel node biopsy alone
- 4.2% for excision of the clipped node alone
- 2.0% for TAD.
- The false-negative rate of this approach was:
- In carefully selected patients TAD alone may offer an accurate approach to axillary staging and assessment of residual disease with limited morbidity.
Rodrigo Arrangoiz MS, MD, FACS a surgical oncologist and is a member of Sociedad Quirúrgica S.C at the America British Cowdray Medical Center in Mexico City:
-
He is an expert in the management of breast cancer.
-
If you have any questions about SLNM and SLNB after neoadjuvant chemotherapy in breast cancer please fill free to contact Dr. Arrangoiz.
-
Training:
• General surgery:
• Michigan State University:
• 2004 al 2010
• Surgical Oncology / Head and Neck Surgery / Endocrine Surgery:
• Fox Chase Cancer Center (Filadelfia):
• 2010 al 2012
• Masters in Science (Clinical research for health professionals):
• Drexel University (Filadelfia):
• 2010 al 2012
• Surgical Oncology / Head and Neck Surgery / Endocrine Surgery:
• IFHNOS / Memorial Sloan Kettering Cancer Center:
• 2014 al 2016
#Arrangoiz
#Surgeon
#Cirujano
#SurgicalOncologist
#CirujanoOncologo
#BreastSurgeon
#CirujanodeMama
#CancerSurgeon
#CirujanodeCancer
http://www.sociedadquirurigca.com










